Advanced Synthetic Route for 3,5-Dihalogen-2-Pentanone: Scaling High-Purity Pharmaceutical Intermediates
Advanced Synthetic Route for 3,5-Dihalogen-2-Pentanone: Scaling High-Purity Pharmaceutical Intermediates
The pharmaceutical and agrochemical industries constantly seek robust, scalable, and environmentally benign synthetic routes for critical building blocks. A significant breakthrough in this domain is detailed in patent CN111205176B, which discloses a novel method for synthesizing 3,5-dihalogen-2-pentanone. This compound serves as a pivotal intermediate for high-value active ingredients, including the fungicide prothioconazole and the sedative-hypnotic drug clomephiazole. The disclosed technology shifts away from hazardous precursors like methyl vinyl ketone, opting instead for a safer, atom-economical pathway starting from acetoacetate esters. By integrating cyclopropanation, ring-opening halogenation, and hydrolysis decarboxylation into a streamlined sequence, this innovation addresses long-standing challenges in impurity control and process safety. For R&D directors and procurement specialists, understanding this methodology is crucial for securing a reliable supply chain of high-purity intermediates while mitigating the risks associated with traditional, toxicity-laden syntheses.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3,5-dihalogen-2-pentanone has relied on pathways fraught with significant operational and safety drawbacks. One prominent prior art method utilizes acetyl-gamma-butyrolactone as a starting material, subjecting it to chlorination and esterification followed by decarboxylation with concentrated hydrochloric acid. This route is economically inefficient due to the high cost of the lactone raw material and generates substantial quantities of corrosive byproducts such as hydrogen chloride and sulfur dioxide, complicating waste treatment and environmental compliance. Another reported approach employs methyl vinyl ketone reacting with dichloromethane under copper catalysis and illumination. While chemically feasible, this method presents severe safety liabilities because methyl vinyl ketone is a highly toxic substance with a low threshold limit value, posing acute health risks to operators and requiring expensive containment infrastructure. Furthermore, these conventional routes often suffer from poor atom utilization and require rigorous purification steps between stages, which erodes overall yield and increases production lead times.
The Novel Approach
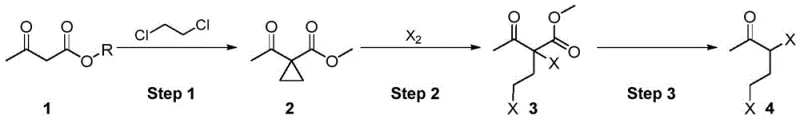
In stark contrast, the methodology described in CN111205176B introduces a paradigm shift by utilizing acetoacetate esters, which are inexpensive, commercially abundant, and significantly safer to handle. The core innovation lies in a three-step cascade that maximizes atom economy and minimizes waste generation. The process initiates with a cyclopropanation reaction using dichloroethane and a beta-cyclodextrin catalyst, followed by a direct ring-opening halogenation and a final hydrolysis decarboxylation. Crucially, this novel approach eliminates the need for intermediate isolation; the reaction mixture from the first step is carried forward directly into the second, and subsequently into the third. This telescoping capability not only drastically reduces solvent consumption and processing time but also prevents the loss of material typically associated with multiple work-up procedures. Additionally, the ability to recycle the beta-cyclodextrin catalyst from the alkaline layer further enhances the sustainability profile, making this a superior choice for cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Beta-Cyclodextrin Catalyzed Cyclopropanation and Halogenation
The chemical elegance of this synthesis is rooted in the efficient generation of a cyclopropane ring followed by its regioselective opening. In the first step, acetoacetate reacts with 1,2-dichloroethane in the presence of beta-cyclodextrin and aqueous sodium hydroxide. The beta-cyclodextrin acts as a phase-transfer catalyst and a supramolecular host, likely stabilizing the reactive carbene or carbenoid species generated from the dichloroethane under basic conditions. This facilitates the insertion of the methylene group into the active methylene position of the acetoacetate, forming a cyclopropane intermediate (Intermediate 2) with high selectivity. The reaction is conducted at mild temperatures ranging from 30°C to 50°C, which suppresses side reactions such as polymerization or over-halogenation. The use of a specific molar ratio of acetoacetate to dichloroethane (1:1.0-3.0) ensures complete conversion while maintaining a manageable exotherm. Following this, the alkaline layer containing the catalyst is separated for recycling, demonstrating a closed-loop catalytic system that aligns with green chemistry principles.
The subsequent ring-opening halogenation is a masterpiece of electrophilic addition control. The cyclopropane ring, possessing significant ring strain, is susceptible to nucleophilic attack or electrophilic addition. By introducing elemental halogens (Cl2, Br2, or I2) at low temperatures (0-5°C), the reaction proceeds via an electrophilic attack on the cyclopropane ring, leading to ring opening and the formation of the dihalogenated keto-ester (Intermediate 3). The low temperature is critical here to prevent poly-halogenation at the alpha-position of the ketone, ensuring the formation of the desired 3,5-dihalogen substitution pattern. Finally, the hydrolysis decarboxylation step utilizes hydrochloric acid at elevated temperatures (70-90°C) to cleave the ester group and remove carbon dioxide. This step is thermodynamically driven by the evolution of CO2 gas, pushing the equilibrium towards the final 3,5-dihalo-2-pentanone product. The entire sequence is designed to minimize impurity profiles, yielding products with GC purities exceeding 98%, which is essential for downstream pharmaceutical applications.
How to Synthesize 3,5-Dihalogen-2-Pentanone Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize yield and safety. The process begins with the preparation of the cyclopropanation mixture, where strict temperature control during the addition of alkali is vital to prevent thermal runaway. Operators must ensure the efficient separation of the catalyst layer to enable its reuse in subsequent batches, a key factor in process economics. The halogenation step demands careful handling of halogen gases or liquids, necessitating appropriate scrubbing systems and corrosion-resistant equipment. Finally, the decarboxylation should be performed with adequate venting to manage CO2 release. For a detailed, step-by-step breakdown of the standardized operating procedures, including specific addition rates and quenching protocols, please refer to the technical guide below.
- Perform cyclopropanation using acetoacetate, dichloroethane, and beta-cyclodextrin catalyst with aqueous alkali at 30-50°C.
- Conduct ring-opening halogenation by adding halogen (Cl2, Br2, or I2) to the intermediate solution at 0-5°C.
- Execute hydrolysis decarboxylation by dropping the halogenated intermediate into preheated hydrochloric acid at 70-90°C.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers transformative benefits that extend beyond simple chemical yield. The shift from toxic, high-cost raw materials to commodity chemicals like acetoacetate fundamentally alters the cost structure of production. By eliminating the dependency on methyl vinyl ketone, companies can avoid the premium pricing and stringent regulatory hurdles associated with hazardous substance management. Furthermore, the telescoped nature of the reaction sequence means fewer unit operations, less solvent usage, and reduced energy consumption per kilogram of product. These factors collectively contribute to a significantly reduced cost of goods sold (COGS), allowing for more competitive pricing in the global market without sacrificing margin. The ability to recycle the beta-cyclodextrin catalyst also implies a lower consumption of auxiliary materials, further driving down operational expenditures.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the use of low-cost starting materials and the elimination of intermediate purification steps. Traditional routes often require chromatography or recrystallization between steps, which are labor-intensive and result in material loss. By contrast, this method allows the crude dichloroethane solution to proceed directly to the next stage, preserving yield and reducing labor costs. Additionally, the recovery and reuse of the beta-cyclodextrin catalyst mean that the effective cost of the catalyst per batch approaches zero after the initial investment. This creates a lean manufacturing model where waste disposal costs are minimized, and raw material efficiency is maximized, leading to substantial long-term savings.
- Enhanced Supply Chain Reliability: Supply chain resilience is heavily dependent on the availability and safety of raw materials. Methyl vinyl ketone, used in older methods, is often subject to strict transportation regulations and limited supplier bases due to its toxicity. Acetoacetate esters and dichloroethane, however, are bulk commodities produced at massive scales globally, ensuring a stable and diversified supply base. This reduces the risk of supply disruptions caused by regulatory shutdowns or single-source failures. Moreover, the simplified process flow reduces the complexity of the manufacturing schedule, allowing for faster turnaround times and more flexible production planning. This reliability is critical for meeting the just-in-time delivery requirements of major pharmaceutical clients.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental issues, but this route is inherently designed for industrial amplification. The reaction conditions are mild, avoiding extreme pressures or temperatures that require specialized high-pressure reactors. The absence of heavy metal catalysts (like copper in prior art) simplifies the removal of trace metals from the final API, a critical quality attribute for regulatory approval. From an environmental standpoint, the high atom utilization and reduced waste generation align with increasingly strict global environmental standards. This facilitates easier permitting for new production facilities and reduces the liability associated with hazardous waste treatment, making the commercial scale-up of complex pharmaceutical intermediates smoother and more sustainable.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its implementation. The following questions address common concerns regarding process safety, purity, and scalability. The answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a factual basis for decision-making. Whether you are concerned about the stability of the intermediates or the feasibility of catalyst recycling, these insights clarify the operational realities of the technology.
Q: What are the key advantages of the beta-cyclodextrin catalyzed route over traditional methods?
A: Unlike traditional methods using toxic methyl vinyl ketone or expensive lactones, this route utilizes inexpensive acetoacetate and allows for the recycling of the beta-cyclodextrin catalyst, significantly reducing waste and raw material costs.
Q: Is intermediate purification required between the three reaction steps?
A: No, the process is designed for telescoping. The dichloroethane solution from the cyclopropanation step is used directly for halogenation, and the resulting solution is subsequently used for hydrolysis, eliminating isolation steps and improving overall yield.
Q: What represents the primary safety improvement in this synthesis compared to prior art?
A: The method avoids the use of methyl vinyl ketone, a highly toxic and hazardous raw material found in older literature routes, thereby enhancing production safety and reducing regulatory compliance burdens.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,5-Dihalogen-2-Pentanone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. We have extensively analyzed the synthetic pathway described in CN111205176B and possess the engineering know-how to optimize this route for large-scale production. Our facilities are equipped to handle the specific requirements of cyclopropanation and halogenation chemistry safely and efficiently. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our commitment to quality is unwavering, supported by stringent purity specifications and rigorous QC labs that verify every batch against the highest industry standards.
We invite you to collaborate with us to leverage this advanced technology for your supply chain. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a sustainable, cost-effective, and high-quality supply of 3,5-dihalogen-2-pentanone, empowering your R&D and production teams to focus on delivering life-saving therapies to the market.
