Advanced Nickel-Catalyzed Synthesis of 2-Trifluoromethylnaphthalene Derivatives for Commercial Scale-Up
The development of efficient synthetic routes for fluorinated aromatic compounds remains a critical priority in modern medicinal chemistry, particularly for constructing metabolically stable scaffolds. Patent CN115010575B introduces a groundbreaking methodology for the synthesis of 2-trifluoromethylnaphthalene derivatives, utilizing a nickel-catalyzed reductive cyclization strategy. This innovation addresses the longstanding challenges associated with introducing trifluoromethyl groups into polycyclic aromatic systems, which are ubiquitous motifs in high-performance agrochemicals and pharmaceutical active ingredients. By leveraging a robust nickel catalytic system coupled with a zinc reductant, this process achieves high yields under relatively mild thermal conditions, marking a significant departure from energy-intensive traditional protocols. For R&D teams seeking to optimize impurity profiles and streamline process chemistry, this patent offers a compelling blueprint for accessing complex fluorinated architectures with precision and reliability.

The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of naphthalene cores functionalized with trifluoromethyl groups has relied heavily on transition-metal catalyzed cycloadditions or electrophilic cyclizations promoted by hypervalent iodine reagents. These conventional pathways often suffer from severe limitations, including the requirement for stringent reaction conditions that demand specialized equipment and rigorous safety protocols. Furthermore, the use of expensive stoichiometric oxidants or precious metal catalysts like palladium can drastically inflate the cost of goods, rendering these methods economically unviable for large-scale manufacturing. The generation of hazardous byproducts and the difficulty in controlling regioselectivity during the cyclization steps further complicate the purification process, leading to lower overall throughput and increased environmental burden. Such inefficiencies create significant bottlenecks for supply chain managers aiming to secure consistent volumes of high-purity intermediates for downstream drug development.
The Novel Approach
In stark contrast, the novel approach detailed in CN115010575B utilizes a nickel-catalyzed difunctionalization of alkynes to construct the naphthalene ring system in a single operational step. This method employs readily available starting materials, specifically 1-bromo-2-(2-chloro-3,3,3-trifluoroprop-1-en-1-yl)benzene and various alkynes, which react efficiently in the presence of a nickel catalyst and zinc powder. The reaction proceeds smoothly at 80°C in polar aprotic solvents like N-methylpyrrolidone, eliminating the need for cryogenic conditions or high-pressure reactors. This shift towards base-metal catalysis not only reduces the reliance on scarce precious metals but also simplifies the workup procedure, as the reaction mixture can be directly quenched and extracted without complex intermediate isolations. The result is a streamlined process that delivers excellent yields, reaching up to 84% in optimized examples, while maintaining a cleaner reaction profile that aligns with green chemistry principles.
Mechanistic Insights into Nickel-Catalyzed Reductive Cyclization
The success of this transformation hinges on a sophisticated catalytic cycle involving multiple oxidation states of nickel, which facilitates the sequential activation of carbon-halogen bonds and the subsequent ring closure. As illustrated in the mechanistic proposal, the cycle initiates with the oxidative addition of a zero-valent nickel species into the aryl bromide bond of the substrate, generating an organonickel(II) intermediate. This species then undergoes a cis-insertion of the alkyne moiety, forming a new carbon-carbon bond and extending the conjugated system. Subsequent single-electron reduction by zinc metal converts the divalent nickel species into a monovalent state, which is crucial for the next activation step. The monovalent nickel then engages in a second oxidative addition with the alkenyl chloride functionality, creating a transient trivalent cyclic nickel species that sets the stage for the final ring closure.
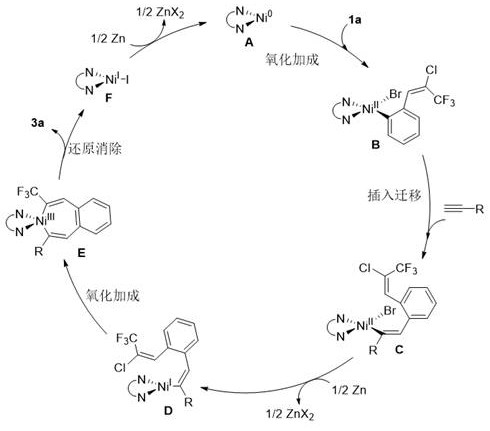
Following the formation of the trivalent intermediate, a reductive elimination step occurs, releasing the desired 2-trifluoromethylnaphthalene product and regenerating the monovalent nickel species. To close the catalytic loop, the monovalent nickel undergoes a final single-electron reduction back to the active zero-valent state, ready to enter another turnover cycle. This intricate dance of electron transfer and bond formation ensures high efficiency and selectivity, minimizing the formation of side products such as homocoupled alkynes or dehalogenated byproducts. Understanding this mechanism is vital for process chemists, as it highlights the importance of maintaining strict anhydrous and anaerobic conditions to preserve the activity of the low-valent nickel species. Moreover, the choice of ligand, such as 2,2'-bipyridine-4,4'-dicarboxylate, plays a pivotal role in stabilizing the nickel center throughout these redox transitions, thereby enhancing the overall robustness of the catalytic system.
How to Synthesize 2-Trifluoromethylnaphthalene Efficiently
Implementing this synthesis requires careful attention to reagent quality and atmospheric control to maximize yield and reproducibility. The protocol involves combining the bromo-chloro precursor with the chosen alkyne in a dry solvent under an inert nitrogen atmosphere, followed by the sequential addition of the catalyst, ligand, and reductant. Detailed standard operating procedures regarding stoichiometry, addition rates, and quenching methods are essential for translating this laboratory-scale success to pilot plant operations.
- Prepare the reaction mixture by combining 1-bromo-2-(2-chloro-3,3,3-trifluoroprop-1-en-1-yl)benzene and the selected alkyne substrate in N-methylpyrrolidone solvent under nitrogen.
- Add the nickel catalyst (e.g., NiI2), bipyridine ligand, and zinc powder as the reducing agent to the reaction vessel.
- Stir the mixture at 80°C for 12 hours, then quench, extract, and purify via silica gel chromatography to isolate the target naphthalene derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented technology offers substantial strategic benefits for organizations focused on cost-effective manufacturing and supply chain resilience. The transition from precious metal catalysts to abundant nickel represents a fundamental shift in cost structure, removing the volatility associated with palladium or rhodium pricing. Additionally, the operational simplicity of the reaction, which avoids extreme temperatures or pressures, reduces the capital expenditure required for specialized reactor infrastructure. This accessibility allows for more flexible production scheduling and faster response times to market demands, ensuring that critical intermediates remain available even during periods of global supply chain disruption. The reduced environmental footprint also translates to lower waste disposal costs and simplified regulatory compliance, further enhancing the economic viability of the process.
- Cost Reduction in Manufacturing: The substitution of expensive noble metal catalysts with economical nickel salts significantly lowers the raw material costs per kilogram of product. Furthermore, the high atom economy of the cyclization reaction minimizes the consumption of starting materials, while the elimination of intermediate isolation steps reduces solvent usage and labor hours. These cumulative efficiencies drive down the overall cost of goods sold, enabling more competitive pricing strategies for downstream pharmaceutical applications without compromising on quality standards.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable reagents, such as zinc powder and common bipyridine ligands, mitigates the risk of supply shortages that often plague specialized chemical markets. The robustness of the reaction conditions ensures consistent batch-to-batch performance, which is critical for maintaining long-term supply agreements with key partners. By diversifying the synthetic toolbox with this reliable method, procurement teams can secure a steady flow of high-value intermediates, safeguarding production timelines against external logistical challenges.
- Scalability and Environmental Compliance: The mild thermal requirements and lack of hazardous gaseous byproducts make this process inherently safer and easier to scale from kilogram to multi-ton quantities. The reduced generation of toxic waste streams aligns with increasingly stringent environmental regulations, facilitating smoother permitting processes for manufacturing facilities. This scalability ensures that the technology can meet the growing demand for fluorinated building blocks in the life sciences sector, supporting the rapid development of new therapeutic candidates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this nickel-catalyzed synthesis. These insights are derived directly from the experimental data and scope defined within the patent documentation, providing clarity for technical decision-makers.
Q: What are the key advantages of this nickel-catalyzed method over traditional naphthalene synthesis?
A: Unlike traditional methods requiring harsh conditions or expensive hypervalent iodine reagents, this nickel-catalyzed protocol operates under mild temperatures (80°C) with high atom economy and significantly reduced waste generation.
Q: Is this process suitable for large-scale manufacturing of pharmaceutical intermediates?
A: Yes, the use of inexpensive nickel catalysts, readily available zinc reductants, and robust reaction conditions makes this method highly scalable for commercial production of complex fluorinated intermediates.
Q: What is the substrate scope for the alkyne component in this reaction?
A: The method demonstrates broad substrate universality, tolerating various substituents including electron-withdrawing groups, alkyl chains, and heterocycles like pyridyl, allowing for diverse structural modifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethylnaphthalene Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the one described in CN115010575B for accelerating drug discovery pipelines. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory reactions are seamlessly translated into robust industrial processes. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Whether you require custom synthesis of complex fluorinated scaffolds or reliable supply of established building blocks, our infrastructure is designed to support your most demanding projects with precision and speed.
We invite you to collaborate with us to leverage this cutting-edge technology for your next development program. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can optimize your supply chain and reduce time-to-market for your critical therapeutic assets.
