Advanced Photoredox Synthesis of Alpha-Difluoro-Beta-Carbonyl Sulfones for Commercial Scale-Up
Advanced Photoredox Synthesis of Alpha-Difluoro-Beta-Carbonyl Sulfones for Commercial Scale-Up
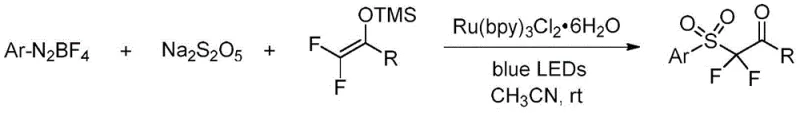
The landscape of fluorinated organic synthesis is undergoing a significant transformation, driven by the urgent need for milder, more sustainable methodologies that can support the rigorous demands of modern drug discovery. A pivotal advancement in this domain is detailed in patent CN111269153A, which discloses a highly efficient visible-light photoredox catalytic system for constructing α,α-difluoro-β-carbonyl sulfone compounds. This technology represents a paradigm shift from classical cryogenic protocols to ambient temperature processes, leveraging the power of blue LED irradiation and earth-abundant sulfur sources. For R&D directors and procurement strategists alike, this innovation offers a compelling pathway to access critical fluorinated building blocks that are essential for enhancing the metabolic stability and oral bioavailability of next-generation bioactive molecules. By utilizing simple aryl diazonium salts and sodium metabisulfite, this method circumvents the logistical nightmares associated with gaseous sulfur dioxide and super-strong bases, positioning itself as a cornerstone technique for the reliable pharmaceutical intermediate supplier seeking to optimize their portfolio.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the α,α-difluoro-β-carbonyl sulfone motif has been plagued by severe operational constraints that hinder both academic exploration and industrial adoption. The seminal work by the Hu group in 2009, while groundbreaking, necessitated the use of lithium hexamethyldisilazide (LHMDS), a potent and moisture-sensitive base, coupled with ultra-low temperature conditions reaching -98°C. Such extreme cryogenic requirements impose a massive burden on energy consumption and specialized equipment infrastructure, making cost reduction in pharmaceutical intermediate manufacturing nearly impossible at scale. Furthermore, the handling of gaseous sulfur dioxide or unstable sulfonyl precursors introduces significant safety hazards and supply chain volatility, often leading to batch-to-batch inconsistencies. The narrow functional group tolerance of these anionic pathways also restricts the structural diversity accessible to medicinal chemists, forcing them to design around synthetic limitations rather than biological optimality. Consequently, the industry has long suffered from a lack of robust, scalable methods that can deliver these high-value motifs without compromising safety or economic viability.
The Novel Approach
In stark contrast, the methodology outlined in CN111269153A introduces a radical-based strategy that operates under remarkably benign conditions, effectively dismantling the barriers erected by traditional anionic chemistry. By employing a ruthenium-based photocatalyst, specifically Ru(bpy)3Cl2·6H2O, in conjunction with 15W blue LED irradiation, the reaction proceeds smoothly at room temperature in acetonitrile. This photoredox manifold utilizes sodium metabisulfite as a convenient solid surrogate for sulfur dioxide, generating aryl sulfonyl radicals in situ that add efficiently to 2,2-difluoro enol silyl ethers. The elimination of cryogenic cooling and strong bases not only drastically simplifies the reactor setup but also enhances the safety profile, making it suitable for commercial scale-up of complex polymer additives and fine chemicals. Moreover, the system exhibits exceptional functional group compatibility, tolerating a wide array of electron-withdrawing and electron-donating substituents on both the aryl diazonium and the enol silane components, thereby expanding the chemical space available for drug design.
Mechanistic Insights into Visible-Light Photoredox Catalysis
The elegance of this transformation lies in its intricate yet efficient radical mechanism, which orchestrates the assembly of the sulfone framework through a series of single-electron transfer events. Upon irradiation with blue light, the ground state Ru(II) photocatalyst is excited to its long-lived triplet state, which possesses sufficient redox potential to facilitate the generation of reactive radical species. Concurrently, the aryl diazonium salt interacts with sodium metabisulfite under these photo-excited conditions to generate an aryl sulfonyl radical. This electrophilic radical species then undergoes a regioselective addition to the electron-rich double bond of the 2,2-difluoro enol silyl ether, forming a new carbon-sulfur bond and a transient carbon-centered radical intermediate. This step is crucial as it establishes the quaternary carbon center bearing the two fluorine atoms, a structural feature highly prized for its ability to mimic hydroxyl groups while resisting metabolic oxidation.
Following the radical addition, the resulting intermediate undergoes a single-electron oxidation mediated by the oxidized form of the photocatalyst, regenerating the ground state Ru(II) species to close the catalytic cycle. This oxidation step triggers the elimination of the trimethylsilyl group and subsequent tautomerization or hydrolysis during workup to yield the final α,α-difluoro-β-carbonyl sulfone product. The beauty of this mechanism is its reliance on mild redox potentials rather than harsh nucleophilic attacks, which inherently minimizes side reactions such as ester hydrolysis or epimerization that often plague basic conditions. From an impurity control perspective, the radical nature of the reaction ensures high chemoselectivity, as the sulfonyl radical preferentially targets the alkene over other sensitive functionalities present in complex molecular scaffolds. This mechanistic robustness translates directly to higher purity profiles and simplified downstream purification, a critical factor for meeting the stringent quality standards of the global pharmaceutical market.
How to Synthesize α,α-Difluoro-β-Carbonyl Sulfones Efficiently
Implementing this photoredox protocol in a laboratory or pilot plant setting requires careful attention to reaction parameters to maximize yield and reproducibility, though the procedure is fundamentally straightforward. The process begins with the preparation of an oxygen-free environment, typically achieved by purging the reaction vessel with high-purity nitrogen or argon, as radical intermediates can be quenched by atmospheric oxygen. The precise stoichiometry involves mixing the aryl diazonium tetrafluoroborate salt, sodium metabisulfite, and the 2,2-difluoro enol silyl ether in acetonitrile, with a catalytic loading of the ruthenium complex. Detailed standardized synthesis steps are provided in the guide below to ensure consistent results across different batches and scales.
- Prepare the reaction mixture by adding aryl diazonium salt, sodium metabisulfite, 2,2-difluoro enol silyl ether, and Ru(bpy)3Cl2·6H2O catalyst into a dry reaction vessel under inert atmosphere.
- Add acetonitrile solvent to the mixture and irradiate the solution with 15W blue LEDs at room temperature while stirring until TLC indicates complete consumption of starting materials.
- Concentrate the reaction mixture under reduced pressure and purify the crude residue via column chromatography using petroleum ether and ethyl acetate to isolate the target sulfone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this photoredox methodology offers tangible strategic benefits that extend far beyond mere chemical curiosity. The primary advantage lies in the drastic simplification of the manufacturing infrastructure; by eliminating the need for cryogenic reactors capable of maintaining -98°C, facilities can utilize standard glass-lined or stainless steel reactors equipped with simple LED arrays. This reduction in capital expenditure and energy overhead contributes to substantial cost savings in fine chemical manufacturing, allowing for more competitive pricing models in a margin-compressed market. Furthermore, the reliance on sodium metabisulfite, a commodity chemical widely used in food preservation and water treatment, ensures a stable and resilient supply chain that is immune to the geopolitical fluctuations often affecting specialized fluorinating reagents.
- Cost Reduction in Manufacturing: The economic impact of replacing cryogenic conditions with room temperature processing cannot be overstated, as it removes the energy-intensive cooling loops that dominate the utility costs of traditional fluorination processes. Additionally, the use of visible light photocatalysis avoids the need for expensive transition metal reagents or hazardous gas handling systems, further lowering the operational expenditure per kilogram of product. The high atom economy of the radical addition also means less waste generation, reducing the costs associated with waste disposal and environmental compliance. By streamlining the synthetic route to fewer steps and milder conditions, manufacturers can achieve a leaner production model that enhances overall profitability without sacrificing quality.
- Enhanced Supply Chain Reliability: Sourcing raw materials for this synthesis is exceptionally straightforward, as aryl diazonium salts can be generated in situ from readily available anilines, and sodium metabisulfite is a globally traded bulk chemical. This abundance mitigates the risk of supply disruptions that frequently occur with niche fluorinated building blocks, ensuring continuous production schedules for critical API intermediates. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, providing a buffer against supply chain volatility. Consequently, partners can rely on a consistent flow of high-purity materials, enabling just-in-time manufacturing strategies that reduce inventory holding costs and improve cash flow.
- Scalability and Environmental Compliance: Scaling photochemical reactions has historically been challenging due to light penetration issues, but the advent of high-power LED arrays and flow chemistry reactors has largely solved this bottleneck, allowing for seamless translation from gram to ton scale. The absence of volatile organic solvents beyond standard acetonitrile, and the elimination of toxic gaseous SO2, significantly improves the environmental footprint of the process, aligning with increasingly strict global regulations on emissions and worker safety. This green chemistry profile not only facilitates regulatory approval but also enhances the brand reputation of manufacturers committed to sustainable practices. The ability to run the reaction at ambient pressure and temperature further reduces the engineering controls required for scale-up, accelerating the time-to-market for new fluorinated therapeutics.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photoredox sulfone synthesis, derived directly from the experimental data and scope analysis within the patent literature. Understanding these nuances is vital for technical teams evaluating the feasibility of integrating this route into their existing production pipelines. The answers provided reflect the specific advantages of the radical mechanism over ionic alternatives, focusing on practical outcomes like yield, purity, and operational safety.
Q: What are the key advantages of this photoredox method over traditional synthesis?
A: Unlike traditional methods requiring strong bases like LHMDS and ultra-low temperatures of -98°C, this photoredox approach operates at room temperature using visible light, significantly reducing energy costs and operational complexity while maintaining broad functional group tolerance.
Q: What is the role of sodium metabisulfite in this reaction?
A: Sodium metabisulfite acts as a solid sulfur dioxide surrogate, generating aryl sulfonyl radicals in situ under blue light irradiation, which eliminates the need for handling hazardous gaseous SO2 and simplifies the reaction setup for industrial applications.
Q: Can this method be scaled for commercial production of pharmaceutical intermediates?
A: Yes, the use of commercially available photocatalysts, mild room temperature conditions, and simple workup procedures makes this route highly amenable to scale-up, offering a reliable supply chain solution for complex fluorinated intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable α,α-Difluoro-β-Carbonyl Sulfones Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced fluorinated intermediates requires more than just a patent; it demands deep process engineering expertise and a commitment to quality that spans the entire value chain. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We understand that every molecule has unique challenges, which is why our rigorous QC labs and stringent purity specifications are designed to meet the exacting demands of top-tier pharmaceutical clients who cannot afford variability in their supply of critical building blocks.
We invite you to collaborate with us to leverage this cutting-edge photoredox technology for your next project, unlocking new possibilities in drug design while optimizing your cost structure. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced manufacturing capabilities can become a strategic asset for your organization, driving innovation and efficiency in equal measure.
