Advanced Photocatalytic Synthesis of Heterocyclyl Cyclopropane Compounds for Commercial Scale-Up
Advanced Photocatalytic Synthesis of Heterocyclyl Cyclopropane Compounds for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust and efficient synthetic routes to access complex molecular scaffolds, particularly those containing strained ring systems like cyclopropanes which are prevalent in bioactive molecules. Patent CN112898202A introduces a groundbreaking methodology for the preparation of heterocyclyl cyclopropane compounds through a visible-light-mediated photocatalytic reaction. This innovation represents a significant departure from conventional synthetic strategies, offering a pathway that is not only chemically elegant but also industrially viable. By utilizing a specific silicon-based reagent in conjunction with modern photocatalysts, the process enables the direct functionalization of heterocyclic precursors under exceptionally mild conditions. For R&D directors and process chemists, this technology opens new avenues for constructing spiro-cyclopropane indole derivatives, a structural motif often associated with enhanced metabolic stability and biological activity in drug candidates. The ability to forge these carbon-carbon bonds without extreme temperatures or hazardous reagents positions this method as a cornerstone for next-generation intermediate synthesis.
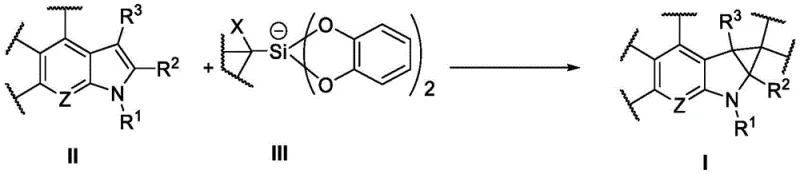
The strategic value of this patent lies in its versatility and the high degree of control it offers over the reaction outcome. The disclosed method accommodates a wide range of substituents on the heterocyclic core, allowing for the rapid generation of diverse chemical libraries essential for lead optimization campaigns. Furthermore, the use of visible light as the energy source aligns perfectly with the global push towards greener chemistry and sustainable manufacturing practices. As a reliable pharmaceutical intermediate supplier, understanding and adopting such cutting-edge technologies is crucial for maintaining a competitive edge in the market. The transition from batch processes relying on thermal activation to continuous flow or batch processes driven by photonics can drastically reduce energy consumption and improve safety profiles, directly impacting the bottom line for procurement and supply chain teams looking for cost-effective solutions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of cyclopropane-fused heterocycles has been fraught with challenges that limit their widespread application in process chemistry. Traditional approaches often rely on the generation of highly reactive carbene intermediates, which typically require harsh reaction conditions such as strong bases, elevated temperatures, or the use of toxic heavy metal catalysts. These severe conditions can lead to poor functional group tolerance, resulting in low yields and complex purification profiles due to the formation of numerous side products. Moreover, the instability of carbene species necessitates careful handling and specialized equipment, increasing the operational complexity and safety risks associated with scale-up. For manufacturing facilities, the disposal of heavy metal waste and the management of exothermic reactions pose significant environmental and regulatory hurdles. Consequently, many promising drug candidates containing these motifs have faced delays or increased costs during the development phase due to the lack of a scalable and robust synthetic route.
The Novel Approach
In stark contrast, the novel photocatalytic approach described in the patent circumvents these issues by employing a mild, redox-neutral mechanism driven by visible light. The reaction utilizes a stable silicon-containing reagent, specifically an iodomethyl silicate derivative, which acts as a radical precursor under photo-irradiation. This eliminates the need for unstable carbenes and allows the reaction to proceed at room temperature, typically between 30 to 40 degrees Celsius. The compatibility with a broad spectrum of functional groups, including esters, ketones, and sulfonamides, ensures that complex molecules can be synthesized without extensive protecting group strategies. This simplification of the synthetic sequence not only enhances the overall yield but also streamlines the downstream processing, making it an ideal candidate for cost reduction in pharmaceutical intermediate manufacturing. The shift towards such mild conditions represents a paradigm shift in how we approach the construction of strained ring systems.
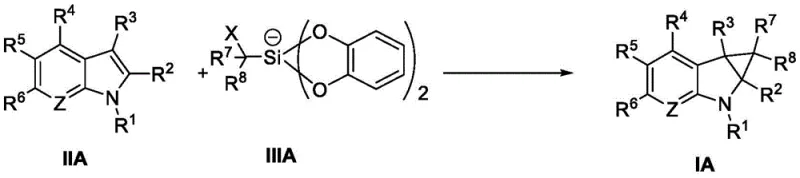
Mechanistic Insights into Visible Light Photocatalytic Cyclopropanation
The core of this transformative technology lies in the intricate interplay between the photocatalyst and the silicon reagent under visible light irradiation. Upon absorption of photons from blue or white LEDs, the photocatalyst enters an excited state, possessing sufficient redox potential to engage in single-electron transfer (SET) processes. In this specific transformation, the excited photocatalyst facilitates the homolytic cleavage of the carbon-iodine bond in the iodomethyl silicate reagent. This generates a reactive carbon-centered radical species along with an iodide anion. The generated radical then adds selectively to the electron-rich double bond of the heterocyclic substrate, such as an indole or pyrrolo[2,3-b]pyridine derivative. This addition step creates a new carbon-carbon bond and a transient radical intermediate on the heterocyclic ring. Subsequently, an intramolecular radical cyclization occurs, closing the three-membered cyclopropane ring and regenerating the aromaticity or stabilizing the system. Finally, the catalytic cycle is closed by the oxidation or reduction of the intermediate, depending on the specific nature of the photocatalyst employed, returning the catalyst to its ground state ready for another turnover. This radical manifold is distinct from ionic pathways and offers unique selectivity patterns that are difficult to achieve with traditional reagents.
From an impurity control perspective, this mechanism offers distinct advantages that are critical for ensuring high-purity pharmaceutical intermediates. Because the reaction proceeds through discrete radical intermediates rather than highly reactive ionic species, there is a reduced tendency for non-specific polymerization or decomposition of sensitive functional groups. The mild conditions prevent thermal degradation of the product, which is a common issue in thermal cyclopropanation reactions. Furthermore, the use of specific photocatalysts like Iridium complexes or organic dyes allows for fine-tuning of the redox potentials, thereby minimizing side reactions such as over-reduction or oxidative degradation of the substrate. The patent data indicates that by optimizing the solvent system, typically a mixture of DMSO and acetonitrile, and the stoichiometry of the reagents, the formation of by-products can be effectively suppressed. This level of control is paramount for meeting the stringent purity specifications required by regulatory bodies for API intermediates, ensuring that the final material is suitable for subsequent coupling reactions without extensive chromatographic purification.
How to Synthesize Heterocyclyl Cyclopropane Efficiently
Implementing this photocatalytic protocol requires attention to detail regarding light source intensity, solvent purity, and reagent addition sequences to maximize efficiency. The general procedure involves dissolving the heterocyclic substrate and the silicon reagent in a degassed solvent mixture under an inert atmosphere to prevent quenching of the excited state by oxygen. A catalytic amount of the photocatalyst is added, and the mixture is irradiated with high-power LEDs. It is often beneficial to add the reagents in portions to maintain a steady concentration of the radical species and prevent dimerization. Detailed standardized synthesis steps for specific derivatives are outlined below to guide process development teams in replicating these results.
- Prepare the reaction mixture by combining the heterocyclic substrate (Formula II) and the silicon reagent (Formula III) in an organic solvent such as DMSO or acetonitrile.
- Add a visible light photocatalyst, such as Ir(dFCF3ppy)2(dtbbpy)PF6 or 4CzIPN, to the solution under an inert atmosphere like argon.
- Irradiate the mixture with blue or white LEDs at room temperature for 24 to 48 hours, monitoring progress via TLC or HPLC until completion.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this photocatalytic technology translates into tangible strategic benefits that extend beyond mere chemical novelty. The primary advantage is the drastic simplification of the manufacturing process, which directly correlates to reduced operational expenditures. By eliminating the need for cryogenic conditions or high-temperature reactors, facilities can utilize standard glass-lined or stainless-steel equipment equipped with LED arrays, which are increasingly becoming standard in modern chemical plants. This reduces the capital expenditure required for specialized infrastructure and lowers the energy costs associated with heating and cooling large reaction volumes. Additionally, the use of commercially available starting materials ensures a stable and reliable supply chain, mitigating the risks associated with sourcing exotic or custom-synthesized reagents that often suffer from long lead times and price volatility.
- Cost Reduction in Manufacturing: The economic impact of this method is profound due to the elimination of expensive and hazardous reagents traditionally used for cyclopropanation. Conventional methods often require diazo compounds or metal carbenoids, which are not only costly but also pose significant safety hazards requiring specialized containment and disposal protocols. By replacing these with a stable silicon reagent and a reusable photocatalyst, the raw material costs are significantly lowered. Furthermore, the mild reaction conditions minimize the formation of degradation products, leading to higher crude yields and reducing the burden on purification units. This efficiency gain means less solvent consumption and lower waste disposal costs, contributing to substantial cost savings in pharmaceutical intermediate manufacturing without compromising on quality or safety standards.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness and scalability of this synthetic route. The reagents involved, such as the iodomethyl silicate and various substituted indoles, are either commodity chemicals or can be synthesized in high volumes using established processes. This availability ensures that production schedules are not disrupted by raw material shortages. Moreover, the reaction's tolerance to various functional groups allows for a convergent synthesis strategy, where complex fragments can be assembled late in the sequence. This flexibility enables manufacturers to respond quickly to changes in demand or to produce multiple analogues from a common intermediate, thereby reducing lead time for high-purity pharmaceutical intermediates and ensuring continuity of supply for downstream API production.
- Scalability and Environmental Compliance: Scaling photochemical reactions has historically been a challenge, but advancements in reactor design and LED technology have made this highly feasible for commercial scale-up of complex pharmaceutical intermediates. The reaction described operates efficiently at room temperature, removing the heat transfer limitations often encountered in exothermic thermal reactions. This makes the transition from gram-scale laboratory experiments to multi-kilogram pilot plant runs much smoother and safer. From an environmental standpoint, the process aligns with green chemistry principles by using visible light as a traceless reagent and generating minimal hazardous waste. The absence of heavy metals in the final product simplifies the purification process and ensures compliance with strict limits on residual metals in drug substances, facilitating faster regulatory approval and market entry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic synthesis method. These insights are derived directly from the experimental data and embodiments disclosed in the patent, providing a clear understanding of the technology's capabilities and limitations for potential partners and licensees.
Q: What are the primary advantages of this photocatalytic method over traditional carbene routes?
A: Unlike traditional methods that require harsh conditions and unstable carbene intermediates, this photocatalytic approach operates under mild room temperature conditions using visible light, significantly improving safety and operational simplicity.
Q: Which photocatalysts are most effective for this transformation?
A: The patent identifies several effective catalysts, with Iridium complexes like Ir(dFCF3ppy)2(dtbbpy)PF6 and organic photocatalysts like 4CzIPN showing superior yields and conversion rates in DMSO/acetonitrile solvent systems.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method utilizes commercially available starting materials, mild reaction conditions, and standard LED light sources, making it highly scalable and environmentally friendly for industrial manufacturing of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Heterocyclyl Cyclopropane Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of photocatalytic technologies in modern drug discovery and development. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN112898202A can be seamlessly translated into robust industrial processes. We are committed to delivering high-purity pharmaceutical intermediates that meet the rigorous quality standards of the global market. Our state-of-the-art facilities are equipped with advanced photochemical reactors and stringent purity specifications are enforced through our rigorous QC labs, guaranteeing that every batch of heterocyclyl cyclopropane compounds we supply is consistent, safe, and ready for the next stage of synthesis.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this cutting-edge synthetic methodology for your pipeline projects. Whether you require custom synthesis of specific analogues or large-scale manufacturing of key intermediates, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis tailored to your specific project needs. We encourage you to reach out to our technical procurement team to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is optimized for efficiency and reliability.
