Revolutionizing Disulfoxide Manufacturing: Metal-Free Electrochemical Oxidation for Global Supply Chains
Revolutionizing Disulfoxide Manufacturing: Metal-Free Electrochemical Oxidation for Global Supply Chains
The landscape of fine chemical synthesis is undergoing a paradigm shift driven by the urgent need for greener, more efficient manufacturing processes. Patent CN112391644B introduces a groundbreaking preparation method for disulfoxide compounds that fundamentally alters the traditional approach to sulfide oxidation. By leveraging electrocatalytic oxidation, this technology replaces conventional chemical oxidants and transition metal catalysts with electricity, offering a pathway to significantly lower production costs and enhanced environmental compliance. For R&D directors and procurement managers in the pharmaceutical and agrochemical sectors, this innovation represents a critical opportunity to optimize supply chains for high-value intermediates. The method utilizes cheap and easily available acetic acid as an acid additive, demonstrating that high-efficiency synthesis does not require exotic or expensive reagents. This report analyzes the technical merits and commercial implications of this patent, providing a strategic roadmap for integrating this technology into global manufacturing networks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of sulfoxides and disulfoxides has relied heavily on the catalytic oxidation of corresponding sulfides using stoichiometric amounts of strong oxidants. Traditional protocols frequently employ hazardous reagents such as trifluoroperacetic acid, nitric acid mixed with sulfuric acid, nitromethane, iodic acid, or various high-valent iodine compounds. These conventional methods present severe drawbacks that impact both the bottom line and operational safety profiles of chemical manufacturers. The reliance on expensive oxidants drives up raw material costs, while the generation of stoichiometric byproducts creates substantial waste disposal challenges and environmental liabilities. Furthermore, many of these oxidation reactions require harsh conditions that can compromise the integrity of sensitive functional groups on the substrate, leading to lower selectivity and difficult purification processes. The inherent risks associated with handling strong oxidizing agents also necessitate specialized equipment and rigorous safety protocols, further inflating the capital expenditure required for production facilities.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in patent CN112391644B utilizes an electrochemical cell to drive the oxidation process, effectively using electrons as clean reagents. This novel approach eliminates the need for transition metal catalysts and external chemical oxidants entirely, relying instead on a simple system comprising an electrolyte, an organic solvent, water, and a benign organic acid additive. The reaction proceeds under mild conditions, specifically at room temperature with constant current stirring, which dramatically reduces energy consumption and thermal hazards. By substituting dangerous chemical oxidants with electrical energy, the process not only enhances safety but also simplifies the reaction workup, as there are no heavy metal residues or complex oxidant byproducts to remove. This shift from chemical to electrochemical oxidation represents a significant technological leap, enabling the production of high-purity disulfoxide compounds with yields reported as high as 90% in specific embodiments, thereby offering a robust solution for the commercial scale-up of complex pharmaceutical intermediates.
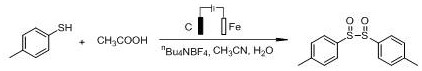
Mechanistic Insights into Electrocatalytic Oxidation of Sulfides
The core mechanism of this invention revolves around the anodic oxidation of sulfide substrates within an undivided electrochemical cell. In this system, the sulfide molecule—whether it is a thiophenol, a disulfide, or a thiol—undergoes electron transfer at the anode surface, generating a reactive cation radical or sulfonium intermediate. The presence of the organic acid, such as acetic acid, plays a crucial role in facilitating proton transfer and stabilizing intermediate species, ensuring the reaction proceeds selectively towards the sulfoxide state rather than over-oxidizing to the sulfone. The electrolyte, typically a quaternary ammonium salt like nBu4NBF4, nBu4NPF6, or nBu4NClO4, ensures sufficient conductivity in the organic solvent medium, allowing for efficient current flow at low amperage (2.5-3.5 mA). This precise control over the electrochemical potential allows for exceptional chemoselectivity, preserving other sensitive functional groups on the aromatic ring that might otherwise be attacked by aggressive chemical oxidants. The inclusion of a small amount of water in the acetonitrile solvent system is also critical, likely serving as the oxygen source for the formation of the sulfoxide moiety through an electro-generated hydroxyl species or direct interaction with the activated sulfide.
From an impurity control perspective, this electrochemical mechanism offers distinct advantages over traditional catalytic cycles. Since no transition metals are involved, the risk of metal contamination in the final active pharmaceutical ingredient (API) intermediate is completely eradicated, a critical parameter for regulatory compliance in drug manufacturing. The absence of stoichiometric oxidant byproducts means the impurity profile is significantly cleaner, primarily consisting of unreacted starting material or minor over-oxidation products that are easily separated. The high reaction selectivity observed, with yields reaching up to 90% in optimized conditions, indicates that side reactions such as C-S bond cleavage or polymerization are effectively suppressed. This mechanistic clarity allows process chemists to predict scalability with greater confidence, as the reaction rate is directly controlled by the applied current, providing a linear and tunable parameter for process optimization that is often lacking in batch chemical oxidation methods.
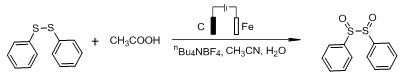
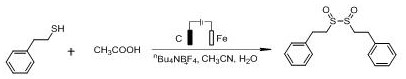
How to Synthesize Disulfoxide Compounds Efficiently
The synthesis protocol outlined in the patent provides a straightforward and reproducible method for generating disulfoxide compounds suitable for industrial application. The process begins with the precise weighing of the sulfide starting material and the electrolyte, which are loaded into a reaction vessel equipped with stirring capabilities. Subsequently, the organic solvent, typically acetonitrile, is added along with the organic acid additive and a controlled amount of water to create the electrolytic medium. The reaction is then initiated by applying a constant current at room temperature, with progress monitored via standard analytical techniques such as TLC or GC-MS. Upon completion, the workup involves a simple concentration step to remove volatile solvents, followed by purification using column chromatography with ethyl acetate and petroleum ether. This streamlined workflow minimizes unit operations and reduces the overall processing time, making it an attractive candidate for rapid process development. Detailed standardized synthesis steps for specific substrates are provided in the technical guide below to ensure consistent replication of these high-yield results.
- Prepare the reaction mixture by adding the sulfide starting material (thiophenol, disulfide, or thiol) and electrolyte (e.g., nBu4NBF4) into a reaction tube, followed by organic solvent, organic acid, and water.
- Conduct the electrolytic reaction under constant temperature and constant current conditions (2.5-3.5 mA) with continuous stirring to facilitate the oxidation process.
- Upon completion, remove volatile solvents from the reaction liquid under reduced pressure and purify the crude product via column chromatography to isolate the high-purity disulfoxide compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this electrochemical synthesis route offers transformative benefits that extend far beyond simple yield improvements. The primary value driver is the drastic simplification of the raw material portfolio; by eliminating the need for expensive, hazardous, and often supply-constrained chemical oxidants, manufacturers can achieve significant cost reduction in pharmaceutical intermediate manufacturing. The reliance on commodity chemicals like acetic acid and common electrolytes ensures a stable and predictable supply chain, mitigating the risks associated with sourcing specialty reagents. Furthermore, the removal of transition metal catalysts eliminates the need for costly and time-consuming metal scavenging steps during downstream processing, which directly translates to reduced production cycles and lower operational expenditures. The mild reaction conditions also imply lower energy costs for heating or cooling, contributing to a more sustainable and economically viable production model that aligns with modern green chemistry mandates.
- Cost Reduction in Manufacturing: The elimination of stoichiometric oxidants and transition metal catalysts fundamentally alters the cost structure of disulfoxide production. Traditional methods incur high costs not only from the reagents themselves but also from the waste treatment required for toxic byproducts. By using electricity as the oxidant and acetic acid as a promoter, the variable cost per kilogram of product is substantially decreased. Additionally, the simplified purification process reduces solvent consumption and labor hours associated with complex workups. This economic efficiency allows suppliers to offer more competitive pricing for high-purity disulfoxide compounds while maintaining healthy margins, providing a strategic advantage in price-sensitive markets.
- Enhanced Supply Chain Reliability: Dependence on specialized oxidants often introduces fragility into the supply chain, as these materials may be subject to regulatory restrictions or logistical bottlenecks. This electrochemical method relies on universally available materials such as acetonitrile, acetic acid, and standard salts, ensuring that production can continue uninterrupted even during global supply disruptions. The robustness of the reaction conditions, which tolerate ambient temperatures and do not require cryogenic cooling or high-pressure vessels, further enhances operational reliability. This stability is crucial for reducing lead time for high-purity disulfoxides, allowing manufacturers to respond more agilely to fluctuating demand from downstream API producers without the risk of raw material stockouts.
- Scalability and Environmental Compliance: Scaling electrochemical processes is increasingly feasible with modern flow chemistry and plate-and-frame reactor technologies, making this method highly suitable for commercial scale-up of complex sulfoxides. The absence of hazardous waste streams simplifies environmental compliance, reducing the burden on wastewater treatment facilities and lowering the carbon footprint of the manufacturing process. High atom economy and the generation of fewer byproducts mean that the process is inherently greener, aligning with the sustainability goals of major multinational corporations. This environmental advantage not only mitigates regulatory risk but also enhances the brand value of the supply chain, appealing to end-users who prioritize eco-friendly sourcing strategies in their vendor selection criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electrochemical synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in patent CN112391644B, ensuring accuracy and relevance for technical decision-makers. Understanding these details is essential for evaluating the feasibility of adopting this method for specific project requirements. The responses cover aspects of safety, cost efficiency, and scalability to provide a comprehensive overview of the technology's value proposition.
Q: How does this electrochemical method improve safety compared to traditional oxidation?
A: Traditional methods often rely on hazardous oxidants like trifluoroperacetic acid or nitric acid, which pose significant safety risks and generate toxic byproducts. This patented electrochemical approach operates at room temperature using electricity as the primary oxidant, eliminating the need for dangerous chemical oxidants and significantly enhancing operational safety.
Q: What are the cost advantages of using acetic acid as an additive?
A: Acetic acid is a cheap, readily available commodity chemical compared to specialized catalytic systems or expensive stoichiometric oxidants. By utilizing acetic acid as the acid additive in conjunction with electrocatalysis, the process drastically reduces raw material costs and simplifies the supply chain for acid reagents.
Q: Can this method be scaled for industrial production of API intermediates?
A: Yes, the method is designed for scalability. It features a simple catalytic system, mild reaction conditions, and high synthesis efficiency with yields reaching up to 90%. The absence of transition metals simplifies downstream purification, making it highly suitable for the commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Disulfoxide Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in the production of high-value fine chemicals. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN112391644B can be successfully translated into robust industrial processes. Our commitment to quality is unwavering, with stringent purity specifications and rigorous QC labs dedicated to verifying the identity and purity of every batch of disulfoxide compounds we produce. We understand that consistency is key in the pharmaceutical supply chain, and our advanced manufacturing facilities are equipped to handle the specific requirements of electrochemical reactions, including precise current control and specialized electrode materials.
We invite global partners to collaborate with us to leverage this cutting-edge technology for their specific intermediate needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis to quantify the economic benefits of switching to this metal-free route for your specific molecule. We encourage you to reach out for specific COA data and route feasibility assessments to determine how this electrochemical oxidation method can optimize your supply chain. Our team is ready to provide the technical support and commercial flexibility required to secure your supply of critical sulfoxide intermediates, ensuring continuity and competitiveness in your final drug product manufacturing.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
