Scalable Synthesis of Isoindoline Acetic Acid Intermediates via Efficient Palladium Catalysis
The pharmaceutical industry continuously seeks robust synthetic pathways for complex heterocyclic intermediates that serve as the backbone for next-generation therapeutics. Patent CN115215835A, published on October 21, 2022, introduces a groundbreaking methodology for the preparation of 2-(2,6-dioxopiperidine-3-yl)-1-oxoisoindoline-4-yl acetic acid, a critical building block in the synthesis of immunomodulatory imide drugs (IMiDs). This innovation addresses long-standing challenges in the field by replacing cumbersome multi-step sequences with a streamlined, two-step catalytic process. The disclosed method leverages advanced organometallic chemistry to achieve superior selectivity and yield, marking a significant leap forward for manufacturers aiming to optimize their production of high-value pharmaceutical intermediates. By focusing on safety and efficiency, this technology offers a compelling value proposition for global supply chains.
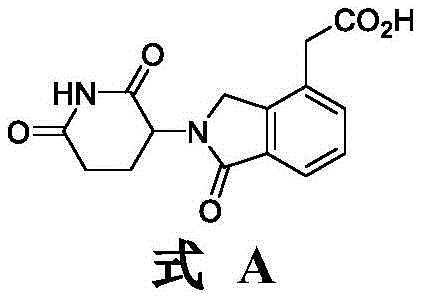
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of isoindoline derivatives such as the target compound has been plagued by inefficient and hazardous protocols. Prior art, specifically referenced as CN107739389A, relies on a tedious four-step sequence initiating from lenalidomide. This legacy route necessitates the use of highly toxic diazotization reagents and peroxides, creating severe safety hazards for plant operators and generating substantial quantities of difficult-to-treat chemical waste. Furthermore, the coupling step in the third stage of the conventional process suffers from low yields and generates byproducts that are notoriously difficult to separate, leading to poor overall purity and increased downstream processing costs. These factors collectively render traditional methods unsuitable for modern, green manufacturing standards and limit the ability to scale production to meet growing market demand without incurring prohibitive expenses.
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent simplifies the entire synthetic landscape into a concise two-step operation that maximizes atom economy and operational safety. The process initiates with a palladium-catalyzed cross-coupling reaction, specifically utilizing a Negishi coupling strategy to install the acetic acid side chain precursor directly onto the isoindoline core. This is immediately followed by a mild acidic hydrolysis to reveal the final carboxylic acid functionality. By bypassing the need for diazotization and reducing the step count by half, the new route drastically minimizes solvent consumption and waste generation. The use of stable zinc reagents and standard palladium catalysts ensures that the reaction proceeds with high conversion rates under controlled conditions, facilitating easier purification and delivering a product with a significantly improved impurity profile suitable for stringent pharmaceutical applications.
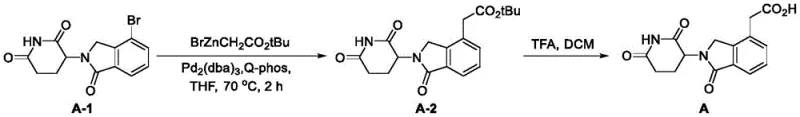
Mechanistic Insights into Pd-Catalyzed Negishi Coupling
The core of this technological breakthrough lies in the efficient execution of the Negishi coupling reaction, which connects the brominated isoindoline scaffold with the zinc-functionalized acetate fragment. The mechanism involves the oxidative addition of the aryl bromide bond of Compound A-1 to the zero-valent palladium center, facilitated by the bulky and electron-rich Q-Phos ligand. This ligand choice is critical as it stabilizes the active catalytic species and promotes the subsequent transmetallation step with the organozinc reagent. The use of THF as the solvent provides an optimal coordination environment for the zinc species, ensuring high solubility and reactivity. Following transmetallation, reductive elimination occurs rapidly to forge the new carbon-carbon bond, releasing the ester-intermediate Compound A-2 and regenerating the active palladium catalyst for the next cycle. This catalytic cycle is highly robust, tolerating the sensitive imide functionalities present in the substrate without causing degradation or racemization.
Impurity control is inherently built into this mechanistic design through the high chemoselectivity of the palladium system. Unlike radical-based or harsh nucleophilic substitutions that might attack the lactam rings, the mild nature of the Negishi coupling preserves the integrity of the piperidine-2,6-dione moiety. The subsequent hydrolysis step utilizes trifluoroacetic acid (TFA) in dichloromethane, a condition that selectively cleaves the tert-butyl ester protecting group while leaving the cyclic imide structures intact. This orthogonality prevents the formation of ring-opened byproducts or hydrolysis of the isoindolinone core, which are common failure modes in less optimized processes. The result is a crude product profile that is exceptionally clean, reducing the burden on final crystallization or chromatography steps and ensuring that the final API intermediate meets rigorous quality specifications.
How to Synthesize 2-(2,6-Dioxopiperidine-3-yl)-1-oxoisoindoline-4-yl Acetic Acid Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize the reported yields of 72% for the coupling step and 99% for the hydrolysis. The process begins by dissolving the bromo-starting material in anhydrous tetrahydrofuran under an inert argon atmosphere to prevent catalyst deactivation. The addition of the palladium source and ligand must be followed by the careful introduction of the organozinc reagent, after which the mixture is heated to 70°C to drive the coupling to completion. Detailed standard operating procedures regarding quenching, extraction, and purification are essential to maintain this high level of performance. For the complete standardized synthesis protocol including specific molar ratios and workup details, please refer to the structured guide below.
- Perform a Negishi coupling reaction by reacting 3-(4-bromo-1-oxoisoindolin-2-yl)piperidine-2,6-dione with (2-(tert-butoxy)-2-oxoethyl)zinc bromide using a Pd2(dba)3 catalyst and Q-Phos ligand in THF at 70°C.
- Quench the reaction with saturated ammonium chloride, extract with ethyl acetate, and purify the intermediate ester (Compound A-2) via column chromatography.
- Hydrolyze the tert-butyl ester group of Compound A-2 using trifluoroacetic acid (TFA) in dichloromethane at room temperature to yield the final carboxylic acid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route translates into tangible strategic benefits that extend far beyond simple chemical transformation. The reduction in synthetic steps from four to two inherently lowers the operational overhead, as fewer unit operations mean less equipment time, reduced labor costs, and lower utility consumption per kilogram of product. Moreover, the elimination of hazardous diazotization reagents removes the need for specialized containment infrastructure and expensive waste disposal protocols associated with toxic nitrogenous byproducts. This shift not only enhances the safety profile of the manufacturing facility but also streamlines regulatory compliance, allowing for faster batch release and reduced risk of production stoppages due to environmental or safety audits.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the dramatic improvement in overall yield and the simplification of the purification workflow. By achieving near-quantitative conversion in the final hydrolysis step and a robust 72% yield in the coupling stage, the amount of raw material required to produce a fixed quantity of the final intermediate is significantly reduced. Additionally, the avoidance of expensive and dangerous reagents like peroxides lowers the direct material costs. The simplified impurity profile means that resource-intensive purification techniques, such as repeated recrystallizations or preparative HPLC, can often be replaced by more economical standard column chromatography or direct crystallization, further driving down the cost of goods sold (COGS) for this critical pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the use of commercially abundant and stable starting materials. The key reagents, including the bromo-isoindolinone precursor and the organozinc species, are readily available from multiple global suppliers, mitigating the risk of single-source bottlenecks. The mild reaction conditions, operating at moderate temperatures and atmospheric pressure, reduce the dependency on specialized high-pressure reactors or cryogenic cooling systems, allowing the synthesis to be performed in a wider range of multipurpose manufacturing facilities. This flexibility ensures that production can be scaled up or shifted between sites with minimal capital expenditure, guaranteeing a steady flow of materials to downstream API manufacturers even during periods of high market volatility.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route represents a substantial advancement towards green chemistry principles. The significant reduction in the number of reaction steps directly correlates to a lower E-factor, meaning less waste is generated per unit of product. The absence of heavy metal contaminants from harsh oxidation steps and the use of standard palladium catalysts, which can often be recovered or scavenged efficiently, simplify the wastewater treatment process. This makes the process highly scalable from pilot plant to commercial tonnage without encountering the exponential increase in waste management costs that typically plagues older, linear synthetic routes, ensuring long-term sustainability and compliance with increasingly strict global environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis technology. These insights are derived directly from the experimental data and technical disclosures within the patent documentation, providing a clear understanding of the process capabilities. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the primary advantages of this new synthesis route over prior art methods?
A: Unlike previous methods requiring four steps and toxic diazotization reagents, this novel route utilizes a direct two-step Negishi coupling and hydrolysis sequence. This significantly reduces waste generation, eliminates hazardous peroxide usage, and improves overall yield and purification efficiency.
Q: What specific catalyst system is recommended for optimal conversion?
A: The patent specifies a robust catalytic system comprising Pd2(dba)3 as the palladium source and Q-Phos as the ligand. This combination ensures high conversion rates at moderate temperatures (70°C) while minimizing side reactions common in complex heterocyclic synthesis.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method is explicitly designed for industrial scalability. It avoids explosive reagents, operates under mild conditions, and utilizes commercially available starting materials like zinc reagents and bromo-isoindolinones, ensuring a reliable supply chain for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(2,6-Dioxopiperidine-3-yl)-1-oxoisoindoline-4-yl Acetic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the development of life-saving medicines. Our team of expert chemists has thoroughly analyzed the technology disclosed in CN115215835A and is fully prepared to leverage this advanced Negishi coupling methodology for your projects. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of isoindoline intermediate we deliver adheres to the highest quality standards required by the global pharmaceutical industry.
We invite you to collaborate with us to unlock the full potential of this cost-effective and safe manufacturing process. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to discuss your project needs, obtain specific COA data for our reference standards, and receive comprehensive route feasibility assessments that will accelerate your drug development timeline and optimize your supply chain economics.
