Advanced Synthesis of 4-(4-Ethoxycarbonyl-1,2,3-triazole)phenylacetylene for Pharmaceutical Intermediates
The pharmaceutical and agrochemical industries are constantly seeking robust synthetic pathways for complex heterocyclic building blocks, particularly those containing the privileged 1,2,3-triazole scaffold. Patent CN101440068A, published in May 2009, introduces a groundbreaking methodology for the preparation of 4-(4-ethoxycarbonyl-1,2,3-triazole)phenylacetylene, a valuable intermediate with significant potential in medicinal chemistry. This compound serves as a critical synthon for further functionalization, enabling the construction of diverse bioactive molecules. The disclosed technology leverages a clever four-step sequence that transforms readily available p-azidocinnamic acid into the target alkyne through a combination of copper-catalyzed click chemistry and strategic functional group manipulations. For R&D directors and procurement specialists alike, this patent represents a shift towards more efficient, cost-effective manufacturing strategies that bypass the limitations of traditional heterocycle synthesis.
Furthermore, the integration of microwave-assisted techniques in the decarboxylation step highlights a modern approach to process intensification, drastically reducing reaction times from hours to mere seconds. This innovation not only enhances throughput but also minimizes energy consumption, aligning with contemporary green chemistry principles. As a reliable pharmaceutical intermediate supplier, understanding such nuanced process improvements is essential for maintaining competitive advantage in the global market. The following analysis dissects the technical merits of this route, providing actionable insights for scaling production and optimizing supply chain logistics for high-purity triazole derivatives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the direct introduction of alkynyl groups into 1,2,3-triazole rings has presented significant synthetic challenges, often requiring harsh conditions that compromise yield and purity. Conventional methods frequently struggle with regioselectivity issues, leading to complex mixtures of 1,4- and 1,5-disubstituted isomers that are difficult and costly to separate. Additionally, traditional alkyne synthesis often relies on expensive palladium catalysts or unstable reagents that pose safety risks during scale-up. The instability of terminal alkynes under certain reaction conditions can lead to polymerization or Glaser coupling side reactions, resulting in substantial material loss. For procurement managers, these inefficiencies translate into higher raw material costs and extended lead times due to the need for rigorous purification protocols. The inability to reliably produce high-purity alkynyl-triazoles has long been a bottleneck in the development of novel antifungal and anticancer agents.
The Novel Approach
The methodology described in CN101440068A circumvents these obstacles by employing an indirect synthetic strategy that builds the triazole ring first, followed by the installation of the alkyne functionality via a stable precursor. This route begins with a highly regioselective copper-catalyzed azide-alkyne cycloaddition (CuAAC), ensuring the exclusive formation of the 1,4-isomer. Subsequent transformations involve bromination and a unique microwave-assisted decarboxylative elimination, which elegantly installs the vinyl bromide moiety before final conversion to the alkyne. 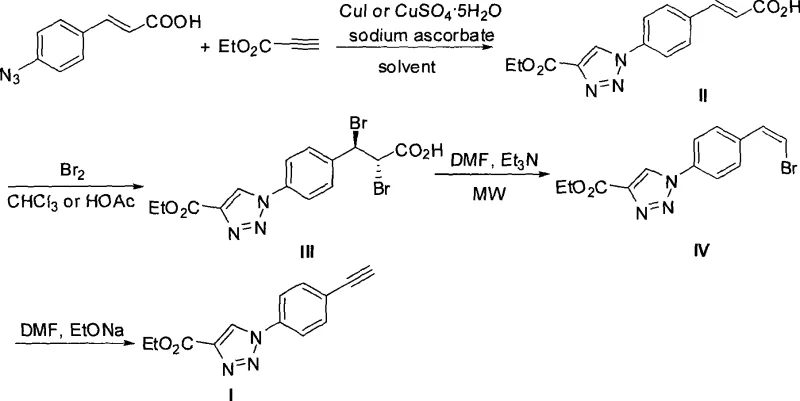 This novel approach offers a streamlined pathway that avoids the direct handling of sensitive alkyne-triazole intermediates until the final step. By utilizing cheap and abundant starting materials like p-azidocinnamic acid and ethyl propiolate, the process significantly lowers the barrier to entry for commercial production. The operational simplicity, characterized by standard solvent systems and straightforward workup procedures, makes this method exceptionally attractive for cost reduction in pharmaceutical intermediate manufacturing.
This novel approach offers a streamlined pathway that avoids the direct handling of sensitive alkyne-triazole intermediates until the final step. By utilizing cheap and abundant starting materials like p-azidocinnamic acid and ethyl propiolate, the process significantly lowers the barrier to entry for commercial production. The operational simplicity, characterized by standard solvent systems and straightforward workup procedures, makes this method exceptionally attractive for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Cu-Catalyzed Cycloaddition and Elimination
The cornerstone of this synthesis is the initial Cu(I)-catalyzed 1,3-dipolar cycloaddition between p-azidocinnamic acid and ethyl propiolate. In this mechanism, sodium ascorbate acts as a crucial reducing agent, maintaining the copper catalyst in its active +1 oxidation state throughout the reaction cycle. This prevents the oxidation of Cu(I) to inactive Cu(II), thereby sustaining high catalytic turnover and ensuring complete conversion of the azide. The reaction proceeds through a metallacycle intermediate that dictates the strict 1,4-regioselectivity observed in the product. For R&D teams, controlling this step is vital, as any deviation could lead to isomeric impurities that are notoriously difficult to remove later in the sequence. The use of solvents like acetonitrile or DMF facilitates the solubility of both organic substrates and the inorganic catalyst, creating a homogeneous reaction environment that maximizes efficiency.
Following the cycloaddition, the mechanism shifts to electrophilic addition and elimination. The treatment with liquid bromine results in the anti-addition across the double bond of the cinnamic acid derivative, forming a vicinal dibromide. The subsequent decarboxylation step is particularly ingenious; under microwave irradiation in the presence of triethylamine, the dibromo acid undergoes rapid loss of carbon dioxide and one equivalent of hydrogen bromide. This generates a cis-bromoolefin intermediate with high stereochemical fidelity. Finally, the treatment with sodium ethoxide induces a second dehydrohalogenation, removing the remaining bromine atom to reveal the terminal alkyne. This stepwise unmasking of the alkyne functionality protects the reactive triple bond during the earlier, more vigorous chemical transformations, thereby preserving the integrity of the final product and minimizing impurity profiles.
How to Synthesize 4-(4-Ethoxycarbonyl-1,2,3-triazole)phenylacetylene Efficiently
Implementing this synthesis requires precise control over reaction parameters, particularly temperature and stoichiometry, to ensure optimal yields at each stage. The process is designed to be modular, allowing for the isolation and characterization of intermediates if necessary, though a telescoped approach may be feasible for experienced process chemists. The initial cycloaddition is performed at room temperature under nitrogen protection to prevent catalyst deactivation by oxygen. Following this, the bromination step requires careful temperature management between 60°C and 80°C to drive the reaction to completion without promoting side reactions. The microwave step is the most time-sensitive, requiring only 25 to 40 seconds of irradiation, which demands specialized equipment but offers unparalleled speed. Detailed standardized operating procedures for these steps are essential for reproducibility.
- Perform Cu-catalyzed 1,3-dipolar cycloaddition between p-azidocinnamic acid and ethyl propiolate using sodium ascorbate.
- Conduct electrophilic addition with liquid bromine in chloroform or acetic acid to form the dibromo intermediate.
- Execute microwave-assisted decarboxylative elimination using triethylamine in DMF to yield the cis-bromoolefin.
- Complete the synthesis via dehydrohalogenation with sodium ethoxide in DMF to obtain the final phenylacetylene product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented route offers compelling advantages that directly address the pain points of global supply chain management and cost control. The reliance on commodity chemicals such as p-azidocinnamic acid and ethyl propiolate ensures a stable and resilient supply base, reducing the risk of raw material shortages that often plague specialty chemical manufacturing. Furthermore, the elimination of expensive noble metal catalysts like palladium in favor of inexpensive copper salts significantly lowers the direct material cost of goods sold. The simplified purification processes, which primarily involve aqueous washes and solvent evaporation rather than complex chromatography, reduce solvent consumption and waste disposal costs. These factors combine to create a manufacturing process that is not only economically viable but also environmentally sustainable, appealing to increasingly eco-conscious stakeholders.
- Cost Reduction in Manufacturing: The substitution of precious metal catalysts with copper-based systems drastically reduces catalyst costs, which are often a major expense in fine chemical synthesis. Additionally, the high yields reported in the patent examples minimize the need for recycling unreacted starting materials, further enhancing overall process economy. The use of common solvents like ethyl acetate and chloroform allows for bulk purchasing and efficient solvent recovery systems, contributing to substantial operational savings. By streamlining the synthetic sequence to just four steps, the process reduces labor hours and equipment occupancy time, maximizing facility throughput.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis are widely available from multiple global suppliers, mitigating the risk of single-source dependency. The robustness of the reaction conditions means that the process is less susceptible to variations in raw material quality, ensuring consistent output even with fluctuating supply inputs. The short reaction times, especially in the microwave-assisted step, allow for rapid response to market demand spikes, effectively reducing lead time for high-purity pharmaceutical intermediates. This agility is crucial for maintaining just-in-time inventory levels and meeting tight delivery schedules for downstream API manufacturers.
- Scalability and Environmental Compliance: The process is inherently scalable, as demonstrated by the use of standard laboratory glassware that translates well to pilot and production-scale reactors. The absence of hazardous reagents beyond standard laboratory bromine simplifies safety compliance and waste treatment protocols. The microwave step, while requiring specific equipment, can be adapted to continuous flow processing for even greater scalability and energy efficiency. Overall, the reduced solvent usage and high atom economy of the click chemistry step align with green chemistry metrics, facilitating easier regulatory approval and environmental permitting for new production facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of 4-(4-ethoxycarbonyl-1,2,3-triazole)phenylacetylene. These answers are derived directly from the technical specifications and experimental data provided in the patent literature. Understanding these details is critical for technical procurement teams evaluating the feasibility of integrating this intermediate into their supply chains. The responses cover aspects of reaction safety, purity standards, and scalability potential.
Q: Why is this indirect synthesis route preferred over direct alkyne introduction?
A: Direct introduction of alkynyl groups into 1,2,3-triazole heterocycles is chemically challenging and often suffers from low regioselectivity. This patented indirect route via a cinnamic acid precursor ensures high structural fidelity and yield.
Q: What are the critical reaction conditions for the decarboxylation step?
A: The decarboxylation step utilizes microwave irradiation in DMF with triethylamine. This specific condition allows for rapid reaction completion (25-40 seconds) compared to traditional thermal heating, minimizing decomposition.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process uses readily available raw materials like p-azidocinnamic acid and common solvents. The operations are simple, involving standard workups like washing and rotary evaporation, making it highly amenable to scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-(4-Ethoxycarbonyl-1,2,3-triazole)phenylacetylene Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthetic methodologies like the one described in CN101440068A for driving innovation in drug discovery. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop to full-scale manufacturing. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Whether you require custom synthesis or established intermediates, our infrastructure is designed to support your long-term growth and stability in the competitive pharmaceutical landscape.
We invite you to engage with our technical procurement team to discuss how we can tailor this synthesis to your specific volume and quality requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how optimizing this route can benefit your bottom line. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, ensuring that our capabilities align perfectly with your project milestones. Let us be your partner in transforming complex chemical challenges into commercial successes.
