Scalable Manufacturing of High-Purity Diazacyclooctatetraene Intermediates for Global Markets
Scalable Manufacturing of High-Purity Diazacyclooctatetraene Intermediates for Global Markets
The rapid evolution of the optoelectronic and pharmaceutical sectors demands access to sophisticated heterocyclic building blocks that combine structural complexity with commercial viability. Patent CN102584725A introduces a transformative methodology for the preparation of diazacyclooctatetraene derivatives, addressing critical bottlenecks in traditional synthetic pathways. This technology leverages a streamlined three-step sequence starting from inexpensive acid anhydrides and aromatic compounds, bypassing the instability issues associated with conventional precursors. For R&D directors and procurement specialists, this represents a significant opportunity to secure a reliable diazacyclooctatetraene supplier capable of delivering high-purity materials essential for ferroelectric liquid crystals and organic drug synthesis. The patent details a robust protocol that not only enhances yield but also drastically simplifies the operational complexity, making it an ideal candidate for cost reduction in electronic chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
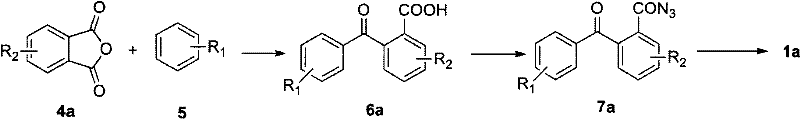
Historically, the synthesis of diazacyclooctatetraene has been plagued by reliance on unstable and expensive starting materials that hinder mass production capabilities. Traditional routes often utilize o-aminobenzaldehyde or o-aminobenzoyl compounds, which are prone to polymerization and require rigorous protection-deprotection strategies that extend the synthetic timeline. As illustrated in the prior art structures, methods employing o-aminobenzophenone derivatives or o-trifluoromethylaniline often necessitate harsh catalytic conditions involving strong Lewis acids like boron trifluoride, which demand strictly anhydrous environments to prevent catalyst deactivation. Furthermore, alternative approaches utilizing palladium-catalyzed hydrogenation introduce prohibitive costs due to the price of precious metals and the safety risks associated with high-pressure hydrogenation processes. These factors collectively result in low overall yields, typically ranging from 40% to 90%, and create significant supply chain vulnerabilities for manufacturers seeking consistent quality.
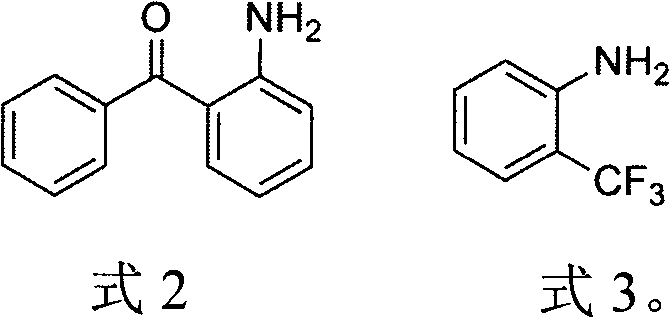
The Novel Approach
In stark contrast to these legacy methods, the patented process utilizes a highly efficient Friedel-Crafts acylation strategy followed by a novel cyclization mechanism that eliminates the need for unstable aldehyde precursors. The core innovation lies in the conversion of readily available phthalic anhydrides and simple aromatics into substituted benzoylbenzoic acids, which serve as stable platforms for subsequent functionalization. This route allows for the introduction of diverse substituents (R1 and R2 groups such as halogens, alkyl, or nitro groups) with high regioselectivity, enabling the customization of electronic properties for specific optoelectronic applications. By shifting the synthetic logic to an acyl azide intermediate, the process achieves a remarkable two-step yield of 80% for the precursor and an additional 80% yield for the final ring-closure step. This modular approach not only reduces the number of unit operations but also ensures that the commercial scale-up of complex diazacyclooctatetraene derivatives is both economically feasible and technically robust.
Mechanistic Insights into Friedel-Crafts Acylation and Acid-Catalyzed Cyclization
The success of this synthetic pathway hinges on the precise control of electrophilic aromatic substitution and the subsequent rearrangement of the acyl azide moiety. In the initial step, anhydrous aluminum chloride acts as a potent Lewis acid to activate the carbonyl group of the phthalic anhydride, facilitating nucleophilic attack by the aromatic ring to form the ketone-acid intermediate. This step is critical for establishing the carbon skeleton, and the use of excess aromatic solvent drives the equilibrium forward while minimizing polyacylation side reactions. Following chlorination with thionyl chloride, the resulting acid chloride undergoes nucleophilic substitution with sodium azide at controlled temperatures between -10°C and 50°C to generate the acyl azide. This temperature window is vital for safety and selectivity, preventing premature decomposition of the energetic azide group while ensuring complete conversion. The final cyclization occurs in an acidic medium, where protonation of the carbonyl oxygen enhances the electrophilicity of the carbonyl carbon, promoting intramolecular attack by the nitrogen species to close the eight-membered ring.
Impurity control is meticulously managed throughout the process to meet the stringent specifications required for high-purity OLED material and pharmaceutical intermediates. The patent specifies recrystallization from ethanol for the benzoylbenzoic acid intermediate, which effectively removes unreacted starting materials and inorganic salts generated during the workup. For the final diazacyclooctatetraene product, column chromatography is employed to separate the target molecule from potential isomers or oligomeric by-products formed during the thermal cyclization. The choice of acid catalyst in the final step, ranging from acetic acid to trifluoroacetic acid, allows for fine-tuning of the reaction kinetics; stronger acids can accelerate the rate but require careful monitoring to prevent degradation of the sensitive heterocyclic core. This level of mechanistic understanding ensures that the process remains reproducible across different batch sizes, providing supply chain heads with the confidence needed for reducing lead time for high-purity diazacyclooctatetraene derivatives.
How to Synthesize Diazacyclooctatetraene Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for transitioning from laboratory discovery to pilot plant production. The procedure begins with the preparation of the key benzoylbenzoic acid scaffold, followed by its conversion to the reactive acyl azide, and concludes with the thermal ring-closing step. Each stage has been optimized to balance reaction rate with safety, particularly regarding the handling of azide intermediates. Operators must adhere to strict temperature controls during the azide formation to mitigate explosion risks, while the final cyclization benefits from the use of common organic solvents that facilitate easy product isolation. The detailed standardized synthesis steps below outline the specific reagents, stoichiometry, and workup procedures required to achieve the reported yields of up to 85% for the final product.
- Perform Friedel-Crafts acylation using acid anhydride and aromatic compounds with AlCl3 to form substituted benzoylbenzoic acid.
- Convert the acid to benzoyl chloride using thionyl chloride, then react with sodium azide at -10 to 50°C to form the acyl azide intermediate.
- Execute ring closure reaction in an acidic system (e.g., acetic acid) at 20-120°C to yield the final diazacyclooctatetraene product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers substantial strategic benefits beyond mere technical feasibility. The shift away from exotic or unstable raw materials towards commodity chemicals like phthalic anhydride and benzene derivatives significantly stabilizes the upstream supply base, reducing the risk of production stoppages due to raw material shortages. Furthermore, the elimination of expensive transition metal catalysts, such as palladium, removes a major cost driver and simplifies the purification process by avoiding the need for specialized metal scavenging resins. This translates directly into cost reduction in electronic chemical manufacturing, allowing companies to maintain healthy margins even in volatile market conditions. The mild reaction conditions also imply lower energy consumption and reduced wear on reactor equipment, contributing to a more sustainable and economically efficient production lifecycle.
- Cost Reduction in Manufacturing: The replacement of precious metal catalysts and complex protecting group chemistry with inexpensive Lewis acids and simple acid anhydrides results in a drastic simplification of the bill of materials. By avoiding the use of palladium and high-pressure hydrogenation equipment, capital expenditure and operating costs are significantly lowered, while the high yields reported in the patent minimize waste disposal costs associated with low-efficiency processes.
- Enhanced Supply Chain Reliability: Sourcing stability is greatly improved as the primary inputs are bulk petrochemical derivatives available from multiple global vendors, eliminating dependence on single-source specialty suppliers. The robustness of the reaction conditions means that production is less susceptible to minor fluctuations in environmental parameters, ensuring consistent output quality and on-time delivery performance for downstream customers in the display and pharmaceutical industries.
- Scalability and Environmental Compliance: The process generates fewer hazardous by-products compared to traditional methods involving toxic amines or heavy metals, simplifying wastewater treatment and regulatory compliance. The ability to run the reaction at moderate temperatures without the need for cryogenic cooling or extreme heating facilitates easier scale-up from kilogram to tonne quantities, supporting the growing demand for these functional materials in next-generation electronic devices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for decision-makers.
Q: What are the key advantages of this synthesis route over conventional methods?
A: This method avoids unstable precursors like o-aminobenzaldehyde and eliminates the need for expensive palladium catalysts or strict anhydrous conditions required by strong Lewis acids like boron trifluoride, significantly simplifying operations.
Q: How is product purity controlled in this process?
A: High purity is achieved through specific recrystallization steps using ethanol for intermediates and column chromatography for the final product, ensuring suitability for sensitive optoelectronic applications.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the use of commercially available raw materials like phthalic anhydride and mild reaction temperatures ranging from -10°C to 120°C makes the process highly scalable and safe for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diazacyclooctatetraene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced heterocyclic intermediates play in driving innovation within the optoelectronic and pharmaceutical sectors. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from patent literature to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which employ state-of-the-art analytical techniques to verify every batch against the highest industry standards. Our infrastructure is designed to handle complex chemistries safely, including the management of energetic intermediates like acyl azides, providing our partners with peace of mind regarding supply continuity and product quality.
We invite you to collaborate with us to unlock the full potential of this innovative synthesis route for your specific applications. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements and quality needs. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize your supply chain and accelerate your time to market.
