Scalable Synthesis of Pyrrolo[1,2-A]quinoxalin-4(5H)-one via Curtius Rearrangement for Pharma Applications
Scalable Synthesis of Pyrrolo[1,2-A]quinoxalin-4(5H)-one via Curtius Rearrangement for Pharma Applications
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for complex heterocyclic scaffolds, particularly those exhibiting potent biological activity. A recent breakthrough detailed in patent CN114560862A introduces a highly efficient methodology for the synthesis of pyrrolo[1,2-A]quinoxalin-4(5H)-one and its derivatives. This chemical class is increasingly recognized for its broad spectrum of pharmacological properties, including significant anticancer activity and protein inhibition capabilities, making it a high-value target for drug discovery programs. The disclosed innovation fundamentally shifts the synthetic paradigm from traditional transition-metal catalyzed couplings to a streamlined cyclization and Curtius rearrangement sequence. For R&D directors and procurement specialists alike, this represents a pivotal opportunity to secure a reliable pharmaceutical intermediate supplier capable of delivering high-purity materials with improved cost structures. By eliminating the reliance on expensive catalysts and harsh reaction conditions, this technology not only enhances process safety but also aligns perfectly with modern green chemistry initiatives, ensuring a sustainable supply chain for critical API intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the pyrrolo[1,2-A]quinoxalin-4(5H)-one core has been fraught with significant technical and economic challenges that hinder large-scale production. As illustrated in the prior art, conventional Route 1 typically employs 2-iodo or 2-bromoaniline as the starting material, necessitating a multi-step sequence involving amino group protection, followed by a copper-catalyzed Ullmann coupling with methyl pyrrole-2-carboxylate. 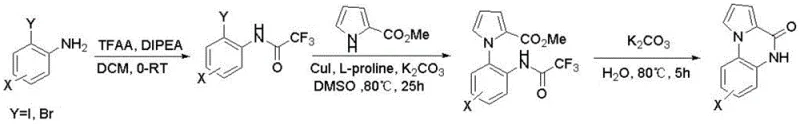 This legacy approach is severely limited by poor atom economy and notoriously low yields; literature reports indicate that the critical Ullmann coupling step often achieves yields of merely 5%, rendering the process economically unviable for commercial manufacturing. Furthermore, the requirement for cuprous iodide and L-proline catalysts introduces the risk of heavy metal contamination, necessitating costly and time-consuming purification steps to meet stringent pharmaceutical specifications. Alternative routes, such as Route 4, attempt to bypass some issues but introduce new bottlenecks, such as the requirement for microwave irradiation at extreme temperatures of 200°C, which is impractical for ton-scale production due to equipment limitations and safety concerns. These cumulative inefficiencies result in extended lead times and inflated costs, creating a substantial barrier for cost reduction in API manufacturing.
This legacy approach is severely limited by poor atom economy and notoriously low yields; literature reports indicate that the critical Ullmann coupling step often achieves yields of merely 5%, rendering the process economically unviable for commercial manufacturing. Furthermore, the requirement for cuprous iodide and L-proline catalysts introduces the risk of heavy metal contamination, necessitating costly and time-consuming purification steps to meet stringent pharmaceutical specifications. Alternative routes, such as Route 4, attempt to bypass some issues but introduce new bottlenecks, such as the requirement for microwave irradiation at extreme temperatures of 200°C, which is impractical for ton-scale production due to equipment limitations and safety concerns. These cumulative inefficiencies result in extended lead times and inflated costs, creating a substantial barrier for cost reduction in API manufacturing.
The Novel Approach
In stark contrast to these cumbersome legacy methods, the novel approach disclosed in CN114560862A leverages readily available anthranilic acid or methyl anthranilate derivatives as the foundational building blocks. This strategy elegantly bypasses the need for halogenated starting materials and transition metal catalysts entirely. The process initiates with a facile cyclization reaction using 2,5-dimethoxytetrahydrofuran in acetic acid, constructing the pyrrole ring under thermal conditions that are easily manageable in standard stainless steel reactors. Following this, the pathway proceeds through a Curtius rearrangement to close the quinoxalinone ring, a transformation known for its reliability and cleanliness. This methodological shift drastically simplifies the operational workflow, reducing the number of unit operations and eliminating the need for specialized microwave equipment. By utilizing commodity chemicals and avoiding toxic reagents like triphosgene, this new route offers a compelling value proposition for supply chain heads focused on commercial scale-up of complex heterocycles, ensuring consistent quality and uninterrupted supply continuity.
Mechanistic Insights into the Cyclization and Curtius Rearrangement Sequence
The core of this technological advancement lies in the precise orchestration of two key chemical transformations: the acid-catalyzed formation of the N-arylpyrrole and the subsequent intramolecular cyclization via the Curtius rearrangement. In the first stage, the amino group of the anthranilate derivative undergoes condensation with 2,5-dimethoxytetrahydrofuran in glacial acetic acid. The reaction is conducted at elevated temperatures between 115°C and 120°C, which provides sufficient thermal energy to drive the elimination of methanol and water, effectively aromatizing the pyrrole ring. This step is critical as it establishes the necessary spatial proximity between the nitrogen atom and the carbonyl carbon for the subsequent ring closure. The use of acetic acid serves a dual purpose as both solvent and catalyst, promoting the reaction kinetics without the need for exotic additives. Following the formation of the N-arylpyrrole intermediate, if the methyl ester variant is used, a hydrolysis step converts the ester to the corresponding carboxylic acid, preparing the molecule for rearrangement. 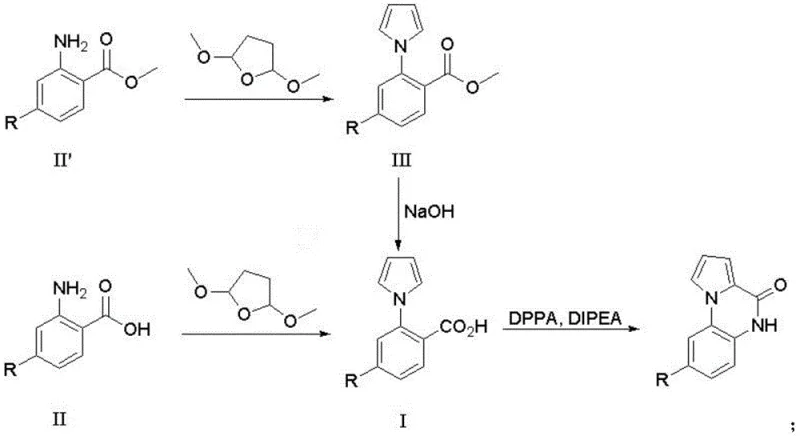 The final and most sophisticated step involves the activation of the carboxylic acid using diphenylphosphoryl azide (DPPA) in the presence of diisopropylethylamine (DIPEA). This generates an acyl azide intermediate which, upon heating to mild temperatures of 40°C to 45°C, undergoes rearrangement to an isocyanate. The proximal pyrrole nitrogen then acts as a nucleophile, attacking the isocyanate carbon to form the final seven-membered fused ring system. This intramolecular capture is highly efficient, minimizing the formation of intermolecular byproducts and ensuring high purity of the final high-purity pyrrolo[1,2-A]quinoxalin-4(5H)-one.
The final and most sophisticated step involves the activation of the carboxylic acid using diphenylphosphoryl azide (DPPA) in the presence of diisopropylethylamine (DIPEA). This generates an acyl azide intermediate which, upon heating to mild temperatures of 40°C to 45°C, undergoes rearrangement to an isocyanate. The proximal pyrrole nitrogen then acts as a nucleophile, attacking the isocyanate carbon to form the final seven-membered fused ring system. This intramolecular capture is highly efficient, minimizing the formation of intermolecular byproducts and ensuring high purity of the final high-purity pyrrolo[1,2-A]quinoxalin-4(5H)-one.
Impurity control is inherently built into this mechanism through the careful optimization of reagent stoichiometry, particularly in the Curtius step. The patent data reveals that the molar ratio of the substrate to DPPA is a critical quality attribute; deviations from the optimal 1:1.3 ratio can lead to either incomplete conversion or the generation of difficult-to-remove side products. By maintaining strict control over this parameter, the process ensures that the crude product often precipitates directly from the reaction mixture upon cooling or anti-solvent addition. This phenomenon is a significant advantage for process chemists, as it allows for purification via simple filtration and washing rather than resource-intensive column chromatography. The ability to isolate the product as a solid with LCMS purity exceeding 98% directly from the reactor demonstrates the robustness of the mechanistic design. Such high levels of purity are essential for downstream applications in medicinal chemistry, where trace impurities can confound biological assay results or compromise the safety profile of the final drug candidate.
How to Synthesize Pyrrolo[1,2-A]quinoxalin-4(5H)-one Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific operational parameters to maximize yield and safety. The process begins with the dissolution of methyl anthranilate in acetic acid, followed by the controlled addition of 2,5-dimethoxytetrahydrofuran. The mixture is then heated to reflux conditions (115-120°C) for approximately 6 hours, monitored by LC-MS to ensure complete consumption of the starting material. Upon completion, the solvent is removed under reduced pressure, and the residue is purified via vacuum distillation to isolate the pyrrole intermediate. Subsequent hydrolysis using aqueous sodium hydroxide in methanol converts the ester to the acid, which is precipitated by acidification. The final cyclization is performed in dichloromethane at 40-45°C using DPPA and DIPEA. 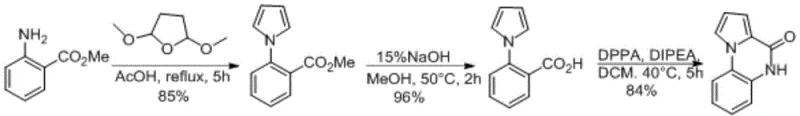
- Cyclize methyl anthranilate with 2,5-dimethoxytetrahydrofuran in acetic acid at 115-120°C to form the pyrrole ring.
- Hydrolyze the resulting ester intermediate using sodium hydroxide in methanol to obtain the carboxylic acid derivative.
- Perform Curtius rearrangement using diphenylphosphoryl azide (DPPA) and DIPEA in dichloromethane at 40-45°C to close the quinoxalinone ring.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route translates into tangible strategic advantages that extend beyond mere technical feasibility. The primary benefit lies in the drastic simplification of the raw material portfolio; by replacing expensive halogenated anilines and precious metal catalysts with commodity chemicals like anthranilic acid and acetic acid, the direct material costs are significantly reduced. This shift not only lowers the bill of materials but also mitigates the supply risk associated with fluctuating prices of specialty reagents. Furthermore, the elimination of copper catalysts removes the need for expensive scavenging resins or complex extraction protocols to meet residual metal specifications, thereby streamlining the post-reaction workup and reducing waste disposal costs. These factors collectively contribute to a more resilient and cost-effective supply chain, enabling reducing lead time for high-purity intermediates and allowing for more competitive pricing strategies in the final API market.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, driven primarily by the avoidance of noble metal catalysts and the improvement in overall yield. Traditional Ullmann couplings often suffer from yields as low as 5% to 15%, meaning that over 80% of the valuable starting material is lost as waste. In contrast, the new route achieves step yields exceeding 85% and 96% for the initial stages, leading to a much higher overall throughput. Additionally, the use of dichloromethane and acetic acid, which are standard solvents with well-established recovery infrastructure, further optimizes the operational expenditure. The simplified purification process, often requiring only filtration and washing, reduces labor hours and solvent consumption, resulting in substantial cost savings per kilogram of produced intermediate.
- Enhanced Supply Chain Reliability: From a sourcing perspective, the reliance on widely available bulk chemicals ensures a stable and continuous supply of raw materials. Unlike specialized ligands or halogenated precursors which may have limited suppliers and long lead times, anthranilic acid derivatives are produced globally at massive scales. This abundance reduces the risk of supply disruptions and provides procurement teams with greater leverage in vendor negotiations. Moreover, the robustness of the reaction conditions means that the process is less sensitive to minor variations in raw material quality, further enhancing supply chain stability. This reliability is crucial for maintaining production schedules and meeting the just-in-time delivery requirements of downstream pharmaceutical manufacturers.
- Scalability and Environmental Compliance: The process is inherently designed for scale, avoiding the pitfalls of batch-limited technologies like microwave synthesis. The thermal requirements (115-120°C and 40-45°C) are easily achievable in standard jacketed reactors, facilitating a seamless transition from gram-scale R&D to multi-ton commercial production. Environmentally, the method aligns with green chemistry principles by minimizing the use of toxic reagents like triphosgene and reducing the generation of heavy metal waste. The high atom economy and the ability to isolate products via crystallization rather than chromatography significantly reduce the volume of organic waste generated. This not only lowers waste treatment costs but also simplifies regulatory compliance regarding environmental discharge, making it an attractive option for manufacturers operating under strict environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a clear understanding of the method's capabilities and constraints. Understanding these details is vital for technical teams evaluating the feasibility of integrating this route into their existing manufacturing workflows.
Q: Why is the Curtius rearrangement route preferred over traditional Ullmann coupling for this scaffold?
A: Traditional Ullmann coupling often suffers from low yields (as low as 5-15%) and requires expensive copper catalysts and ligands. The novel Curtius rearrangement route described in patent CN114560862A offers significantly higher overall yields, avoids heavy metal contamination, and utilizes milder reaction conditions suitable for large-scale manufacturing.
Q: What are the critical process parameters for the final cyclization step?
A: The stoichiometry of diphenylphosphoryl azide (DPPA) is critical. The patent specifies that a molar ratio of 1:1.3 (substrate to DPPA) is optimal. Using less than 1.0 equivalent results in incomplete reaction, while exceeding 1.5 equivalents generates excessive impurities that complicate purification. The reaction temperature should be maintained between 40°C and 45°C in dichloromethane.
Q: Is this synthesis method suitable for industrial scale-up?
A: Yes, the method is highly scalable. Unlike prior art routes that require microwave irradiation at 200°C or toxic triphosgene, this process operates at moderate temperatures (115-120°C for cyclization, 40-45°C for rearrangement) using common solvents like acetic acid and dichloromethane. The final product often precipitates directly from the reaction mixture, simplifying isolation and reducing waste.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrrolo[1,2-A]quinoxalin-4(5H)-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the development of next-generation therapeutics. Our team of expert process chemists has thoroughly analyzed the technology disclosed in CN114560862A and is fully equipped to translate this laboratory-scale innovation into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from clinical trials to market launch without supply bottlenecks. Our state-of-the-art facilities are designed to handle the specific thermal and solvent requirements of this Curtius rearrangement process, while our rigorous QC labs guarantee that every batch meets stringent purity specifications required by global regulatory bodies.
We invite you to collaborate with us to leverage this advanced synthesis method for your drug development programs. By partnering with us, you gain access to a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this superior route. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments tailored to your specific derivative needs. Let us help you optimize your supply chain and accelerate your time to market with our reliable and high-quality pharmaceutical intermediates.
