Advanced Manufacturing of Rosuvastatin Sodium: A Novel Synthetic Route for Global Pharma Supply Chains
Advanced Manufacturing of Rosuvastatin Sodium: A Novel Synthetic Route for Global Pharma Supply Chains
The pharmaceutical industry constantly seeks more efficient, safer, and cost-effective pathways for producing high-value active pharmaceutical ingredients (APIs) and their precursors. Patent CN109574938B, published in April 2022, introduces a groundbreaking method for synthesizing Rosuvastatin Sodium, a critical precursor to the widely prescribed lipid-lowering agent Rosuvastatin Calcium. This technology addresses long-standing challenges in statin synthesis, specifically targeting the issues of harsh reaction conditions, expensive reagents, and poor stereoselectivity that have plagued previous generations of manufacturing processes. By leveraging a novel coupling strategy involving trimercapto-s-triazine and a refined Julia olefination sequence, this patent offers a robust framework for producing high-purity intermediates suitable for global supply chains.
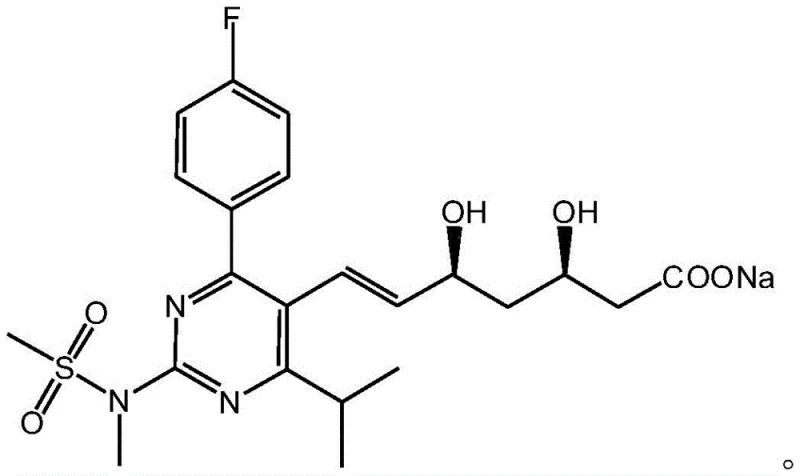
For R&D directors and process chemists, the significance of this patent lies in its ability to bypass the limitations of classical Wittig and Horner-Wadsworth-Emmons reactions, which often require cryogenic temperatures and generate significant waste. The disclosed method not only improves atom economy but also streamlines the purification process, resulting in a final product with exceptional purity profiles. As we delve deeper into the technical specifics, it becomes clear that this route represents a substantial leap forward in the commercial scale-up of complex pharmaceutical intermediates, offering a viable solution for manufacturers aiming to optimize their production lines while adhering to stringent environmental and safety standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical approaches to synthesizing Rosuvastatin Sodium have been fraught with significant technical and economic hurdles that hinder efficient large-scale production. For instance, early methodologies described in European patent EP0521471 relied heavily on Wittig olefination reactions that necessitated the use of hydrofluoric acid (HF) for deprotection steps. The corrosive nature of HF poses severe risks to reactor integrity and requires specialized, expensive equipment lining, thereby driving up capital expenditure. Furthermore, other routes, such as those disclosed in WO0049014 and WO2004052867, demanded extremely low reaction temperatures, often reaching -75°C, to achieve acceptable stereoselectivity. Maintaining such cryogenic conditions on an industrial scale is energy-intensive and operationally complex, leading to increased utility costs and potential batch inconsistencies. Additionally, many of these legacy routes utilized side-chain starting materials that were either difficult to source or prohibitively expensive, creating bottlenecks in the reliable API intermediate supplier network and compromising supply chain continuity.
The Novel Approach
In stark contrast to these cumbersome legacy methods, the synthesis route detailed in CN109574938B introduces a streamlined and chemically elegant solution that mitigates these risks through innovative intermediate design. The core of this novelty lies in the initial coupling of a bromomethyl-pyrimidine derivative with trimercapto-s-triazine, followed by a controlled oxidation to generate a stable sulfone intermediate (Substance B). This strategic shift allows for a subsequent Julia olefination reaction that proceeds with high stereoselectivity under much milder thermal conditions, typically ranging from -60°C to 0°C, which is significantly more manageable than the -75°C required by prior art. The elimination of hazardous HF deprotection and the use of readily available oxidants like ammonium molybdate or m-CPBA drastically simplify the operational workflow. This new pathway not only enhances the overall yield but also ensures that the final Rosuvastatin Sodium possesses the rigorous purity specifications demanded by regulatory bodies, making it an ideal candidate for cost reduction in pharmaceutical intermediates manufacturing.
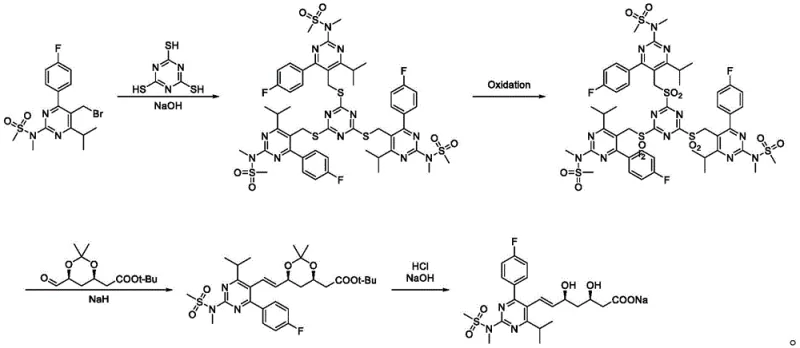
Mechanistic Insights into Trimercapto-s-triazine Coupling and Julia Olefination
The chemical elegance of this patented process is rooted in its precise control over reaction mechanisms to maximize efficiency and minimize impurity formation. The synthesis begins with a nucleophilic substitution where 5-bromomethyl-4-(4-fluorophenyl)-6-isopropyl-2-[methyl(methylsulfonyl)amino]pyrimidine reacts with trimercapto-s-triazine under sodium hydroxide catalysis. This step is critical as it installs the sulfur-containing scaffold necessary for the subsequent olefination, forming Substance A with high fidelity. The reaction is conducted in alcohol solvents at moderate temperatures (10-50°C), which facilitates rapid kinetics without promoting degradation of the sensitive pyrimidine core. Following this, the oxidation of Substance A to Substance B is meticulously managed using oxidants such as a mixture of ammonium molybdate tetrahydrate and hydrogen peroxide. This oxidation system is chosen for its ability to selectively convert the sulfide linkages into sulfones without over-oxidizing other functional groups, a common pitfall in less selective oxidation protocols. The resulting sulfone (Substance B) serves as a potent nucleophile precursor for the pivotal Julia olefination step.
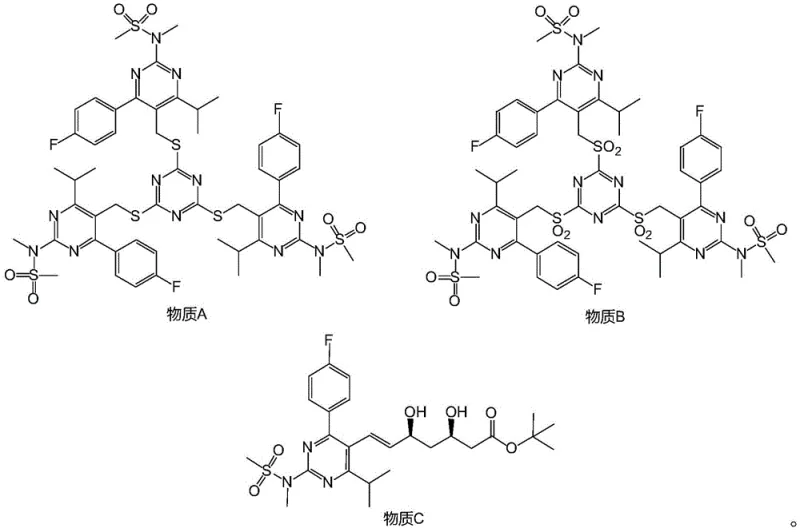
The final assembly of the carbon skeleton occurs through the reaction of Substance B with a chiral aldehyde ester, specifically (4R-Cis)-6-aldehyde-2,2-dimethyl-1,3-dioxane-4-tert-butyl acetate, mediated by sodium hydride. This Julia olefination is renowned for its ability to establish the crucial trans-double bond geometry found in the statin side chain with excellent stereocontrol. The mechanism involves the formation of a beta-hydroxy sulfone intermediate which undergoes elimination to yield the desired alkene. By optimizing the molar ratios of sodium hydride and the aldehyde component, the process minimizes the formation of cis-isomers and other diastereomeric impurities. This high level of stereochemical purity is paramount for the biological activity of the final drug, ensuring that the high-purity pharmaceutical intermediates produced meet the strict enantiomeric excess requirements necessary for clinical efficacy. The subsequent deprotection and hydrolysis steps are equally refined, utilizing mild acidic and basic conditions to reveal the final carboxylic acid salt without compromising the integrity of the sensitive fluorophenyl and pyrimidine moieties.
How to Synthesize Rosuvastatin Sodium Efficiently
Implementing this synthesis route in a production environment requires strict adherence to the optimized parameters outlined in the patent to ensure reproducibility and safety. The process is divided into four distinct stages: the formation of the trithiol-linked intermediate, the oxidative conversion to the sulfone, the stereoselective coupling with the chiral side chain, and the final deprotection and salt formation. Each stage has been fine-tuned to balance reaction rate with impurity control, utilizing common solvents like isopropanol, tetrahydrofuran, and ethyl acetate to facilitate easy workup and solvent recovery. The detailed standardized synthesis steps below provide a roadmap for process chemists to replicate the high yields and purity levels reported in the patent examples, serving as a foundational guide for reducing lead time for high-purity pharmaceutical intermediates in your own facilities.
- React 5-bromomethyl-4-(4-fluorophenyl)-6-isopropyl-2-[methyl(methylsulfonyl)amino]pyrimidine with trimercapto-s-triazine under NaOH catalysis to form Substance A.
- Oxidize Substance A using ammonium molybdate/hydrogen peroxide or m-CPBA to obtain the sulfone intermediate, Substance B.
- Perform Julia olefination between Substance B and a chiral aldehyde ester using NaH, followed by deprotection and hydrolysis to yield Rosuvastatin Sodium.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this novel synthesis route offers transformative benefits that directly impact the bottom line and operational resilience. Traditional manufacturing methods for Rosuvastatin Sodium often rely on exotic reagents and extreme conditions that introduce volatility into the supply chain, leading to unpredictable lead times and inflated costs. By shifting to a process that utilizes commodity chemicals and operates under mild conditions, manufacturers can significantly stabilize their raw material sourcing strategies. The elimination of corrosive acids like HF not only reduces the need for specialized containment infrastructure but also lowers the long-term maintenance costs associated with reactor refurbishment. Furthermore, the improved atom economy of this route means that less raw material is wasted as byproduct, translating to a more sustainable and cost-efficient production model that aligns with modern green chemistry initiatives.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the replacement of expensive and hazardous reagents with more economical alternatives. By avoiding the use of hydrofluoric acid and cryogenic cooling systems that demand excessive energy consumption, the overall utility and material costs are substantially reduced. The simplified purification steps, which rely on standard extraction and recrystallization techniques rather than complex chromatographic separations, further decrease the operational expenditure per kilogram of product. This efficiency gain allows for a more competitive pricing structure without sacrificing quality, providing a distinct advantage in the global market for cardiovascular drug intermediates.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the reliance on single-source suppliers for niche chiral reagents or hazardous materials. This patented method mitigates such risks by utilizing widely available starting materials such as trimercapto-s-triazine and standard oxidants. The robustness of the reaction conditions, which tolerate a broader range of temperatures compared to the fragile -75°C protocols of the past, ensures that production schedules are less susceptible to disruptions caused by equipment failures or environmental fluctuations. This reliability is crucial for maintaining consistent inventory levels and meeting the rigorous delivery commitments expected by major pharmaceutical clients.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to multi-ton production often reveals hidden inefficiencies, but this route is inherently designed for scalability. The use of benign solvents and the generation of less hazardous waste streams simplify the environmental compliance burden, reducing the costs associated with waste treatment and disposal. The high yield and selectivity of the Julia olefination step mean that fewer batches need to be reprocessed or discarded due to quality failures, maximizing the throughput of existing manufacturing assets. This combination of scalability and environmental stewardship positions the technology as a future-proof solution for long-term commercial production.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of a new synthesis pathway is essential for making informed procurement and development decisions. The following questions address common inquiries regarding the practical implementation, quality control, and strategic benefits of the Rosuvastatin Sodium synthesis method described in CN109574938B. These answers are derived directly from the patent's experimental data and technical disclosures, providing a transparent view of the technology's capabilities and limitations for potential partners and stakeholders.
Q: How does this new synthesis route improve upon traditional methods involving HF acid?
A: Traditional routes often utilize hazardous hydrofluoric acid (HF) for deprotection, which causes severe equipment corrosion and safety risks. The patented method (CN109574938B) eliminates the need for HF by employing a trimercapto-s-triazine coupling strategy followed by mild oxidation, significantly enhancing operational safety and reducing equipment maintenance costs.
Q: What are the stereoselectivity advantages of this Julia olefination approach?
A: The process utilizes a specific Julia olefination reaction between a sulfone intermediate and a chiral aldehyde ester under controlled temperatures (-60 to 0°C). This ensures excellent docking stereoselectivity, minimizing the formation of unwanted diastereomers and simplifying downstream purification compared to older Wittig-based routes.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the route is designed for scalability. It avoids extreme cryogenic conditions (like -75°C required in some prior art) and uses readily available raw materials. The reaction conditions are mild and controllable (10-50°C for initial steps), making it highly feasible for commercial manufacturing environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rosuvastatin Sodium Supplier
The technological advancements presented in patent CN109574938B underscore the dynamic evolution of statin intermediate manufacturing, offering a pathway that balances high performance with operational safety. At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting such innovative routes to maintain competitiveness in the global pharmaceutical landscape. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our commitment to quality is unwavering, supported by stringent purity specifications and rigorous QC labs that verify every batch against the highest international standards, guaranteeing that the intermediates we supply are ready for immediate integration into your API synthesis workflows.
We invite you to explore how our expertise in advanced organic synthesis can drive value for your organization. By leveraging our technical capabilities, you can access a Customized Cost-Saving Analysis tailored to your specific production volumes and quality requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized manufacturing processes can enhance your supply chain resilience and support your long-term strategic goals in the cardiovascular therapeutic sector.
