Scalable Manufacturing of Halofuginone Intermediate via Advanced Grignard Technology
The global demand for effective anticoccidial agents in the livestock and poultry industry has necessitated the development of robust supply chains for key intermediates like halofuginone. Patent CN112279805B introduces a groundbreaking preparation method for 1-(3-methoxypyridin-2-yl)-2-propanone, a critical building block in the synthesis of this broad-spectrum antiparasitic drug. Historically, the industrialization of this intermediate has been hindered by complex reaction conditions and suboptimal yields, creating bottlenecks for pharmaceutical manufacturers. This new technical disclosure outlines a novel synthetic route that leverages advanced Grignard chemistry to overcome these traditional limitations. By utilizing a specific exchange reaction followed by controlled acylation, the process achieves remarkable efficiency and purity. For R&D directors and procurement specialists, this patent represents a pivotal shift towards more reliable and cost-effective manufacturing strategies for veterinary pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the domestic and international markets faced significant challenges in sourcing high-quality 1-(3-methoxypyridin-2-yl)-2-propanone. Existing preparation processes were often characterized by harsh reaction conditions that required specialized equipment and rigorous safety protocols, driving up operational expenditures. Furthermore, the low yields associated with traditional synthetic pathways resulted in substantial material waste and inconsistent batch-to-batch quality. These inefficiencies not only inflated the cost of goods sold but also compromised the reliability of the supply chain for downstream halofuginone production. The difficulty in realizing stable industrialization meant that many manufacturers remained dependent on imports, exposing them to geopolitical risks and logistical delays. Consequently, there was an urgent need for a method that could simplify the process while enhancing overall throughput and economic viability.
The Novel Approach
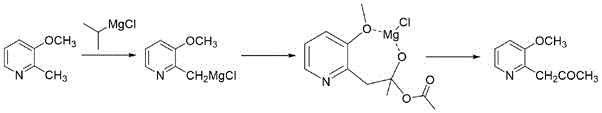
The methodology described in the patent offers a transformative solution by employing a streamlined, one-pot synthetic strategy that significantly mitigates the drawbacks of legacy techniques. By initiating the sequence with 2-methyl-3-methoxypyridine and utilizing an isopropyl magnesium chloride exchange in the presence of lithium chloride, the process generates a highly reactive Grignard species in situ. This intermediate is then immediately subjected to acylation with acetic anhydride under mild thermal conditions, eliminating the need for isolation steps that typically incur yield losses. The reaction conditions are notably gentle, operating effectively at near-ambient or slightly cooled temperatures, which reduces energy consumption and equipment stress. This approach not only simplifies the operational workflow but also ensures that the final product meets stringent purity specifications required for pharmaceutical applications, thereby facilitating the localization and scale-up of halofuginone production.
Mechanistic Insights into Grignard Exchange and Acylation
The core of this technological advancement lies in the precise execution of the Grignard exchange reaction, facilitated by the presence of lithium chloride. In this mechanism, 2-methyl-3-methoxypyridine undergoes a metal-halogen exchange-like transformation with isopropyl magnesium chloride in 2-methyltetrahydrofuran. The addition of lithium chloride is critical, as it enhances the solubility and reactivity of the organomagnesium species, promoting the formation of the ((3-methoxypyridin-2-yl)methyl)magnesium chloride intermediate. This activation allows the reaction to proceed efficiently under reflux conditions for 6 to 10 hours, ensuring complete conversion of the starting material. The resulting Grignard reagent is stabilized within the solvent matrix, ready for the subsequent nucleophilic attack without the need for purification, which preserves the integrity of the reactive species and minimizes handling hazards.
Following the formation of the Grignard reagent, the mechanism proceeds through a controlled nucleophilic acyl substitution. The organomagnesium intermediate attacks the carbonyl carbon of acetic anhydride, which is added in batches to manage the exothermic nature of the reaction. Maintaining the temperature between -5°C and 5°C during addition, and strictly below 10°C thereafter, is essential to prevent side reactions such as over-addition or decomposition of the sensitive pyridine ring. The tetrahedral intermediate formed collapses to release acetate and generate the desired ketone functionality. Subsequent hydrolysis with ammonium chloride solution quenches any remaining magnesium species and facilitates the separation of the organic product. This meticulous control over reaction kinetics and thermodynamics ensures a clean impurity profile and high isolated yields.
How to Synthesize 1-(3-Methoxypyridin-2-yl)-2-Propanone Efficiently
The synthesis protocol detailed in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing the importance of reagent ratios and thermal management. The process begins with the charging of 2-methyl-3-methoxypyridine and the Grignard source into a reactor equipped with precise heating and stirring capabilities. The addition of lithium chloride is performed in batches to ensure uniform dispersion before heating to reflux. Once the Grignard formation is complete, the mixture is cooled, and acetic anhydride is introduced carefully to maintain the low-temperature window. Detailed standardized synthesis steps regarding specific molar equivalents, stirring speeds, and distillation parameters are provided in the guide below to ensure reproducibility and safety during scale-up operations.
- Preparation of the Grignard reagent ((3-methoxypyridin-2-yl)methyl)magnesium chloride via exchange reaction using isopropyl magnesium chloride and lithium chloride in 2-methyltetrahydrofuran under reflux.
- Controlled addition of acetic anhydride to the Grignard solution at low temperatures (-5 to 5°C) to form the ketone intermediate while maintaining strict thermal control below 10°C.
- Quenching the reaction mixture with ammonium chloride solution, followed by phase separation, drying, and vacuum distillation to isolate the high-purity final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers compelling strategic benefits that extend beyond simple chemical transformation. The primary value proposition lies in the drastic simplification of the manufacturing workflow, which directly correlates to reduced operational complexity and lower overhead costs. By eliminating multiple isolation and purification steps between the Grignard formation and the final acylation, the process minimizes solvent usage and waste generation, aligning with modern green chemistry principles. Furthermore, the reliance on mature, commercially available raw materials ensures that production schedules are not held hostage by niche reagent shortages. This stability is crucial for maintaining continuous supply lines to downstream pharmaceutical clients who require just-in-time delivery of critical intermediates.
- Cost Reduction in Manufacturing: The implementation of this one-pot strategy significantly lowers the cost of goods by maximizing atom economy and minimizing unit operations. The high yield demonstrated in the patent examples, reaching over 90% in optimized conditions, means that less raw material is required to produce the same amount of finished product compared to legacy methods. Additionally, the mild reaction conditions reduce the energy burden associated with extreme heating or cooling, leading to tangible savings in utility costs. The elimination of transition metal catalysts or exotic reagents further simplifies the bill of materials, making the process economically robust against market fluctuations in specialty chemical pricing.
- Enhanced Supply Chain Reliability: Sourcing stability is greatly improved as the key starting material, 2-methyl-3-methoxypyridine, is a commodity chemical with a well-established global supply network. Unlike processes dependent on custom-synthesized precursors with long lead times, this route allows for rapid procurement and inventory turnover. The robustness of the reaction conditions also means that production is less susceptible to minor variations in environmental factors or equipment performance, ensuring consistent output quality. This reliability empowers supply chain planners to forecast demand more accurately and reduce safety stock levels, freeing up working capital for other strategic investments.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up, utilizing standard reactor configurations and common solvents like 2-methyltetrahydrofuran. The absence of hazardous byproducts and the use of aqueous ammonium chloride for quenching simplify wastewater treatment and effluent management. This ease of compliance with environmental regulations reduces the administrative burden and potential fines associated with chemical manufacturing. Moreover, the straightforward workup procedure involving phase separation and distillation is easily transferable from pilot plants to multi-ton production facilities, enabling rapid capacity expansion to meet growing market demand for halofuginone.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 1-(3-methoxypyridin-2-yl)-2-propanone based on the patented technology. These insights are derived directly from the experimental data and process descriptions to provide clarity for potential partners and technical evaluators. Understanding these details is essential for assessing the feasibility of integrating this intermediate into your existing supply chain or product portfolio.
Q: What is the primary advantage of this new synthetic route for 1-(3-methoxypyridin-2-yl)-2-propanone?
A: The primary advantage is the significant improvement in total yield, reaching up to 91.2% in optimized examples, compared to traditional methods which suffer from harsh conditions and low conversion rates. The process utilizes a one-pot strategy that simplifies operations and reduces waste.
Q: Are the raw materials for this process readily available for large-scale production?
A: Yes, the process relies on commercially mature raw materials such as 2-methyl-3-methoxypyridine and isopropyl magnesium chloride. These inputs have sufficient market supply and wide sources, ensuring supply chain stability for industrial manufacturing.
Q: How does the process ensure safety and controllability during the exothermic reaction?
A: Safety is ensured by strict temperature control during the acylation step. The addition of acetic anhydride is performed at -5 to 5°C, and the reaction mixture temperature is maintained below 10°C throughout the addition and subsequent stirring, preventing thermal runaway and ensuring consistent product quality.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-(3-Methoxypyridin-2-yl)-2-Propanone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the efficacy and safety of final veterinary pharmaceutical products. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory success to industrial reality is seamless. We are committed to delivering 1-(3-methoxypyridin-2-yl)-2-propanone with stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical instrumentation. Our capability to replicate and optimize the Grignard-based synthesis described in recent patents allows us to offer a product that meets the highest standards of the global agrochemical and pharmaceutical industries.
We invite you to collaborate with us to leverage this advanced technology for your supply chain needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our manufacturing excellence can drive value and reliability for your organization.
