Advanced Manufacturing of Pyrazole-Flavonoid Derivatives Using Novel BTC Vilsmeier Chemistry
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic routes that balance high purity with environmental sustainability. Patent CN101759702A introduces a significant advancement in the preparation of flavonoid derivatives containing pyrazole structural units, which are critical scaffolds in medicinal chemistry known for their diverse biological activities including anti-cancer and anti-inflammatory properties. This technology leverages a novel application of bis(trichloromethyl) carbonate (BTC) combined with N,N-dimethylformamide (DMF) to generate a Vilsmeier reagent in situ, effectively replacing the traditionally hazardous phosphorus oxychloride (POCl3). By shifting away from phosphorus-based reagents, this method addresses growing regulatory pressures regarding phosphorus discharge while maintaining excellent reaction yields under mild thermal conditions. For R&D teams and procurement specialists, understanding this transition is vital for securing a reliable pharmaceutical intermediate supplier capable of delivering high-quality materials without the ecological baggage of legacy processes.
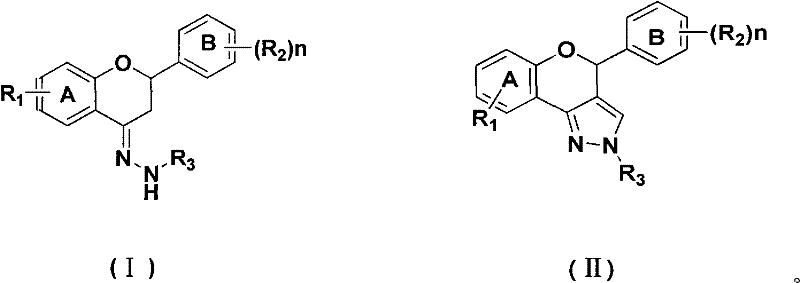
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of heterocyclic-fused flavonoids has relied heavily on the classic Vilsmeier-Haack reaction utilizing phosphorus oxychloride (POCl3) and DMF. While chemically effective, this conventional approach presents severe operational and environmental drawbacks that complicate large-scale manufacturing. The reaction generates substantial quantities of phosphoric acid as a byproduct, creating a highly acidic waste stream that requires complex and costly neutralization and disposal procedures to meet modern environmental standards. Furthermore, POCl3 is extremely corrosive, necessitating the use of specialized glass-lined or Hastelloy reactors to prevent equipment degradation, which drives up capital expenditure and maintenance overheads. The harsh acidic conditions can also lead to the decomposition of sensitive functional groups on the flavonoid scaffold, resulting in lower selectivity and a more difficult purification process to remove phosphorus-containing impurities from the final active pharmaceutical ingredient.
The Novel Approach
The methodology described in patent CN101759702A offers a transformative solution by employing bis(trichloromethyl) carbonate (BTC), also known as triphosgene, as a safe and solid alternative to liquid phosgene or corrosive phosphorus halides. In this innovative route, BTC reacts with DMF to form the active chloroiminium species required for cyclization without introducing phosphorus into the reaction matrix. This fundamental change in reagent chemistry eliminates the formation of phosphoric acid, drastically simplifying the downstream workup and wastewater treatment protocols. The process operates under significantly milder conditions, typically ranging from 30°C to 80°C, which enhances the stability of the product and reduces energy consumption. For manufacturers, this represents a strategic shift towards greener chemistry that aligns with global sustainability goals while improving the overall economic efficiency of producing complex pyrazole-flavonoid intermediates.
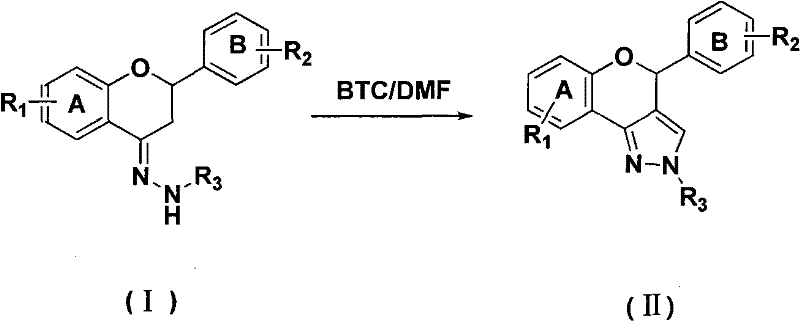
Mechanistic Insights into BTC-Mediated Vilsmeier Cyclization
The core of this synthetic breakthrough lies in the generation of the Vilsmeier reagent through the interaction of BTC and DMF. Mechanistically, BTC serves as a source of phosgene equivalents upon thermal decomposition or nucleophilic attack, which then reacts with the carbonyl oxygen of DMF to form a highly electrophilic chloroiminium ion. This activated species attacks the electron-rich positions of the flavanone-4-hydrazone substrate, initiating a cascade of cyclization and dehydration steps that construct the fused pyrazole ring system. Unlike the POCl3 pathway, which often leads to over-chlorination or phosphorylation side reactions, the BTC-mediated pathway is cleaner and more selective. The absence of strong Lewis acidic phosphorus species minimizes the risk of skeletal rearrangement or degradation of the sensitive hydrazone linkage, ensuring that the structural integrity of the flavonoid core is preserved throughout the transformation.
From an impurity control perspective, this mechanism offers distinct advantages for quality assurance teams. Traditional methods often leave trace amounts of organophosphorus compounds that are notoriously difficult to purge to ppm levels required for drug substances. By utilizing a carbon-based leaving group (carbonate/chloride) instead of phosphate, the impurity profile is significantly simplified, consisting mainly of organic byproducts that are more easily separated via standard extraction and chromatography techniques. The patent specifies a molar ratio of substrate to BTC to DMF ranging from 1:0.6-2:2.4-7.2, allowing for precise tuning of the reaction kinetics to maximize conversion while minimizing excess reagent waste. This level of control is essential for achieving the stringent purity specifications demanded by regulatory bodies for clinical trial materials and commercial API production.
How to Synthesize Pyrazolo-Flavonoid Derivatives Efficiently
The practical execution of this synthesis involves a straightforward two-stage protocol that is amenable to both laboratory scale-up and industrial manufacturing. Initially, the Vilsmeier reagent is prepared by dissolving BTC in a solvent such as dichloromethane or dichloroethane at low temperatures (0-5°C) and adding it to DMF, followed by warming to room temperature to ensure complete activation. Subsequently, the flavanone-4-hydrazone precursor is introduced, and the mixture is heated to promote cyclization. The detailed standardized synthesis steps are outlined below to guide process engineers in replicating these results with high fidelity.
- Preparation of Vilsmeier Reagent: Dissolve bis(trichloromethyl) carbonate in an organic solvent like dichloromethane at 0-5°C and add dropwise to diluted DMF, then warm to room temperature.
- Cyclization Reaction: Add the flavanone-4-hydrazone compound to the prepared reagent at 0-5°C, then heat the mixture to 30-80°C for 2-6 hours to facilitate ring closure.
- Workup and Purification: Quench the reaction in ice water, neutralize with sodium bicarbonate, extract with organic solvent, and purify the residue via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this BTC-based technology translates into tangible operational improvements beyond mere chemical elegance. The elimination of phosphorus reagents removes a major bottleneck in waste management, allowing facilities to operate with greater flexibility and reduced environmental compliance risks. Additionally, the use of solid BTC improves logistics and storage safety compared to handling corrosive liquid POCl3, streamlining the inbound supply chain for raw materials. These factors collectively contribute to a more resilient manufacturing process that can adapt quickly to market demands without being hindered by hazardous material restrictions or complex effluent treatment requirements.
- Cost Reduction in Manufacturing: The substitution of phosphorus oxychloride with bis(trichloromethyl) carbonate leads to significant cost optimization by removing the need for expensive corrosion-resistant reactor linings and extensive neutralization chemicals. Since the process avoids the generation of phosphoric acid waste, the associated costs for hazardous waste disposal and wastewater treatment are drastically simplified, resulting in substantial operational savings. Furthermore, the milder reaction conditions reduce energy consumption for heating and cooling cycles, contributing to a lower overall cost of goods sold (COGS) for the final intermediate.
- Enhanced Supply Chain Reliability: BTC is a widely available commodity chemical with a stable global supply chain, reducing the risk of raw material shortages that can plague specialized phosphorus reagents. The robustness of the reaction conditions means that production schedules are less likely to be disrupted by equipment failures caused by corrosion, ensuring consistent on-time delivery for downstream customers. This reliability is crucial for maintaining continuous production lines in the fast-paced pharmaceutical sector where delays can have cascading effects on drug development timelines.
- Scalability and Environmental Compliance: The process demonstrates excellent scalability, having been validated across various substrate derivatives with consistent yields, making it suitable for commercial scale-up of complex pharmaceutical intermediates. The environmentally friendly nature of the reaction, characterized by the absence of persistent organic pollutants like phosphates, ensures full compliance with increasingly strict international environmental regulations. This green chemistry profile enhances the marketability of the product to eco-conscious partners and facilitates easier permitting for new manufacturing capacity expansions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity for stakeholders evaluating this technology for potential integration into their supply chains.
Q: What is the primary environmental advantage of using BTC over POCl3 in this synthesis?
A: The use of bis(trichloromethyl) carbonate (BTC) eliminates the generation of phosphoric acid byproducts, thereby avoiding severe organic phosphorus pollution and reducing the burden on wastewater treatment systems compared to traditional phosphorus oxychloride methods.
Q: What are the optimal reaction conditions for the cyclization step?
A: The reaction typically proceeds by adding the substrate at 0-5°C followed by heating to a range of 30-80°C for a duration of 2 to 6 hours, ensuring mild conditions that preserve sensitive functional groups while achieving high conversion.
Q: How does this method impact equipment maintenance costs?
A: By replacing highly corrosive phosphorus oxychloride with the milder BTC-based reagent system, the process significantly reduces corrosion risks to reactor vessels and piping, leading to lower long-term maintenance costs and extended equipment lifespan.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrazolo-Flavonoid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the evolving needs of the global pharmaceutical industry. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of pyrazolo-flavonoid derivatives meets the highest international standards for safety and efficacy.
We invite potential partners to engage with our technical procurement team to discuss how this BTC-mediated technology can be tailored to your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this greener synthesis route. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and innovation in your drug development pipeline.
