Advanced Synthesis of Chiral Sulfinamide Catalysts for Scalable Pharmaceutical Manufacturing
Introduction to Next-Generation Chiral Organocatalysis
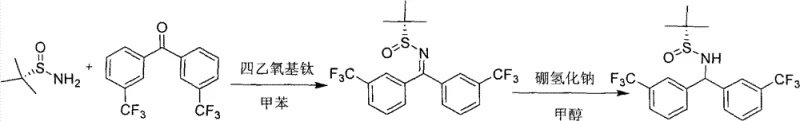
The landscape of asymmetric synthesis is undergoing a paradigm shift, driven by the urgent need for sustainable and cost-effective manufacturing processes in the pharmaceutical industry. Patent CN101863805B introduces a breakthrough methodology for the preparation of (S)-N-(bis(3-(trifluoromethyl)phenyl)methyl)-tert-butyl-2-sulfinamide, a sophisticated chiral organocatalyst designed to facilitate high-efficiency carbon-carbon bond formation. This specific molecular architecture leverages the steric bulk and electronic properties of the trifluoromethyl-substituted phenyl rings to enforce rigorous stereocontrol during catalytic cycles. Unlike traditional methods that rely on scarce transition metals, this innovation utilizes a robust sulfinamide scaffold that offers exceptional stability and ease of handling. For R&D directors and process chemists, this represents a critical advancement in accessing high-purity chiral building blocks without the regulatory burden of heavy metal clearance. The technology underscores a move towards greener chemistry, where atom economy and operational simplicity are paramount for maintaining competitive advantage in the global supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral amines and the execution of asymmetric aldol reactions have heavily depended on transition metal complexes or stoichiometric chiral auxiliaries that present significant logistical and economic challenges. Traditional protocols often necessitate the use of expensive noble metals such as palladium, rhodium, or iridium, which not only inflate raw material costs but also introduce severe contamination risks that require complex and costly purification steps to meet stringent pharmaceutical standards. Furthermore, many conventional chiral ligands are sensitive to moisture and air, demanding inert atmosphere conditions that complicate reactor operations and increase energy consumption. The disposal of metal-laden waste streams also poses environmental compliance issues, creating bottlenecks for manufacturers aiming to scale up production while adhering to increasingly strict green chemistry regulations. These cumulative factors often result in prolonged lead times and reduced overall process efficiency, hindering the rapid deployment of new drug candidates.
The Novel Approach
In stark contrast, the methodology outlined in CN101863805B presents a streamlined, two-step synthetic route that circumvents these traditional pitfalls through the strategic use of organocatalysis. The process initiates with a titanium-catalyzed condensation between readily available m-trifluoromethylbenzophenone and tert-butylsulfinamide in toluene, forming a stable sulfinylimine intermediate under reflux conditions. This is followed by a highly selective reduction using sodium borohydride in methanol, a reagent known for its safety profile and cost-effectiveness compared to complex hydrides. The resulting catalyst features a rigid chiral environment induced by the tert-butylsulfinyl group, which effectively directs the stereochemical outcome of subsequent reactions like the Aldol condensation. By eliminating the need for precious metals and utilizing commodity solvents, this novel approach drastically simplifies the supply chain requirements and enhances the economic viability of producing high-value chiral intermediates. The operational simplicity allows for seamless integration into existing manufacturing infrastructure without the need for specialized equipment.
Mechanistic Insights into Sulfinamide-Mediated Asymmetric Catalysis
The efficacy of (S)-N-(bis(3-(trifluoromethyl)phenyl)methyl)-tert-butyl-2-sulfinamide lies in its unique ability to activate substrates through a combination of steric hindrance and hydrogen bonding interactions. The bulky bis(3-trifluoromethylphenyl)methyl group creates a well-defined chiral pocket that restricts the approach of nucleophiles to the electrophilic center, thereby ensuring high enantioselectivity. The electron-withdrawing nature of the trifluoromethyl groups further modulates the electronic density of the aromatic rings, enhancing the acidity of the N-H proton and strengthening hydrogen bond donation to the substrate. This dual activation mechanism allows the catalyst to organize the transition state with precision, favoring the formation of one enantiomer over the other with remarkable fidelity. For process developers, understanding this mechanistic nuance is crucial for optimizing reaction parameters such as temperature and solvent polarity to maximize yield and optical purity. The robustness of the sulfinamide linkage ensures that the catalyst remains intact throughout the reaction cycle, preventing decomposition and maintaining consistent performance over extended periods.
Furthermore, the impurity profile generated during the synthesis is inherently cleaner due to the specificity of the borohydride reduction step, which selectively targets the imine functionality without affecting other sensitive groups. The use of titanium tetraethoxide as a Lewis acid promoter in the first step facilitates the dehydration process efficiently, driving the equilibrium towards the desired imine intermediate with minimal side reactions. Any unreacted starting materials can be easily removed during the aqueous workup, as the organic product exhibits distinct solubility characteristics in the toluene and ethyl acetate system. This inherent selectivity reduces the burden on downstream purification units, such as chromatography or crystallization, leading to higher overall recovery rates. For quality control teams, this translates to a more predictable and manageable impurity spectrum, simplifying the validation process for regulatory filings and ensuring batch-to-batch consistency essential for commercial supply.
How to Synthesize (S)-N-(bis(3-(trifluoromethyl)phenyl)methyl)-tert-butyl-2-sulfinamide Efficiently
The synthesis protocol detailed in the patent provides a reproducible framework for generating this high-performance catalyst, emphasizing the importance of precise stoichiometry and temperature control to achieve optimal results. The initial condensation step requires careful monitoring of the reflux progress to ensure complete conversion of the ketone to the imine, indicated by the formation of a yellow solid precipitate. Subsequent reduction must be conducted under controlled conditions, typically in an ice-water bath, to manage the exothermic nature of the borohydride reaction and prevent thermal degradation of the chiral center. Detailed standardized operating procedures for this synthesis are critical for maintaining the integrity of the chiral information and ensuring safety during scale-up operations. The following guide outlines the critical process parameters derived from the patent examples to assist technical teams in implementing this route effectively.
- Condense m-trifluoromethylbenzophenone with tert-butylsulfinamide in toluene using catalytic titanium tetraethoxide under reflux.
- Quench the reaction with saturated sodium bicarbonate, filter solids, and isolate the sulfinylimine intermediate.
- Reduce the intermediate imine using sodium borohydride in methanol to yield the final chiral sulfinamide catalyst.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the adoption of this organocatalytic technology offers profound strategic benefits by decoupling production from the volatile markets of precious metals. The reliance on commodity chemicals such as toluene, methanol, and sodium borohydride ensures a stable and resilient supply chain that is less susceptible to geopolitical disruptions or mining shortages. This shift not only mitigates the risk of raw material scarcity but also provides significant leverage in negotiating pricing with suppliers, as these inputs are produced at massive global scales with established logistics networks. For supply chain heads, the predictability of lead times is vastly improved, allowing for more accurate inventory planning and reduced safety stock requirements. The elimination of complex metal scavenging steps further accelerates the manufacturing cycle time, enabling faster response to market demands and shorter time-to-market for new pharmaceutical products.
- Cost Reduction in Manufacturing: The economic model of this process is fundamentally superior due to the complete avoidance of expensive transition metal catalysts and ligands, which traditionally account for a substantial portion of COGS in asymmetric synthesis. By replacing these high-cost inputs with inexpensive organic precursors and simple inorganic reductants, manufacturers can achieve drastic reductions in direct material expenses. Additionally, the simplified workup procedure reduces the consumption of auxiliary materials like filtration aids and chromatography media, further lowering operational expenditures. The ability to recover and recycle solvents like toluene adds another layer of cost efficiency, making the overall process highly competitive for large-scale commercial production. These savings can be reinvested into R&D or passed on to customers to enhance market positioning.
- Enhanced Supply Chain Reliability: The robustness of the synthetic route ensures that production schedules are not compromised by the sensitivity issues often associated with air- or moisture-sensitive metal catalysts. The reagents used are shelf-stable and widely available from multiple qualified vendors, reducing the risk of single-source dependency. This diversification of the supply base enhances business continuity and allows for greater flexibility in sourcing strategies. Furthermore, the straightforward nature of the reaction conditions minimizes the likelihood of batch failures due to operational deviations, ensuring a consistent flow of high-quality product to downstream users. This reliability is critical for maintaining long-term contracts with major pharmaceutical clients who demand unwavering supply security.
- Scalability and Environmental Compliance: Scaling this process from laboratory to industrial production is facilitated by the use of standard unit operations such as reflux, filtration, and distillation, which are well-understood and easily engineered. The absence of toxic heavy metals simplifies waste treatment protocols, reducing the environmental footprint and associated disposal costs. This alignment with green chemistry principles not only satisfies regulatory requirements but also enhances the corporate sustainability profile, which is increasingly important for stakeholders and investors. The process generates minimal hazardous waste, and the solvents employed are amenable to efficient recovery systems, supporting a circular economy approach within the manufacturing facility. This scalability ensures that the technology can meet growing market demands without compromising on quality or compliance standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this sulfinamide catalyst technology, drawing directly from the experimental data and claims presented in the patent literature. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing portfolios. The answers provided reflect the specific capabilities and limitations observed during the development of this methodology, offering a realistic perspective on its industrial potential. We encourage stakeholders to review these details carefully to assess the fit for their specific application requirements.
Q: What are the key advantages of this sulfinamide catalyst over transition metal catalysts?
A: This organocatalyst eliminates the need for expensive and toxic transition metals like palladium or rhodium, significantly reducing heavy metal residue concerns in API manufacturing and simplifying downstream purification processes.
Q: What is the expected yield for the catalyst synthesis described in CN101863805B?
A: The patent documentation indicates a robust two-step total yield ranging from 70% to 80%, demonstrating high efficiency and material throughput suitable for industrial scaling.
Q: Can this catalyst be used for large-scale Aldol reactions?
A: Yes, the technology has been validated for Aldol condensations with yields reaching up to 85%, utilizing common solvents like toluene and methanol which are easily managed in large-scale reactor systems.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Organocatalyst Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced organocatalysis in modern drug discovery and development, and we are committed to bridging the gap between academic innovation and industrial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory techniques like the one described in CN101863805B can be successfully translated into robust manufacturing processes. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee the quality of every batch. Our expertise in handling sensitive chiral molecules allows us to navigate the complexities of asymmetric synthesis with precision, delivering products that meet the highest international standards for pharmaceutical intermediates.
We invite you to collaborate with us to explore how this cost-effective catalytic technology can optimize your supply chain and reduce your overall manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your decision-making process. Let us be your partner in achieving sustainable growth and operational excellence in the competitive landscape of fine chemical manufacturing.
