Advanced Silver-Catalyzed Synthesis of Hydroindole Imine Diastereoisomers for Pharma
Advanced Silver-Catalyzed Synthesis of Hydroindole Imine Diastereoisomers for Pharma
The pharmaceutical industry continuously seeks robust and efficient synthetic routes for complex heterocyclic scaffolds, particularly those found in bioactive alkaloids. Patent CN114716359B introduces a groundbreaking preparation method for hydroindole imine diastereoisomers, addressing critical bottlenecks in current organic synthesis technologies. This innovation leverages a silver compound catalyst to facilitate a one-step reaction between cyclohexenone amine compounds and pyridine ketene imine salt compounds. Unlike traditional methods that often require harsh conditions or multiple steps, this novel approach operates under mild conditions without the need for alkali or chiral auxiliary agents. The result is a highly efficient process capable of generating structurally diverse diastereomers with excellent yields and purity, marking a significant advancement for the production of high-purity pharmaceutical intermediates.
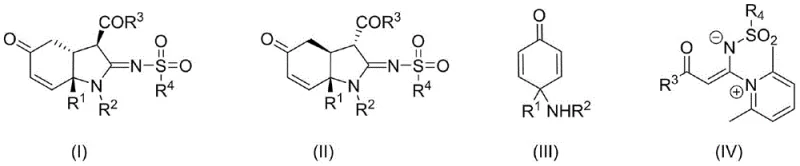
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of hydroindole derivatives has relied heavily on hydrogenation reduction, noble metal catalysis, or photocatalysis. These conventional pathways are fraught with significant disadvantages that hinder efficient commercial scale-up of complex pharmaceutical intermediates. Common issues include excessively harsh reaction conditions that compromise safety, convoluted multi-step routes that lower overall throughput, and a tendency to produce products with单一 stereostructures, limiting the diversity available for drug screening. Furthermore, these methods often suffer from low reaction yields and extended reaction times, which drastically increase manufacturing costs and lead times. The inability to efficiently synthesize diastereomerically diverse hydroindole imines has been a persistent challenge, restricting the ability of R&D teams to fully explore the structure-activity relationships of this important pharmacophore.
The Novel Approach
The method disclosed in CN114716359B represents a paradigm shift by employing a silver-catalyzed divergent synthesis strategy. This novel approach allows for the direct reaction of readily available cyclohexenone amines and pyridine ketene imine salts in an organic solvent to obtain two mutually diastereomeric hydroindole imines in a single step. By eliminating the requirement for external bases or chiral auxiliaries, the process simplifies the reaction matrix and reduces waste generation. The method boasts outstanding advantages such as simple operating conditions, high reaction efficiency, and broad substrate applicability. This capability to access diverse three-dimensional structures from a single reaction setup provides a powerful tool for medicinal chemists, enabling rapid access to various stereoisomers for biological evaluation without the need for separate, labor-intensive synthetic lines.
Mechanistic Insights into Silver-Catalyzed Cyclization
The core of this technological breakthrough lies in the unique catalytic cycle driven by the silver compound. Under the action of the silver catalyst, the pyridine ketene imine salt initially eliminates a molecule of 2,6-lutidine to generate a silver-complexed ketene imine intermediate. This activated species then undergoes a nucleophilic addition reaction with the nitrogen lone pair of the cyclohexenone amine, forming a key intermediate via an aza-Michael addition. Subsequently, the 2,6-lutidine present in the system acts as an internal organic base to abstract a proton from the quaternary ammonium nitrogen, facilitating the formation of a neutral intermediate ready for cyclization. This intricate interplay between the silver catalyst and the organic base ensures high selectivity and conversion rates, distinguishing it from non-catalyzed or poorly catalyzed alternatives.
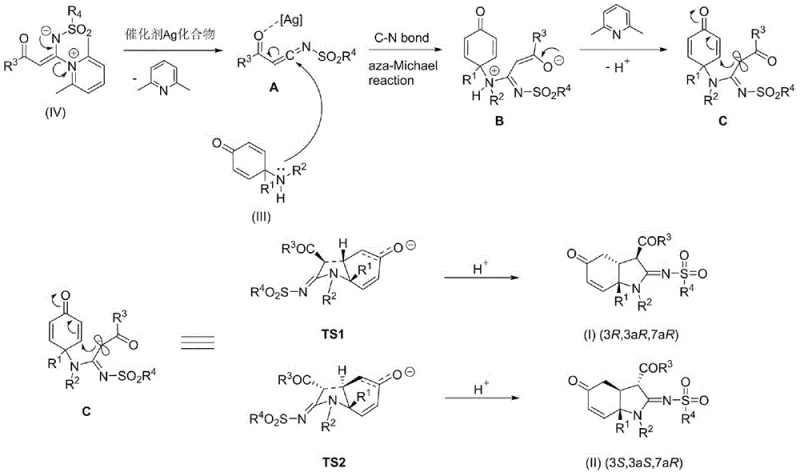
Following the initial addition, the intermediate undergoes an intramolecular cyclization reaction, passing through two stable transition states, TS1 and TS2, to yield the final diastereomeric hydroindole imines. This mechanistic pathway explains the high diastereoselectivity and the ability to produce distinct stereoisomers simultaneously. Understanding this mechanism is crucial for impurity control, as the specific transition states dictate the stereochemical outcome. By optimizing the silver catalyst loading and solvent environment, manufacturers can steer the reaction towards the desired transition state, thereby minimizing the formation of unwanted byproducts and ensuring the final product meets stringent purity specifications required for API intermediates. This level of mechanistic control is essential for maintaining batch-to-batch consistency in large-scale production.
How to Synthesize Hydroindole Imine Efficiently
To implement this synthesis effectively, precise control over reaction parameters is essential. The process typically involves mixing the cyclohexenone amine and pyridine ketene imine salt in a solvent like 1,2-dichloroethane, followed by the addition of a silver catalyst such as silver trifluoroacetate. The reaction is maintained at temperatures between 25°C and 70°C for a duration of 1 to 24 hours. Post-reaction processing involves standard workup procedures including cooling, extraction with dichloromethane and saturated saline, drying, and concentration. The crude product is then purified via crystallization or silica gel column chromatography to isolate the high-purity target compounds. For detailed operational parameters and specific stoichiometric ratios tailored to your specific substrate, please refer to the standardized guide below.
- Mix cyclohexenone amine compounds and pyridine ketene imine salt compounds in an organic solvent such as 1,2-dichloroethane.
- Add a silver compound catalyst, preferably silver trifluoroacetate, and stir the mixture at 25-70°C for 1-24 hours.
- Upon completion, cool the reaction, perform extraction and drying, and purify the crude product via crystallization or column chromatography to isolate the target diastereomers.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this silver-catalyzed methodology offers substantial strategic benefits beyond mere chemical efficiency. The elimination of expensive chiral auxiliaries and the reduction in catalyst loading directly translate to significant cost reduction in pharmaceutical intermediate manufacturing. By streamlining the synthesis into a one-step process, the method drastically reduces the number of unit operations required, which lowers energy consumption and labor costs associated with prolonged reaction times and complex separations. Furthermore, the use of common organic solvents and mild reaction temperatures enhances process safety, reducing the need for specialized high-pressure or cryogenic equipment, thereby lowering capital expenditure for production facilities.
- Cost Reduction in Manufacturing: The removal of noble metal catalysts like palladium and the avoidance of chiral resolving agents significantly lowers the raw material bill of materials. The high reaction yield and simplified purification process mean less solvent waste and higher throughput per batch. This efficiency allows for a more competitive pricing structure for the final hydroindole imine intermediates, providing a clear economic advantage in cost-sensitive generic drug markets.
- Enhanced Supply Chain Reliability: The starting materials, cyclohexenone amines and pyridine ketene imine salts, are readily accessible and stable, reducing the risk of supply disruptions associated with exotic or unstable reagents. The robustness of the reaction conditions ensures consistent output even with minor variations in feedstock quality, enhancing the reliability of supply for downstream API manufacturers. This stability is critical for maintaining continuous production schedules and meeting strict delivery deadlines.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work effectively across a wide range of scales without loss of efficiency. The simplified workup and the ability to recycle solvents contribute to a reduced environmental footprint, aligning with increasingly strict global environmental regulations. This compliance minimizes the risk of regulatory delays and ensures long-term sustainability of the supply chain for these critical pharmaceutical building blocks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on its practical application and advantages over legacy technologies. Understanding these details is vital for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Why is silver catalysis preferred over traditional noble metal catalysts for this synthesis?
A: Silver catalysis eliminates the need for expensive palladium or platinum complexes and avoids harsh hydrogenation conditions. It enables a divergent synthesis pathway that efficiently produces multiple diastereomers in a single step without chiral auxiliaries, significantly simplifying the process and reducing raw material costs.
Q: Can this method control the stereochemistry of the hydroindole imine products?
A: Yes, the method utilizes a desymmetrization and divergent synthesis strategy. By adjusting reaction parameters and substrate structures, it allows for the selective formation of specific diastereomers (Formula I and Formula II), providing crucial flexibility for optimizing the biological activity of downstream pharmaceutical candidates.
Q: Is this synthesis protocol suitable for large-scale commercial production?
A: Absolutely. The process operates under mild temperatures (25-70°C) using common organic solvents like 1,2-dichloroethane. The workup involves standard extraction and crystallization techniques, avoiding complex purification steps, which makes it highly scalable and compliant with industrial environmental and safety standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Hydroindole Imine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the silver-catalyzed synthesis described in CN114716359B for the production of advanced pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs, guaranteeing that every batch of hydroindole imine delivered meets the highest international standards for safety and efficacy.
We invite you to collaborate with us to leverage this cutting-edge technology for your drug development programs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to request specific COA data and comprehensive route feasibility assessments, and let us help you optimize your supply chain for the next generation of therapeutic agents.
