Scalable Synthesis of Cis-7,7-Difluoro-Hexahydro-Pyrrolo-Pyridine Derivatives for Pharma
Scalable Synthesis of Cis-7,7-Difluoro-Hexahydro-Pyrrolo-Pyridine Derivatives for Pharma
The rapid evolution of medicinal chemistry has placed a premium on fluorinated heterocyclic scaffolds due to their ability to modulate lipophilicity and metabolic stability in drug candidates. Patent CN111620866A discloses a groundbreaking methodology for the preparation of cis-7,7-difluoro-hexahydro-1H-pyrrolo[3,4-c]pyridine derivatives, addressing a significant gap in the availability of these high-value building blocks. This technology leverages a strategic 3+2 cycloaddition followed by selective gem-difluorination to construct the core bicyclic system with precise stereochemical control. For R&D teams targeting Menin/MLL protein interaction inhibitors or thrombopoietin receptor agonists, access to such specialized intermediates is critical for accelerating lead optimization campaigns. As a reliable pharmaceutical intermediate supplier, understanding the nuances of this synthetic route allows us to offer superior supply chain solutions for complex fluorinated APIs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing fluorinated pyrrolo-pyridine cores often suffer from harsh reaction conditions that compromise yield and stereochemical integrity. Conventional routes may rely on multi-step sequences involving unstable intermediates or require expensive transition metal catalysts that introduce difficult-to-remove impurities. Furthermore, achieving the specific cis-7,7-difluoro configuration without generating trans-isomers or over-fluorinated byproducts has historically been a formidable challenge in process chemistry. These limitations frequently result in prolonged development timelines and inflated costs for active pharmaceutical ingredient manufacturing, creating bottlenecks for procurement managers seeking cost reduction in API manufacturing. The lack of a direct, high-yielding pathway has previously forced reliance on inefficient linear syntheses that are ill-suited for commercial scale-up.
The Novel Approach
The methodology outlined in the patent introduces a streamlined three-step sequence that dramatically simplifies the construction of the target scaffold. By utilizing N-substituted-3-oxo-3,6-dihydro-2H-pyridine as a starting material, the process initiates with a highly efficient 3+2 cycloaddition with a silylated amine precursor. This key transformation rapidly assembles the bicyclic framework under mild acidic catalysis, bypassing the need for complex ring-closing metathesis or high-pressure cyclizations. Subsequent fluorination installs the critical difluoro motif with high fidelity, while the final deprotection step is adaptable to various protecting groups (Boc, Cbz, Benzoyl), offering flexibility for downstream derivatization. This novel approach not only enhances the overall yield but also significantly reduces the environmental footprint by minimizing waste generation and energy consumption.
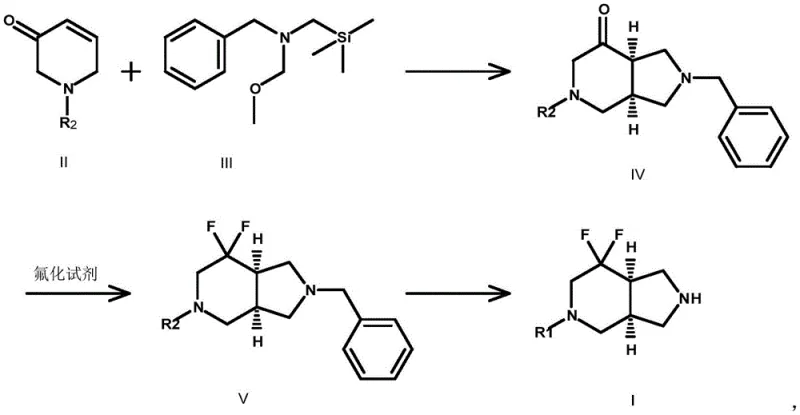
Mechanistic Insights into 3+2 Cycloaddition and Gem-Difluorination
The core of this synthetic strategy lies in the mechanistic elegance of the initial cycloaddition step. The reaction between the electron-deficient enone system of Compound II and the nucleophilic silyl amine (Compound III) proceeds via a concerted or stepwise dipolar mechanism facilitated by trifluoroacetic acid (TFA). The acid activates the carbonyl group, promoting the attack of the amine nitrogen and subsequent ring closure to form the pyrrolidine fused system. This step is crucial as it establishes the relative stereochemistry of the bridgehead hydrogens, which is preserved throughout the subsequent transformations. The use of molar ratios ranging from 1:1 to 1:5 ensures complete consumption of the valuable enone starting material, driving the equilibrium towards the desired bicyclic ketone intermediate (Compound IV).
Following the ring construction, the installation of the fluorine atoms is achieved using robust fluorinating agents such as diethylaminosulfur trifluoride (DAST) or its tetrafluoroborate salt. The mechanism involves the activation of the ketone carbonyl at cryogenic temperatures (-78°C to 20°C), followed by nucleophilic attack by fluoride ions to generate the gem-difluoro species. This low-temperature protocol is essential to prevent elimination reactions or decomposition of the sensitive heterocyclic ring. The final deprotection, whether via catalytic hydrogenation using Pd/C or chemical cleavage with 1-chloroethyl chloroformate, cleanly removes the nitrogen protecting group to reveal the free amine or the desired protected derivative (Compound I), ready for coupling in final drug assembly.
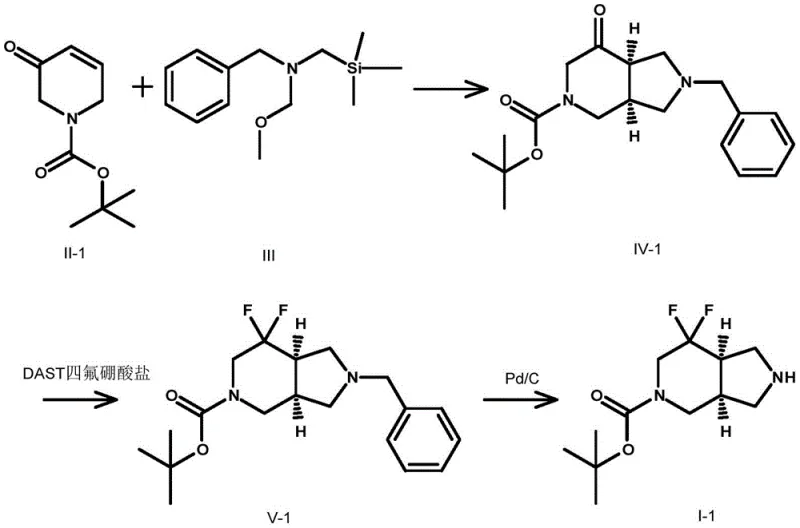
How to Synthesize Cis-7,7-Difluoro-Hexahydro-1H-Pyrrolo[3,4-c]Pyridine Efficiently
Executing this synthesis requires careful attention to temperature control and reagent stoichiometry to maximize yield and purity. The process begins with the preparation of the bicyclic ketone intermediate, followed by the critical fluorination step which demands strict anhydrous conditions. Detailed operational parameters, including solvent choices like acetonitrile or dichloromethane and specific workup procedures, are essential for reproducibility. For process chemists aiming to implement this route, adhering to the standardized protocols ensures consistent quality. The detailed standardized synthesis steps are provided in the guide below.
- Perform a 3+2 cycloaddition between N-substituted-3-oxo-3,6-dihydro-2H-pyridine and N-(methoxymethyl)-N-(trimethylsilylmethyl)benzylamine using TFA catalysis.
- React the resulting intermediate with a fluorinating reagent such as DAST or BAST at low temperatures (-78°C to 20°C) to install the gem-difluoro motif.
- Execute final deprotection via hydrogenation with Pd/C or chemical cleavage to yield the target cis-7,7-difluoro derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented route offers substantial benefits for supply chain stability and cost management. The reliance on readily available starting materials, such as substituted pyridones and simple silyl amines, mitigates the risk of raw material shortages that often plague specialty chemical supply chains. Furthermore, the avoidance of precious metal catalysts in the early stages reduces the burden on metal scavenging processes, directly lowering the cost of goods sold. The modular nature of the protecting group strategy allows manufacturers to adapt the process based on the specific needs of the downstream API synthesis, providing flexibility that is invaluable for contract development and manufacturing organizations (CDMOs).
- Cost Reduction in Manufacturing: The elimination of complex multi-step sequences in favor of a convergent 3+2 cycloaddition strategy significantly reduces the number of unit operations required. By streamlining the synthesis to three primary steps, labor costs and solvent consumption are drastically minimized. Additionally, the use of standard fluorinating reagents like DAST, which are commercially sourced at scale, prevents the need for custom reagent synthesis, further driving down material costs and enhancing the economic viability of producing high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, particularly the tolerance for varying temperatures in the cycloaddition step (-10°C to 50°C), ensures that the process can be maintained even with minor fluctuations in plant utilities. This operational resilience translates to fewer batch failures and more predictable delivery schedules for clients. The ability to produce the intermediate with purity levels reaching 98% reduces the need for extensive reprocessing, ensuring a steady flow of quality material to support continuous API manufacturing pipelines without interruption.
- Scalability and Environmental Compliance: The synthetic route is inherently scalable, moving seamlessly from gram-scale laboratory experiments to multi-kilogram pilot runs. The purification methods described, primarily column chromatography, can be transitioned to crystallization or continuous extraction processes upon scale-up, aligning with green chemistry principles. This adaptability facilitates the commercial scale-up of complex pharmaceutical intermediates while maintaining strict adherence to environmental regulations regarding solvent disposal and waste management.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthetic pathway. Understanding these details helps R&D and procurement teams assess the feasibility of integrating this intermediate into their specific drug development programs. The answers are derived directly from the experimental data and beneficial effects reported in the patent documentation.
Q: What are the key advantages of this fluorination method?
A: The method utilizes mild reaction conditions (-78°C to 20°C) and commercially available fluorinating reagents like DAST, ensuring high selectivity for the cis-7,7-difluoro configuration without compromising the sensitive bicyclic scaffold.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the route employs readily available starting materials and avoids exotic catalysts. The purification steps involve standard column chromatography which can be adapted to crystallization or preparative HPLC for ton-scale production.
Q: What is the typical purity achieved?
A: Experimental data indicates that the final compounds (e.g., Compound I-1, I-2) can be isolated with purity levels reaching 98% after standard purification protocols, meeting stringent pharmaceutical intermediate specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cis-7,7-Difluoro-Hexahydro-1H-Pyrrolo[3,4-c]Pyridine Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality fluorinated intermediates play in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and speed. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of cis-7,7-difluoro-hexahydro-1H-pyrrolo[3,4-c]pyridine derivative meets the highest industry standards. Our commitment to technical excellence allows us to navigate the complexities of fluorine chemistry effectively, delivering results that accelerate your time to market.
We invite you to collaborate with us to leverage this advanced synthetic technology for your upcoming projects. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can support your supply chain goals and drive innovation in your drug discovery pipeline.
