Scalable Synthesis of Chiral Beta-Amino Acids via Mild Wolff Rearrangement for Global Pharma Supply Chains
Introduction to Advanced Chiral Synthesis Technologies
The pharmaceutical industry's relentless pursuit of high-purity chiral intermediates has led to significant innovations in synthetic methodology, particularly for complex amino acid derivatives. Patent CN112778162B introduces a robust and highly efficient preparation method for chiral beta-(Boc-amino)-5-hexynoic acid, a critical building block for various bioactive molecules. This technology leverages a sophisticated sequence involving carboxyl activation, diazomethane substitution, and a pivotal Wolff rearrangement to achieve chain homologation while strictly preserving stereochemical integrity. Unlike traditional resolution methods that often suffer from theoretical yield limitations of 50%, this synthetic route starts with readily available chiral alpha-amino acids and extends the carbon skeleton with exceptional efficiency. For R&D directors and procurement specialists seeking reliable sources of complex chiral scaffolds, this patent represents a significant advancement in process chemistry, offering a pathway that balances high yield with operational simplicity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of beta-amino acids with specific chirality has posed substantial challenges for process chemists, often relying on the resolution of racemic mixtures or asymmetric hydrogenation which can be cost-prohibitive at scale. Traditional resolution techniques, such as those utilizing chiral amines to separate enantiomers, inherently cap the maximum yield at 50% unless dynamic kinetic resolution is employed, which adds further complexity and catalyst costs. Furthermore, many existing routes require harsh reaction conditions, strong acids or bases, and elevated temperatures that can lead to racemization of the sensitive chiral centers, compromising the optical purity required for modern API manufacturing. These inefficiencies result in higher production costs, increased waste generation, and longer lead times, creating bottlenecks for supply chain managers who need consistent, high-volume delivery of key intermediates.
The Novel Approach
The methodology disclosed in CN112778162B circumvents these traditional bottlenecks by employing a direct homologation strategy that transforms chiral alpha-(Boc-amino)-4-pentynoic acid into the target beta-analogue through a streamlined four-step sequence. This novel approach utilizes mild reaction conditions, with temperatures carefully controlled between -15°C and 35°C, ensuring that the delicate Boc-protecting group and the alkyne functionality remain intact throughout the synthesis. By activating the carboxyl group with chloroformates and subsequently reacting with diazomethane, the process generates a reactive diazoketone intermediate that undergoes a silver-catalyzed Wolff rearrangement. This elegant transformation extends the carbon chain by one unit without disturbing the adjacent chiral center, delivering the target product with a combined yield of greater than or equal to 77%.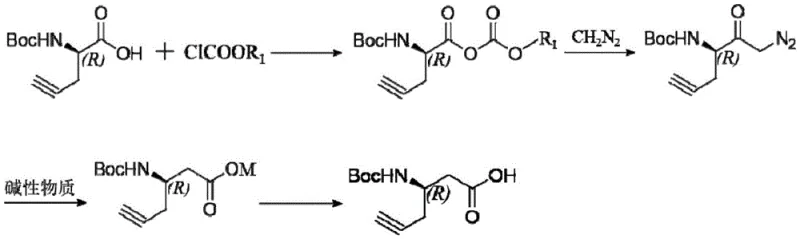
Mechanistic Insights into Silver-Catalyzed Wolff Rearrangement
The core of this synthetic innovation lies in the precise execution of the Wolff rearrangement, a powerful tool for converting alpha-diazoketones into ketenes which are subsequently trapped to form carboxylic acid derivatives. In this specific protocol, the diazoketone intermediate, generated from the reaction of the mixed anhydride with diazomethane, is subjected to a silver catalyst (such as silver acetate or silver oxide) in the presence of a mild alkaline substance. The silver catalyst facilitates the loss of nitrogen gas from the diazo group, generating a highly reactive carbene or metal-carbenoid species that rapidly rearranges to form a ketene. This rearrangement occurs with retention of configuration at the migrating chiral center, a critical feature for maintaining the optical purity of the final pharmaceutical intermediate. The subsequent nucleophilic attack by water or hydroxide ions on the ketene yields the homologated carboxylic acid salt, effectively shifting the carboxyl group from the alpha to the beta position relative to the amine.
Impurity control is meticulously managed through the selection of specific reaction parameters and workup procedures. The use of organic bases like triethylamine or N-methylmorpholine during the activation and rearrangement steps helps to neutralize acidic byproducts and stabilize the reaction mixture, preventing side reactions such as alkyne polymerization or Boc-deprotection. Furthermore, the patent specifies a post-treatment step involving phase separation where the aqueous phase containing the product salt is retained, allowing for the removal of organic-soluble impurities and silver residues before the final acidification. This careful control of the reaction environment ensures that the final product meets stringent purity specifications required for downstream coupling reactions in peptide synthesis or drug discovery programs.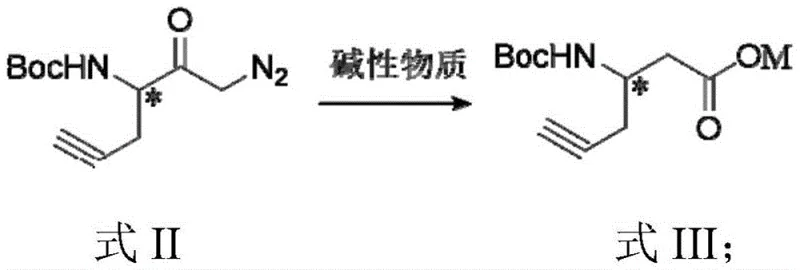
How to Synthesize Chiral Beta-(Boc-amino)-5-hexynoic Acid Efficiently
Implementing this synthesis requires strict adherence to the temperature profiles and molar ratios defined in the patent to ensure optimal yield and safety, particularly when handling reactive species like diazomethane. The process begins with the activation of the starting amino acid at low temperatures to form a mixed anhydride, followed by the careful addition of diazomethane solution to generate the diazoketone. The subsequent rearrangement step is the critical junction where the carbon chain is extended, requiring precise thermal control at 30-35°C in the presence of the silver catalyst. Detailed standard operating procedures regarding solvent selection, such as the use of tetrahydrofuran or methyl tert-butyl ether, and specific quenching protocols are essential for successful replication. For a comprehensive breakdown of the standardized synthesis steps including exact reagent quantities and safety precautions, please refer to the guide below.
- Activate chiral alpha-(Boc-amino)-4-pentynoic acid with chloroformate at 0-5°C to form the mixed anhydride intermediate.
- React the activated intermediate with diazomethane at -15 to -10°C to generate the diazoketone species.
- Perform Wolff rearrangement using a silver catalyst and alkaline substance at 30-35°C to extend the carbon chain.
- Acidify the resulting salt solution to pH 2-3 to isolate the final chiral beta-(Boc-amino)-5-hexynoic acid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible strategic benefits beyond mere chemical elegance. The reliance on easily available starting materials, such as chiral alpha-(Boc-amino)-4-pentynoic acid and common reagents like ethyl chloroformate and sodium bicarbonate, significantly de-risks the supply chain against raw material shortages. The mild reaction conditions eliminate the need for specialized high-pressure or cryogenic equipment, allowing for production in standard glass-lined or stainless steel reactors found in most multi-purpose chemical facilities. This compatibility with existing infrastructure reduces capital expenditure requirements and accelerates the timeline from process development to commercial manufacturing, ensuring a more reliable supply of critical intermediates for global pharmaceutical partners.
- Cost Reduction in Manufacturing: The elimination of expensive chiral resolution reagents and the avoidance of yield-limiting separation steps directly translate to lower cost of goods sold (COGS). By achieving high combined yields through a convergent synthetic strategy, the process minimizes raw material consumption per kilogram of final product. Furthermore, the ability to perform the reaction in common organic solvents and the use of catalytic amounts of silver salts reduce waste disposal costs and solvent recovery burdens, contributing to substantial overall cost savings in API manufacturing.
- Enhanced Supply Chain Reliability: The robustness of this method against minor fluctuations in reaction parameters enhances batch-to-b consistency, a key metric for supply chain reliability. Since the process does not rely on scarce natural products or complex biocatalysts that may have long lead times, manufacturers can maintain higher inventory levels of starting materials and respond more agilely to demand spikes. The simplified purification workflow, which relies on phase separation and crystallization rather than complex chromatography, further shortens the production cycle time, reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The process is explicitly designed for industrial scale-up, with reaction exotherms managed through controlled addition rates and moderate temperature ranges. The use of aqueous workups and the potential for solvent recycling align with green chemistry principles, reducing the environmental footprint of the manufacturing process. This compliance with increasingly stringent environmental regulations ensures long-term operational continuity and mitigates the risk of production shutdowns due to regulatory non-compliance, securing the supply chain for long-term contracts.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its integration into their supply networks. The following questions address common concerns regarding scalability, safety, and quality control associated with the Wolff rearrangement pathway. These answers are derived directly from the technical disclosures and experimental data provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers.
Q: What are the key advantages of this Wolff rearrangement method over traditional resolution?
A: This method avoids the low yields and high costs associated with chiral resolution of racemic mixtures. By starting with chiral alpha-amino acids and extending the chain via Wolff rearrangement, optical purity is preserved without the need for expensive resolving agents or multiple recrystallization steps.
Q: Is the process suitable for large-scale industrial production?
A: Yes, the patent explicitly states the process uses easily available raw materials and mild reaction conditions (temperatures ranging from -15°C to 35°C). The combined yield is reported to be greater than or equal to 77%, indicating high efficiency suitable for commercial scale-up.
Q: How is the chirality maintained during the carbon chain extension?
A: The reaction conditions are carefully controlled to prevent racemization. The use of mild bases and specific silver catalysts during the Wolff rearrangement ensures that the chiral center at the alpha-position remains intact while the carboxyl group is homologated to the beta-position.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Beta-(Boc-amino)-5-hexynoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality chiral intermediates play in the development of next-generation therapeutics. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and reliability. Our state-of-the-art facilities are equipped to handle the specific requirements of this synthesis, including the safe handling of diazomethane and the precise temperature control needed for the Wolff rearrangement. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of chiral beta-(Boc-amino)-5-hexynoic acid meets the highest international standards for pharmaceutical applications.
We invite you to collaborate with us to leverage this advanced synthetic technology for your drug development programs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this efficient route can optimize your budget. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us support your journey from clinical trials to commercial success with our superior manufacturing capabilities.
