Advanced Copper-Catalyzed Synthesis of Tetra-Substituted Z-Beta-Sulfonyl Enamines for Commercial Scale
Advanced Copper-Catalyzed Synthesis of Tetra-Substituted Z-Beta-Sulfonyl Enamines for Commercial Scale
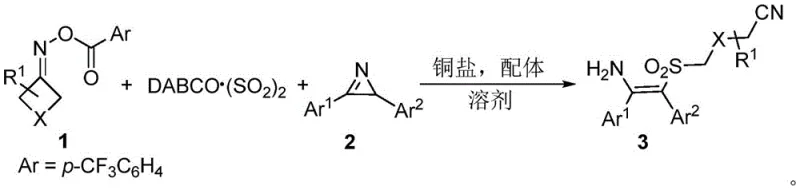
The pharmaceutical and agrochemical industries are constantly seeking robust methodologies for constructing complex sulfone-containing scaffolds, which are critical motifs in numerous bioactive molecules. Patent CN111138325A introduces a groundbreaking preparation method for (Z)-β-sulfonyl enamine compounds, addressing significant challenges in stereoselectivity and operational safety. This technology leverages a copper-catalyzed three-component coupling reaction involving aziridines, O-acyl cyclic ketoximes, and the solid sulfur dioxide surrogate DABCO(SO2)2. By utilizing this innovative approach, manufacturers can access tetra-substituted alkenyl sulfones with high precision under mild conditions. The process represents a substantial leap forward for reliable pharmaceutical intermediate supplier networks, offering a pathway to synthesize diverse molecular architectures that were previously difficult to access efficiently. The integration of solid SO2 sources not only enhances laboratory safety but also simplifies the engineering controls required for large-scale production, making it an attractive option for industrial adoption.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of alkenyl sulfones has relied heavily on the direct use of gaseous sulfur dioxide, which presents severe handling difficulties and safety risks in a manufacturing environment. Conventional routes often require high pressures, specialized corrosion-resistant equipment, and rigorous safety protocols to manage the toxicity and volatility of SO2 gas. Furthermore, achieving high stereoselectivity, particularly for tetra-substituted alkene systems, has been a persistent challenge, often resulting in mixtures of E/Z isomers that require costly and yield-reducing separation processes. Many existing methods also suffer from limited substrate tolerance, failing to accommodate sensitive functional groups commonly found in advanced drug candidates. These limitations collectively increase the cost reduction in API manufacturing barriers and extend the lead time for high-purity pharmaceutical intermediates, creating bottlenecks in the supply chain for complex therapeutic agents.
The Novel Approach
The methodology disclosed in the patent overcomes these hurdles by employing DABCO(SO2)2 as a stable, solid source of sulfur dioxide, effectively bypassing the need for hazardous gas handling. This copper-catalyzed system facilitates a radical cross-coupling reaction that proceeds under remarkably mild conditions, typically between 20°C and 50°C, significantly reducing energy consumption compared to thermal-intensive traditional methods. The reaction exhibits excellent chemoselectivity and stereoselectivity, predominantly yielding the desired (Z)-isomer without the formation of complex byproduct mixtures. By enabling the direct construction of tetra-substituted frameworks from readily available aziridines and oximes, this approach streamlines the synthetic route and enhances the overall atom economy. This innovation supports the commercial scale-up of complex polymer additives and pharmaceutical intermediates by providing a scalable, safe, and efficient protocol that aligns with modern green chemistry principles.
Mechanistic Insights into Copper-Catalyzed Radical Cross-Coupling
The core of this transformation lies in a sophisticated radical mechanism initiated by the single-electron reduction capabilities of the copper catalyst. The process begins with the activation of the O-acyl cyclic ketoxime by the monovalent copper species, leading to the elimination of the N-O bond and the generation of an iminyl radical. This intermediate rapidly undergoes ring-opening to form a carbon-centered alkyl radical, which subsequently traps the sulfur dioxide molecule released from the DABCO adduct to generate a sulfonyl radical species. Simultaneously, the aziridine substrate is reduced by the copper catalyst to produce a benzyl radical through ring opening. The convergence of these two distinct radical species—the sulfonyl radical and the benzyl radical—via cross-coupling forms the new carbon-sulfur and carbon-carbon bonds essential for the target structure. The catalytic cycle is completed as the divalent copper species generated during oxidation is reduced back to the active monovalent state by the DABCO(SO2)2 system, ensuring continuous turnover and high efficiency throughout the reaction duration.
Controlling the stereochemical outcome is paramount in the synthesis of bioactive intermediates, and this mechanism inherently favors the formation of the (Z)-configuration through specific transition state geometries during the radical recombination and subsequent isomerization steps. The presence of the amino group introduced from the aziridine ring opening adds significant value to the final scaffold, providing a handle for further derivatization in downstream medicinal chemistry campaigns. Understanding these mechanistic nuances allows process chemists to fine-tune reaction parameters such as ligand selection and solvent polarity to maximize yield and purity. For instance, the use of 1,10-phenanthroline as a ligand stabilizes the copper center and modulates its redox potential, which is critical for maintaining the delicate balance between radical generation and coupling. This deep mechanistic understanding ensures that the production of high-purity OLED material precursors or drug intermediates remains consistent and reproducible across different batch sizes.
How to Synthesize (Z)-β-sulfonyl enamine Efficiently
To implement this synthesis effectively, operators must adhere to precise stoichiometric ratios and environmental controls to ensure optimal conversion and selectivity. The standard protocol involves charging the reactor with the O-acyl cyclic ketoxime, the specific aziridine derivative, and the solid DABCO(SO2)2 adduct under an inert atmosphere to prevent oxidative degradation of the radical intermediates. Following the addition of the copper catalyst and the bidentate nitrogen ligand, the mixture is suspended in a polar aprotic solvent such as acetonitrile, which facilitates the solubility of the ionic intermediates and stabilizes the radical species. The detailed standardized synthesis steps see the guide below for specific temperature profiles and workup procedures tailored to different substrate classes.
- Combine O-acyl cyclic ketoxime, aziridine, and DABCO(SO2)2 in a reaction vessel under inert gas protection.
- Add copper salt catalyst (e.g., CuOAc) and ligand (e.g., 1,10-phenanthroline) along with an organic solvent like acetonitrile.
- Stir the mixture at 20-80°C for 2-20 hours, then perform extractive workup and column chromatography to isolate the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the shift towards this solid-state sulfur dioxide insertion methodology offers profound benefits regarding operational safety and logistical simplicity. By eliminating the need for pressurized gas cylinders and specialized scrubbing systems for toxic SO2 emissions, facilities can significantly reduce their capital expenditure on safety infrastructure and regulatory compliance measures. This simplification of the process hardware directly translates to lower overhead costs and a more agile manufacturing footprint, allowing suppliers to respond more rapidly to fluctuating market demands. Furthermore, the use of earth-abundant copper catalysts instead of precious metals like palladium or rhodium drastically reduces the raw material cost base, making the final intermediates more price-competitive in the global marketplace. These factors combined create a resilient supply chain capable of sustaining long-term production schedules without the vulnerabilities associated with hazardous reagent sourcing.
- Cost Reduction in Manufacturing: The replacement of expensive noble metal catalysts with inexpensive copper salts, coupled with the use of commodity chemicals like DABCO and simple oximes, drives down the bill of materials substantially. The mild reaction temperatures eliminate the need for extensive heating or cooling utilities, further lowering the energy intensity of the production process. Additionally, the high stereoselectivity minimizes the loss of material during purification, improving the overall mass balance and reducing waste disposal costs associated with failed batches or difficult separations.
- Enhanced Supply Chain Reliability: Utilizing solid reagents like DABCO(SO2)2 ensures stable inventory management, as these materials have long shelf lives and do not require specialized storage conditions like cryogenic tanks or high-pressure vaults. This stability mitigates the risk of supply disruptions caused by transportation regulations on hazardous gases, ensuring a continuous flow of raw materials to the production line. The robustness of the reaction conditions also means that the process is less susceptible to minor variations in utility supply, guaranteeing consistent output quality even in diverse manufacturing environments.
- Scalability and Environmental Compliance: The absence of gaseous effluents simplifies the environmental permitting process for new production lines, accelerating the time-to-market for new products. The reaction generates minimal hazardous waste, aligning with increasingly stringent global environmental standards and corporate sustainability goals. The straightforward workup procedure, involving simple extraction and chromatography, is easily adaptable to continuous flow processing or large-batch reactors, facilitating seamless technology transfer from pilot plant to full commercial scale.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented synthesis technology. These answers are derived directly from the experimental data and claims within the patent documentation to provide accurate guidance for process development teams. Understanding these specifics helps in evaluating the feasibility of adopting this route for specific target molecules in your pipeline.
Q: What is the primary advantage of using DABCO(SO2)2 in this synthesis?
A: DABCO(SO2)2 serves as a solid, easy-to-handle source of sulfur dioxide, eliminating the safety hazards and equipment corrosion associated with using gaseous SO2 directly.
Q: Does this method offer high stereoselectivity for the product?
A: Yes, the copper-catalyzed radical cross-coupling process described in the patent specifically yields the (Z)-isomer with high spatial configuration control, minimizing side reactions.
Q: What types of substrates are compatible with this reaction protocol?
A: The method demonstrates a wide substrate scope, tolerating various electron-donating and electron-withdrawing groups on the aromatic rings of both the aziridine and the oxime components.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (Z)-β-sulfonyl enamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing advanced synthetic technologies to maintain a competitive edge in the fine chemical sector. Our team of expert process chemists has extensively evaluated the copper-catalyzed sulfonylation protocol and is fully equipped to translate this laboratory-scale innovation into robust commercial manufacturing processes. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. Our state-of-the-art facilities are designed to handle sensitive radical chemistries safely, supported by stringent purity specifications and rigorous QC labs that verify every batch against the highest industry standards.
We invite you to collaborate with us to leverage this cutting-edge methodology for your next project. By partnering with our technical procurement team, you can obtain a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this greener, more efficient route. We encourage you to contact us today to request specific COA data and route feasibility assessments tailored to your unique molecular targets, ensuring a seamless path from concept to commercial reality.
