Scalable Electrocatalytic Synthesis of Xanthoisoxazoline B for Commercial Pharmaceutical Applications
The pharmaceutical industry is currently witnessing a paradigm shift towards sustainable manufacturing processes, driven by the urgent need to reduce environmental footprints while maintaining high efficiency. Patent CN112609202B introduces a groundbreaking method for the total synthesis of the natural product Xanthoisoxazoline B, utilizing advanced organic electrocatalysis. This technology represents a significant departure from traditional thermal synthesis, offering a cleaner, more energy-efficient pathway to access complex spiroisoxazoline alkaloids. The method leverages electricity as a traceless reagent to drive oxidative dearomatization, a critical step in constructing the spirocyclic core, thereby eliminating the need for stoichiometric amounts of hazardous chemical oxidants. For R&D directors and procurement managers alike, this patent signals a new era of viable commercial production for high-value natural product derivatives with potent antibacterial and anticancer properties.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for complex spiroisoxazoline alkaloids often rely on harsh reaction conditions that pose significant challenges for industrial scale-up. Conventional methods typically require high temperatures, high pressures, and the use of toxic heavy metal oxidants or stoichiometric reagents that generate substantial amounts of hazardous waste. These factors not only increase the operational costs associated with waste treatment and regulatory compliance but also introduce safety risks in large-scale manufacturing environments. Furthermore, traditional thermal oxidation processes often suffer from poor selectivity, leading to complex impurity profiles that require extensive and costly downstream purification steps. The reliance on non-renewable chemical oxidants also contradicts the growing global mandate for green chemistry principles in pharmaceutical manufacturing, making these older routes less attractive for long-term commercial investment.
The Novel Approach
The novel approach detailed in patent CN112609202B overcomes these historical barriers by integrating electrochemical technology into the synthetic workflow. By utilizing anodic oxidation, the process achieves dearomatization under mild conditions, typically at room temperature, which significantly reduces energy consumption compared to thermal methods. The use of electricity as the primary oxidant means that the only byproduct is often hydrogen gas, drastically simplifying the waste stream and reducing the environmental impact. This method also demonstrates excellent functional group tolerance, allowing for the use of diverse 4-substituted phenethylamine starting materials without the need for extensive protecting group strategies. The result is a streamlined, three-step synthesis that is not only chemically elegant but also economically superior for commercial production.
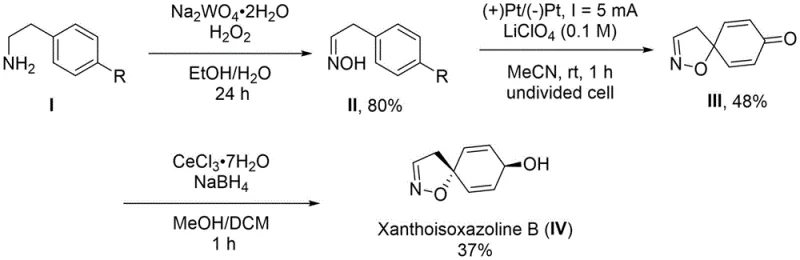
Mechanistic Insights into Electrocatalytic Dearomatization and Luche Reduction
The core innovation of this synthesis lies in the electrochemical dearomatization step, which constructs the spiroisoxazoline skeleton with high precision. In this stage, the oxime intermediate undergoes anodic oxidation at a platinum electrode surface in the presence of a supporting electrolyte like lithium perchlorate. The electrochemical potential drives the removal of electrons from the aromatic system, generating a reactive cationic intermediate that spontaneously undergoes intramolecular cyclization. This mechanism avoids the use of aggressive chemical oxidants that might over-oxidize sensitive functional groups, thereby preserving the integrity of the molecule. The control over the current density allows chemists to fine-tune the reaction rate and selectivity, ensuring that the desired spirocyclic product is formed preferentially over potential side products, which is critical for maintaining high yields in a manufacturing setting.
Following the electrochemical step, the synthesis employs a Luche reduction to establish the final stereochemistry of the Xanthoisoxazoline B product. This step utilizes cerium chloride and sodium borohydride in a mixed solvent system of methanol and dichloromethane at low temperatures. The presence of cerium salts modifies the reactivity of the borohydride, favoring 1,2-reduction of the ketone over 1,4-reduction, which is essential for obtaining the correct allylic alcohol configuration. This chemoselective reduction ensures that the sensitive isoxazoline ring remains intact while the ketone is efficiently converted to the hydroxyl group. The combination of electrochemical oxidation and chemoselective reduction creates a robust synthetic sequence that minimizes impurity generation and simplifies the purification process, directly addressing the purity concerns of R&D directors.

How to Synthesize Xanthoisoxazoline B Efficiently
The synthesis of Xanthoisoxazoline B is achieved through a concise three-step sequence that begins with the oxidation of readily available 4-substituted phenethylamine. The initial step involves treating the amine with sodium tungstate and hydrogen peroxide in ethanol to generate the corresponding oxime intermediate in high yield. This oxime is then subjected to the key electrochemical dearomatization step in an undivided cell using platinum electrodes and acetonitrile as the solvent. Finally, the resulting spiroisoxazoline ketone is reduced using cerium chloride and sodium borohydride to afford the target natural product. The detailed standardized synthesis steps, including specific molar ratios, current densities, and workup procedures, are provided in the guide below for technical reference.
- Oxidize 4-substituted phenethylamine using sodium tungstate and hydrogen peroxide to form the oxime intermediate.
- Perform anodic oxidation dearomatization under electrochemical catalysis using platinum electrodes to generate the spiroisoxazoline core.
- Execute a stereoselective Luche reduction using cerium chloride and sodium borohydride to yield the final Xanthoisoxazoline B product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this electrocatalytic route offers tangible strategic advantages beyond mere chemical novelty. The primary benefit lies in the drastic simplification of the supply chain for reagents; by replacing expensive and hazardous chemical oxidants with electricity, the process reduces dependency on volatile raw material markets. This shift significantly lowers the cost of goods sold (COGS) by eliminating the procurement and disposal costs associated with toxic oxidizing agents. Furthermore, the mild reaction conditions reduce the wear and tear on manufacturing equipment, extending asset life and reducing maintenance downtime. The stability of the intermediates also allows for more flexible production scheduling, enhancing the overall reliability of the supply chain for this high-value pharmaceutical intermediate.
- Cost Reduction in Manufacturing: The elimination of stoichiometric chemical oxidants removes a major cost driver from the bill of materials. Additionally, the reduced need for complex waste treatment infrastructure lowers the overhead costs associated with environmental compliance. The energy efficiency of the electrochemical step further contributes to lower utility costs per kilogram of product produced. These factors combine to create a more cost-competitive manufacturing process that can withstand market fluctuations in raw material pricing.
- Enhanced Supply Chain Reliability: The starting materials, specifically 4-substituted phenethylamines, are commodity chemicals with robust global supply chains, ensuring consistent availability. The synthetic route avoids the use of exotic catalysts or reagents that are prone to supply shortages. The simplicity of the three-step process also reduces the number of unit operations required, minimizing the risk of bottlenecks or delays in production. This reliability is crucial for maintaining continuous supply to downstream pharmaceutical customers who require just-in-time delivery of critical intermediates.
- Scalability and Environmental Compliance: Electrochemical reactors are inherently scalable, allowing for a smooth transition from laboratory benchtop to pilot plant and full commercial production. The clean nature of the reaction, producing minimal hazardous waste, simplifies the permitting process for new manufacturing facilities. This environmental advantage aligns with the sustainability goals of major pharmaceutical companies, making this supplier a preferred partner for green chemistry initiatives. The ability to scale without compromising safety or environmental standards ensures long-term viability of the production process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of Xanthoisoxazoline B synthesized via this patented method. These answers are derived directly from the technical specifications and beneficial effects outlined in the patent documentation. They are intended to provide clarity on the feasibility, safety, and quality aspects of the manufacturing process for potential partners and stakeholders.
Q: What are the key advantages of using electrocatalysis for Xanthoisoxazoline B synthesis?
A: The electrocatalytic method replaces toxic chemical oxidants with electrons, significantly reducing hazardous waste generation and lowering energy consumption compared to traditional high-temperature thermal processes.
Q: Is the starting material for this synthesis readily available for large-scale production?
A: Yes, the process utilizes 4-substituted phenethylamine derivatives which are commercially abundant and cost-effective, ensuring a stable supply chain for manufacturing scale-up.
Q: How does this route ensure high purity for pharmaceutical applications?
A: The mild reaction conditions and specific selectivity of the Luche reduction step minimize byproduct formation, facilitating easier purification and meeting stringent pharmaceutical purity specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Xanthoisoxazoline B Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating advanced academic research into commercial reality, leveraging deep expertise in organic electrocatalysis and natural product synthesis. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the innovative route described in patent CN112609202B can be effectively implemented at an industrial level. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Xanthoisoxazoline B meets the exacting standards required for pharmaceutical research and development. Our commitment to quality and technical excellence makes us the ideal partner for companies seeking to secure a stable supply of this bioactive natural product.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By partnering with our technical procurement team, you can access specific COA data and route feasibility assessments that demonstrate the tangible benefits of this electrocatalytic process. Let us help you optimize your supply chain and reduce costs while accessing high-purity intermediates for your next generation of therapeutic candidates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
