Scalable Iridium-Catalyzed Synthesis of Carboxymethyl Ketones for Pharmaceutical Applications
Introduction to Advanced Carboxymethyl Ketone Synthesis
The landscape of organic synthesis for bioactive molecules is constantly evolving, driven by the need for safer, more efficient, and scalable methodologies. A significant breakthrough in this domain is documented in patent CN115974758A, which discloses a novel preparation method for carboxymethyl ketone compounds. This technology leverages an iridium-catalyzed reaction between carboxylic acid compounds and sulfur ylide compounds, successfully introducing heterocyclic structures under mild conditions. For R&D directors and procurement specialists in the fine chemical sector, this represents a pivotal shift away from hazardous traditional reagents towards a more robust catalytic system. The method addresses critical pain points in the industry, such as low yields, narrow substrate scope, and the use of excessive oxidants, offering a pathway to high-purity pharmaceutical intermediates with improved economic viability.
The strategic importance of this patent lies in its ability to generate diverse heterocyclic carboxymethyl ketones, which are privileged scaffolds in medicinal chemistry. These structures are foundational for developing anti-inflammatory, antihypertensive, and antitumor agents. By utilizing readily available carboxylic acids and stable sulfur ylides, the process simplifies the supply chain for raw materials. Furthermore, the operational simplicity—requiring standard heating and inert atmosphere conditions rather than cryogenic temperatures or high-pressure equipment—makes it an attractive candidate for immediate technology transfer and commercial adoption by reliable pharmaceutical intermediate suppliers seeking to optimize their manufacturing portfolios.
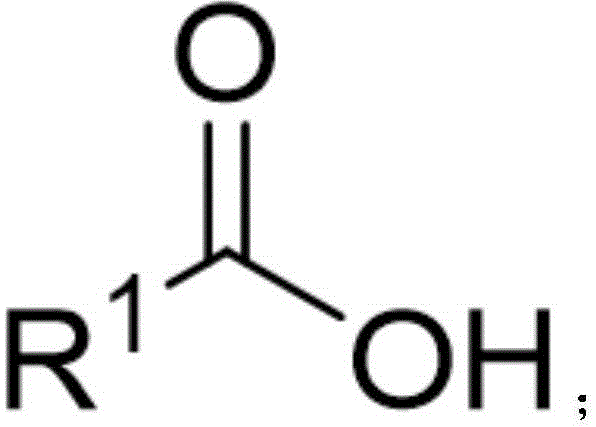
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of carboxymethyl ketones has been fraught with significant technical and safety challenges that hinder large-scale production. Traditional approaches often rely on the oxidative coupling of ketones and carboxylic acids, a process that necessitates the use of excess strong oxidants. These harsh conditions not only limit the functional group tolerance, leading to side reactions with sensitive moieties like amines or hydroxyls, but also generate substantial chemical waste, complicating environmental compliance. Another prevalent method involves the metal-catalyzed O-H bond insertion of diazo compounds. While effective in small-scale laboratory settings, diazo compounds are inherently unstable, toxic, and potentially explosive, posing unacceptable risks for commercial scale-up in pharmaceutical intermediate manufacturing.
Recent alternatives, such as gold-catalyzed oxidative coupling of terminal alkynes or ruthenium-catalyzed reactions, have attempted to mitigate some issues but often fail to accommodate heterocyclic substrates effectively. For instance, vanadium-catalyzed methods reported in prior art typically suffer from low yields, often below 55%, and exhibit poor compatibility with crucial drug-like functional groups such as indoles or alkenyl chains. These limitations result in complex purification processes, increased cost of goods sold (COGS), and extended lead times, creating a bottleneck for supply chain heads who require consistent, high-volume delivery of complex intermediates for downstream API synthesis.
The Novel Approach
The methodology outlined in patent CN115974758A offers a transformative solution by employing an iridium catalyst to facilitate the reaction between carboxylic acids and sulfur ylides. This novel approach eliminates the need for dangerous diazo reagents and excessive oxidants, fundamentally altering the safety profile of the synthesis. The reaction proceeds smoothly in common solvents like DMF or dioxane at moderate temperatures ranging from 90°C to 120°C. Crucially, this system demonstrates remarkable substrate generality, successfully synthesizing a wide array of heterocyclic carboxymethyl ketones that were previously difficult to access. The ability to tolerate diverse functional groups ensures that complex molecular architectures can be constructed without extensive protecting group strategies, thereby streamlining the synthetic route.
From a commercial perspective, this new route translates directly into cost reduction in pharmaceutical intermediate manufacturing. The high yields reported, frequently exceeding 70% and reaching up to 96% in specific embodiments, minimize raw material waste and maximize throughput. The ease of product isolation, often achievable through simple washing and extraction followed by column chromatography, reduces the burden on downstream processing units. For procurement managers, this means a more reliable source of high-purity intermediates with a lower risk of supply disruption due to safety incidents or regulatory hurdles associated with hazardous reagents. The method's robustness makes it an ideal candidate for the commercial scale-up of complex polymer additives and specialty chemicals as well.
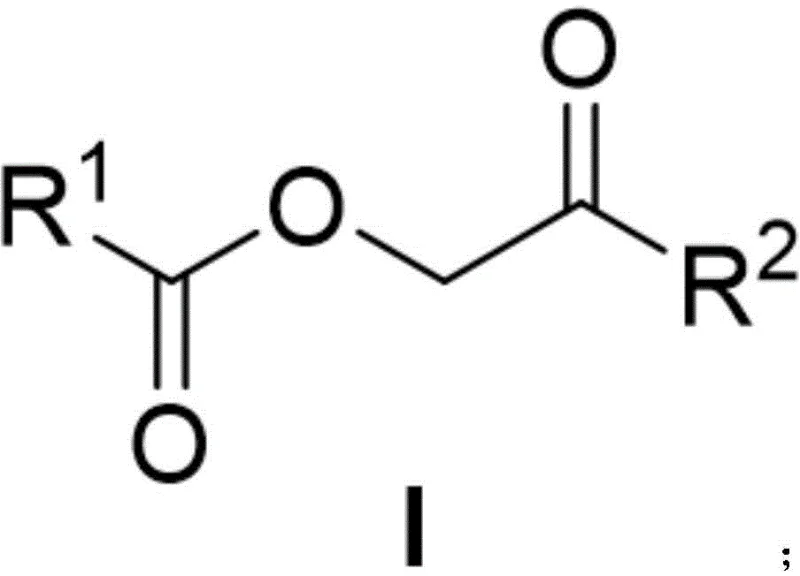
Mechanistic Insights into Iridium-Catalyzed O-H Bond Insertion
The core innovation of this technology rests on the unique reactivity of the iridium catalyst in generating metal-carbene intermediates from sulfur ylides. Unlike rhodium or copper catalysts often used in carbene chemistry, the iridium system described herein exhibits a distinct affinity for O-H bond insertion when paired with specific ligands such as 1,10-phenanthroline or triphenylphosphine. The mechanism initiates with the coordination of the sulfur ylide to the iridium center, followed by the extrusion of the sulfide group to form a reactive iridium-carbene species. This electrophilic intermediate then undergoes insertion into the O-H bond of the carboxylic acid, forming the desired ester-linked ketone structure. This pathway avoids the radical mechanisms typical of oxidative couplings, thereby preserving the integrity of oxidation-sensitive functional groups present on the substrate.
Impurity control is a critical aspect of this mechanistic pathway, particularly for R&D directors focused on purity specifications. The high selectivity of the iridium catalyst minimizes the formation of byproducts such as homocoupling dimers or over-oxidized species. The use of mild reaction conditions prevents thermal degradation of the heterocyclic cores, which is a common issue in high-temperature oxidative processes. Furthermore, the choice of ligand plays a pivotal role in tuning the electronic properties of the metal center, ensuring that the insertion occurs regioselectively at the carboxylic acid oxygen rather than at other nucleophilic sites like amines or indole nitrogens. This precise control results in a cleaner crude reaction profile, simplifying the purification process and ensuring that the final product meets the stringent quality standards required for clinical trial materials.
How to Synthesize Carboxymethyl Ketones Efficiently
Implementing this synthesis route requires careful attention to catalyst loading and reaction atmosphere to maximize efficiency. The standardized protocol involves dissolving the carboxylic acid and sulfur ylide in a polar aprotic solvent, followed by the addition of the iridium precatalyst and ligand. The detailed standardized synthesis steps see the guide below.
- Dissolve carboxylic acid compound, sulfur ylide compound, and ligand in a suitable solvent such as DMF or dioxane.
- Add the iridium catalyst (e.g., [Ir(cod)Cl]2) to the mixture under an inert nitrogen or argon atmosphere.
- Heat the reaction mixture to 90°C-120°C for 8-16 hours, then cool, wash, extract, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement managers, the adoption of this iridium-catalyzed technology offers substantial strategic advantages beyond mere chemical novelty. The primary benefit is the drastic simplification of the raw material supply chain. By replacing unstable diazo compounds with stable, commercially available sulfur ylides and carboxylic acids, the process mitigates the risks associated with storing and transporting hazardous explosives. This stability allows for just-in-time manufacturing models and reduces the need for specialized storage infrastructure, leading to significant overhead cost savings. Additionally, the broad substrate scope means that a single platform technology can be used to produce a diverse library of intermediates, consolidating vendor relationships and simplifying inventory management for multi-product facilities.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous oxidants, combined with the use of earth-abundant iridium catalysts at low loadings, drives down the direct material costs. The high atom economy of the insertion reaction ensures that a larger proportion of the starting mass ends up in the final product, reducing waste disposal fees. Furthermore, the mild reaction temperatures reduce energy consumption compared to high-heat oxidative processes, contributing to a lower carbon footprint and reduced utility costs per kilogram of product produced.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions ensures consistent batch-to-batch reproducibility, a critical factor for maintaining supply continuity. The tolerance for various functional groups means that raw material specifications can be slightly relaxed without compromising product quality, allowing procurement teams to source starting materials from a wider pool of qualified suppliers. This flexibility reduces the risk of supply bottlenecks caused by shortages of highly specialized reagents, ensuring that production schedules for critical API intermediates are met without delay.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work efficiently with diverse substrates including indoles and pyridines. The absence of heavy metal oxidants and toxic diazo byproducts simplifies wastewater treatment and aligns with increasingly strict environmental regulations. This green chemistry profile facilitates faster regulatory approval for new manufacturing sites and reduces the long-term liability associated with hazardous waste management, making it a sustainable choice for long-term commercial partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for technical teams evaluating this route for potential licensing or contract manufacturing opportunities.
Q: Why is the iridium-catalyzed method superior to traditional diazo compound routes?
A: Traditional methods often rely on unstable and explosive diazo compounds. The iridium-catalyzed route utilizes stable sulfur ylides, significantly enhancing operational safety and eliminating the risks associated with diazo handling during scale-up.
Q: What is the functional group compatibility of this synthesis method?
A: This method demonstrates exceptional compatibility with sensitive functional groups including phenolic hydroxyls, amino groups, indoles, alkynyl, and alkenyl moieties, which are often problematic in oxidative coupling reactions.
Q: Can this process be scaled for commercial production of API intermediates?
A: Yes, the process operates under mild temperatures (90°C-120°C) without requiring excessive strong oxidants, making it highly suitable for commercial scale-up with simplified purification and waste treatment protocols.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carboxymethyl Ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the iridium-catalyzed synthesis described in patent CN115974758A for the production of high-value pharmaceutical intermediates. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to market-ready supply is seamless. Our state-of-the-art facilities are equipped to handle air- and moisture-sensitive catalytic reactions under strict inert atmospheres, guaranteeing the stringent purity specifications required for oncology and cardiovascular drug candidates. Our rigorous QC labs utilize advanced analytical techniques to verify the absence of residual metals and impurities, delivering a product that exceeds industry standards.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific molecule, demonstrating how this route can optimize your COGS. Please contact our technical procurement team today to request specific COA data for similar heterocyclic ketones and to discuss route feasibility assessments for your target compounds. Let us help you secure a reliable, cost-effective, and scalable supply chain for your critical carboxymethyl ketone intermediates.
