Advanced Base-Promoted Cyclization for High-Purity 4,5-Diaryl-2H-1,2,3-Triazole Intermediates
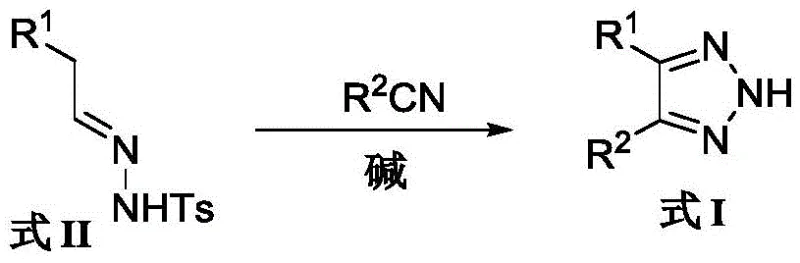
The pharmaceutical and agrochemical industries continuously demand efficient pathways to access bioactive heterocyclic scaffolds, among which the 1,2,3-triazole motif stands out for its exceptional metabolic stability and diverse biological activities. Patent CN108794412B introduces a transformative preparation method for 4,5-diaryl-2H-1,2,3-triazole compounds, addressing critical bottlenecks in existing synthetic methodologies. This innovation leverages a base-promoted cyclization between aromatic aldehyde sulfonyl hydrazones and aromatic nitriles, bypassing the safety hazards associated with azide chemistry. For R&D directors and procurement specialists, this technology represents a pivotal shift towards safer, higher-yielding, and more cost-effective manufacturing of key pharmaceutical intermediates. The method demonstrates remarkable versatility across various substituted aryl groups, ensuring broad applicability in drug discovery pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,2,3-triazoles has relied heavily on the cycloaddition of aryl alkynes with sodium azide or its derivatives. While chemically feasible, this classical approach suffers from severe drawbacks, primarily the high toxicity and explosive nature of azide reagents, which impose stringent safety protocols and inflate operational costs. Alternative strategies involving functionalized alkenes still necessitate azide participation and require complex multi-step preparations of the alkene precursors. More recently, methods utilizing cesium carbonate to promote the coupling of two aryl aldehyde sulfonyl hydrazone molecules were explored; however, these reactions frequently generate significant amounts of self-coupled byproducts alongside the desired cross-coupled triazoles. These self-coupling impurities possess polarity and molecular sizes nearly identical to the target compound, rendering purification via standard chromatography extremely difficult and drastically reducing the overall isolated yield to below 50 percent in many cases.
The Novel Approach
In stark contrast, the methodology disclosed in CN108794412B utilizes strong non-nucleophilic bases such as potassium tert-butoxide (t-BuOK) or sodium bis(trimethylsilyl)amide (NaHMDS) to drive the reaction between a sulfonyl hydrazone and a nitrile. This strategic shift eliminates the formation of self-coupled hydrazone byproducts entirely, leading to a much cleaner reaction profile. The process operates under mild thermal conditions, typically between 60°C and 100°C, and completes within a short timeframe of 3 to 4 hours. By avoiding the use of hazardous azides and minimizing side reactions, this novel route not only enhances operator safety but also significantly simplifies the workup and purification stages, thereby improving the overall process mass intensity (PMI).
Mechanistic Insights into Base-Promoted Cyclization
The core of this technological advancement lies in the precise selection of the base and the reaction stoichiometry. The use of strong bases like t-BuOK or NaHMDS facilitates the deprotonation of the sulfonyl hydrazone to generate a reactive diazo intermediate or a corresponding anionic species in situ. This activated species then undergoes a nucleophilic attack on the electrophilic carbon of the aromatic nitrile (R2CN). The molar ratio is critically optimized to 1:1.0-1.2 for the hydrazone to nitrile, with 2.5-3.0 equivalents of base, ensuring complete conversion without excessive reagent waste. This mechanistic pathway effectively suppresses the competing self-condensation of the hydrazone, which is the primary failure mode of previous cesium carbonate-catalyzed methods. The result is a highly selective formation of the 4,5-diaryl-2H-1,2,3-triazole ring system with minimal structural impurities.
From an impurity control perspective, the absence of self-coupled byproducts is a game-changer for quality assurance. In conventional hydrazone coupling, separating the target cross-coupled product from the self-coupled homodimer often requires repetitive recrystallization or preparative HPLC, which destroys yield. In this new process, the primary impurities are likely unreacted starting materials or minor decomposition products that differ significantly in polarity from the triazole product. This allows for straightforward purification using standard silica gel column chromatography with common eluent systems like petroleum ether and ethyl acetate. The high purity achieved, as evidenced by clean NMR spectra in the patent examples, ensures that the final intermediate meets the rigorous specifications required for subsequent API synthesis steps.
How to Synthesize 4,5-Diaryl-2H-1,2,3-Triazole Efficiently
To implement this synthesis effectively, one must strictly adhere to the optimized parameters regarding solvent choice and base addition. The patent highlights that solvents such as toluene, xylene, DMF, dioxane, and N-methylpyrrolidone are suitable, with DMF and toluene showing particularly robust performance across different substrates. The reaction is initiated by dissolving the sulfonyl hydrazone and nitrile in the chosen solvent, followed by the controlled addition of the base under stirring. Maintaining the temperature within the 60-80°C window is crucial for balancing reaction rate and selectivity. Detailed standardized operating procedures for scaling this reaction from gram to kilogram scale are provided in the technical guide below.
- Dissolve the aromatic aldehyde sulfonyl hydrazone and aromatic nitrile in an organic solvent such as toluene, DMF, or dioxane.
- Add a strong base, specifically potassium tert-butoxide (t-BuOK) or sodium bis(trimethylsilyl)amide (NaHMDS), under stirring conditions.
- Heat the reaction mixture to 60-100°C for 3-4 hours, then quench, extract, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers substantial strategic benefits beyond mere chemical elegance. The elimination of sodium azide removes a major regulatory and safety burden, reducing the costs associated with hazardous material handling, storage, and disposal. Furthermore, the high conversion rates and simplified purification translate directly into lower raw material consumption and reduced solvent usage, driving down the overall cost of goods sold (COGS). The reliance on readily available aromatic nitriles and aldehyde-derived hydrazones ensures a stable and resilient supply chain, mitigating the risks associated with sourcing specialized or restricted reagents.
- Cost Reduction in Manufacturing: The process achieves significantly higher yields compared to prior art, with specific examples demonstrating yields exceeding 90 percent and reaching up to 96 percent in optimized cases. This drastic improvement in efficiency means less raw material is wasted per kilogram of product produced. Additionally, the avoidance of expensive and hazardous azide reagents, combined with the elimination of complex purification steps required to remove self-coupling impurities, leads to substantial cost savings in both material and labor. The use of common organic solvents and standard bases further contributes to a leaner manufacturing budget.
- Enhanced Supply Chain Reliability: The starting materials, specifically aromatic nitriles and sulfonyl hydrazones, are commodity chemicals available from multiple global suppliers, ensuring continuity of supply. Unlike methods requiring custom-synthesized functionalized alkenes or restricted azides, this route relies on a robust and diversified vendor base. The mild reaction conditions (60-100°C) and short reaction times (3-4 hours) allow for faster batch turnover, enabling manufacturers to respond more agilely to fluctuating market demands and reduce lead times for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The protocol is inherently scalable, having been demonstrated effectively across a wide range of substrates including electron-rich, electron-deficient, and heteroaromatic systems. The absence of heavy metal catalysts simplifies the removal of trace metals, a critical requirement for API intermediates, and reduces the environmental footprint of the waste stream. The cleaner reaction profile minimizes the generation of hazardous waste, aligning with modern green chemistry principles and facilitating easier compliance with increasingly strict environmental regulations in chemical manufacturing zones.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity on process robustness and product quality. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for commercial production.
Q: How does this method improve upon traditional azide-based triazole synthesis?
A: Unlike traditional methods relying on toxic and explosive sodium azide, this patent utilizes stable aromatic nitriles and sulfonyl hydrazones. Furthermore, it avoids the self-coupling impurities common in cesium carbonate-promoted reactions, resulting in significantly higher purity and easier downstream processing.
Q: What are the optimal reaction conditions for maximizing yield?
A: The patent specifies using strong bases like t-BuOK or NaHMDS in solvents such as DMF or toluene. The optimal temperature range is 60-80°C with a reaction time of 3-4 hours, achieving yields up to 96% depending on the substrate substituents.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process operates at moderate temperatures (60-100°C) and uses commercially available reagents. The elimination of hazardous azides and the high conversion rates make it highly scalable and safer for industrial production environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,5-Diaryl-2H-1,2,3-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your drug development programs. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to manufacturing plant is seamless. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 4,5-diaryl-2H-1,2,3-triazole delivered meets the highest international standards. We are committed to leveraging advanced synthetic technologies like the one described in CN108794412B to provide our clients with superior value.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your project volume. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing us to demonstrate our capability to deliver cost reduction in pharmaceutical intermediate manufacturing while maintaining uncompromised quality and safety.
