Scalable Iron-Catalyzed Synthesis of Beta-Aryl-Alpha-Naphthol Intermediates for Global Supply Chains
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and economically viable synthetic routes for complex aromatic scaffolds. A significant breakthrough in this domain is detailed in Chinese Patent CN111704531B, which discloses a novel method for synthesizing β-aryl-α-naphthol compounds. These structural motifs are pivotal in the development of bioactive molecules, including natural products with antimalarial and antileishmanial properties, as well as key intermediates for functional materials and chiral ligands. The patent introduces a transformative approach that utilizes inexpensive and earth-abundant iron catalysts to facilitate a domino dehydrocyclization reaction. This methodology stands in stark contrast to traditional reliance on scarce and toxic noble metals, offering a pathway that is not only environmentally benign but also highly efficient in terms of atom economy and operational simplicity.
For R&D directors and process chemists, the implications of this technology are profound. The ability to construct the naphthol core in a single step from readily available deoxybenzoin and alkyne precursors eliminates multiple synthetic operations. This reduction in step count directly correlates to improved overall yield and reduced waste generation. Furthermore, the reaction demonstrates exceptional substrate tolerance, accommodating various substituents such as halogens, alkoxy groups, and esters without compromising efficiency. As global supply chains face increasing pressure to reduce dependency on critical raw materials, adopting iron-catalyzed processes represents a strategic move towards sustainability and supply security.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of β-aryl-α-naphthol derivatives has relied heavily on transition metal-catalyzed cross-coupling reactions, most notably the Suzuki-Miyaura coupling. While effective, these conventional methods suffer from several inherent drawbacks that hinder large-scale commercial adoption. Primarily, they necessitate the use of precious metal catalysts such as palladium, rhodium, or ruthenium, which are not only exorbitantly expensive but also subject to volatile market pricing and supply constraints. The patent background explicitly notes that noble metal resources are scarce, and their usage introduces significant toxicity concerns, requiring rigorous and costly purification steps to meet stringent pharmaceutical residual metal limits. Additionally, traditional routes often demand pre-functionalization of the substrate, involving the installation of protecting groups on phenolic hydroxyls and the synthesis of unstable or toxic aryl halides. These extra steps increase the material footprint, extend production lead times, and lower the overall atom economy of the process.
The Novel Approach
The methodology described in CN111704531B offers a paradigm shift by employing a domino dehydrocyclization strategy driven by iron catalysis. This novel approach bypasses the need for pre-functionalized halides and protecting groups, allowing for the direct coupling of deoxybenzoin compounds with alkynes. The use of ferric oxide (Fe2O3) as the catalyst is particularly advantageous; it is cheap, non-toxic, and biocompatible, addressing both cost and environmental safety concerns simultaneously. The reaction proceeds under relatively mild conditions in xylene at 135°C under an inert atmosphere, delivering high yields with complete regioselectivity. This streamlined process not only simplifies the operational workflow but also enhances the purity profile of the final product by minimizing side reactions associated with complex multi-step sequences.
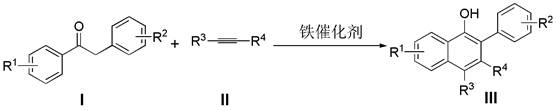
The visual representation of the general reaction scheme underscores the elegance of this transformation. It illustrates how diverse substituents on both the deoxybenzoin and alkyne components are tolerated, leading to a wide array of functionalized naphthol derivatives. This versatility is crucial for medicinal chemistry campaigns where rapid exploration of structure-activity relationships (SAR) is required. By eliminating the bottlenecks associated with noble metal catalysis and complex substrate preparation, this iron-catalyzed route provides a reliable foundation for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Fe2O3-Catalyzed Domino Dehydrocyclization
From a mechanistic perspective, the iron-catalyzed domino dehydrocyclization represents a sophisticated orchestration of C-H activation and cyclization events. Although the precise catalytic cycle may involve multiple oxidation states of iron, the overarching process facilitates the construction of the aromatic naphthol ring system through a concerted sequence. The iron catalyst likely activates the alkyne moiety, promoting nucleophilic attack by the carbonyl or enol form of the deoxybenzoin substrate. This initial interaction triggers a cascade of intramolecular cyclization and subsequent dehydrogenation steps, ultimately aromatizing the system to form the stable naphthol core. The use of ferric oxide, a stable and easily handled solid, suggests a heterogeneous or semi-heterogeneous mechanism that may simplify catalyst recovery and recycling, although the patent describes its use in a homogeneous-like manner within the solvent matrix.
Impurity control is a critical aspect of this mechanism, particularly given the potential for polymerization of alkynes or over-oxidation of the substrate. The patent data indicates that the reaction exhibits complete regioselectivity, which is a testament to the specific interaction between the iron center and the substrates. This high selectivity ensures that the desired β-aryl-α-naphthol isomer is formed exclusively, avoiding the generation of regioisomers that would be challenging to separate via standard chromatography or crystallization. Furthermore, the reaction conditions—specifically the use of an inert argon atmosphere and controlled temperature—prevent oxidative degradation of sensitive functional groups. This robustness allows for the incorporation of electron-withdrawing and electron-donating groups alike, maintaining high fidelity in the final product structure.
How to Synthesize Beta-Aryl-Alpha-Naphthol Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific operational parameters to ensure reproducibility and safety. The general procedure involves charging a reaction vessel with the deoxybenzoin substrate and the ferric oxide catalyst, followed by a rigorous degassing protocol to establish an oxygen-free environment. The addition of xylene as the solvent and the alkyne reactant is performed under inert gas flow to maintain anaerobic conditions throughout the process. The mixture is then heated to reflux temperatures, typically around 135°C, and stirred for an extended period, usually up to 24 hours, to drive the reaction to completion. Monitoring via thin-layer chromatography (TLC) is essential to determine the exact endpoint, ensuring maximum conversion before workup. Detailed standardized synthesis steps are provided in the guide below.
- Charge a reaction vessel with deoxybenzoin compound and ferric oxide catalyst under inert atmosphere.
- Add xylene solvent and alkyne substrate, then seal and purge with argon to ensure oxygen-free conditions.
- Heat the mixture to 135°C for approximately 20-24 hours, monitoring progress via TLC until completion.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this iron-catalyzed methodology offers tangible strategic benefits that extend beyond mere technical feasibility. The primary driver for adoption is the drastic reduction in raw material costs associated with the catalyst system. By replacing palladium or rhodium with ferric oxide, manufacturers can insulate their production costs from the volatility of the precious metals market. Moreover, the elimination of complex substrate pre-functionalization reduces the number of starting materials required, simplifying the supply chain and reducing the risk of delays caused by the sourcing of specialized reagents. The simplicity of the workup and purification process further contributes to operational efficiency, potentially reducing solvent consumption and waste disposal costs.
- Cost Reduction in Manufacturing: The economic impact of switching from noble metals to iron cannot be overstated. As highlighted in the patent background, iron is approximately one-five-thousandth the price of palladium, representing a fundamental shift in the cost structure of the synthesis. This substitution eliminates the need for expensive ligands often required for noble metal catalysis and removes the costly downstream processing steps necessary to remove trace heavy metals to ppm levels. Consequently, the overall cost of goods sold (COGS) for the final naphthol intermediate is significantly lowered, enhancing profit margins or allowing for more competitive pricing in the marketplace.
- Enhanced Supply Chain Reliability: Relying on earth-abundant metals like iron mitigates the geopolitical and supply risks associated with platinum group metals, which are often sourced from limited geographic regions. Iron oxide is a commodity chemical with a stable and robust global supply network, ensuring consistent availability and preventing production stoppages due to catalyst shortages. Additionally, the use of common solvents like xylene and commercially available deoxybenzoins and alkynes means that the entire bill of materials is composed of readily accessible commodities, further strengthening supply chain resilience against market disruptions.
- Scalability and Environmental Compliance: The environmental profile of this process aligns perfectly with modern green chemistry principles and increasingly strict regulatory frameworks. The absence of toxic heavy metals simplifies wastewater treatment and reduces the environmental footprint of the manufacturing facility. The high atom economy of the domino reaction means less waste is generated per kilogram of product, lowering disposal costs. Furthermore, the operational simplicity—heating a mixture in a standard solvent—makes the process highly amenable to scale-up from gram to ton quantities without requiring specialized high-pressure equipment or cryogenic conditions, facilitating rapid technology transfer to commercial production sites.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this iron-catalyzed synthesis. These answers are derived directly from the experimental data and technical disclosures within the patent documentation, providing clarity on reaction scope, catalyst performance, and product quality. Understanding these details is essential for evaluating the feasibility of integrating this technology into existing manufacturing portfolios.
Q: What are the primary advantages of using iron catalysts over palladium for naphthol synthesis?
A: Iron catalysts offer significant cost reductions as iron is abundant and roughly one-five-thousandth the price of palladium. Additionally, iron is biocompatible and eliminates the need for complex heavy metal removal steps required for pharmaceutical grades.
Q: Does this method require pre-functionalization of the substrates?
A: No, unlike traditional Suzuki coupling methods that require halide pre-functionalization and protecting groups, this iron-catalyzed domino dehydrocyclization utilizes readily available deoxybenzoins and alkynes directly.
Q: What is the regioselectivity profile of this synthesis?
A: The patent data indicates complete regioselectivity in the formation of the beta-aryl-alpha-naphthol scaffold, ensuring high purity and minimizing the formation of difficult-to-separate isomeric impurities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Aryl-Alpha-Naphthol Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the iron-catalyzed synthesis described in CN111704531B for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate this innovative academic research into robust commercial processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this cost-effective and environmentally friendly method are realized at an industrial level. We are committed to delivering high-purity beta-aryl-alpha-naphthol derivatives that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities.
We invite global partners to collaborate with us to leverage this advanced synthetic technology for their drug development pipelines. Whether you require custom synthesis of specific analogs or large-scale supply of the core scaffold, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments to understand how this iron-catalyzed approach can optimize your supply chain and reduce manufacturing costs effectively.
