Scalable Synthesis of Novel 2-Indazole-4-Azaindole Derivatives for Oncology Drug Development
Introduction to Novel CDK7 Inhibitor Scaffolds
The landscape of oncology drug discovery is constantly evolving, driven by the need for more selective and potent kinase inhibitors. A pivotal advancement in this field is documented in Chinese Patent CN101440092B, which discloses a novel class of 2-indazole-4-azaindole-5-amino derivatives. These compounds represent a significant structural innovation over traditional indole-indazole scaffolds, specifically targeting the CDK7 enzyme, a critical regulator of cell cycle progression and transcription. The core innovation lies in the bioisosteric replacement of a carbon atom with a nitrogen atom within the pyrrole ring, creating a pyrrolo[3,2-b]pyridine system that offers superior physicochemical properties.
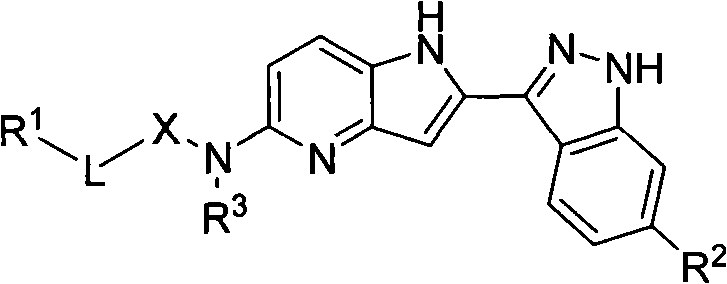
This structural modification is not merely academic; it addresses key pharmacokinetic challenges often faced by early-stage drug candidates, such as poor solubility and rapid metabolic clearance. The patent details a comprehensive library of derivatives where the N5 position is substituted with various amide and sulfonamide groups, allowing for fine-tuning of potency and selectivity. For pharmaceutical companies seeking a reliable pharmaceutical intermediate supplier, understanding the depth of this chemistry is crucial. The disclosed compounds have demonstrated impressive in vitro proliferation inhibitory effects against multiple tumor cell lines, including HL60, KB, and HCT-116, validating their potential as next-generation antitumor agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of CDK7 inhibitors relied heavily on indole-based scaffolds. While effective, these traditional structures often suffer from inherent limitations regarding metabolic stability and off-target toxicity. The indole nucleus, being electron-rich, is susceptible to oxidative metabolism by cytochrome P450 enzymes, leading to short half-lives in vivo. Furthermore, achieving high selectivity against other CDK family members (such as CDK1, 2, 4, and 6) has been a persistent challenge, often resulting in dose-limiting side effects related to bone marrow suppression. Conventional synthetic routes to these indole derivatives frequently involve harsh conditions that limit the diversity of functional groups that can be introduced, thereby restricting the chemical space available for optimization.
The Novel Approach
The methodology presented in CN101440092B overcomes these hurdles through a strategic shift to an azaindole core. By incorporating a nitrogen atom into the five-membered ring, the electronic density of the system is modulated, reducing susceptibility to oxidative degradation. The synthetic strategy employs a convergent approach, building the core pyrrolo[3,2-b]pyridine ring first before introducing the complex indazole moiety via palladium-catalyzed cross-coupling. This modular design allows for the late-stage diversification of the N5 position with various acyl and sulfonyl chlorides. This flexibility is paramount for cost reduction in API manufacturing, as it enables the rapid generation of analogs without redesigning the entire synthetic pathway. The result is a robust platform technology capable of producing high-purity intermediates with consistent quality.
Mechanistic Insights into Stille Coupling and Reductive Cyclization
The synthesis of these sophisticated molecules relies on two critical mechanistic transformations: reductive cyclization and Stille coupling. The formation of the azaindole core begins with the reduction of a nitro-vinyl intermediate. The patent specifies the use of reducing agents such as stannous chloride or palladium on carbon in protic solvents like methanol or ethanol. This step is mechanistically intricate, involving the reduction of the nitro group to an amine, which then spontaneously cyclizes onto the adjacent vinyl ether functionality to close the five-membered ring. Controlling the stoichiometry and temperature during this phase is essential to prevent over-reduction or polymerization, ensuring the integrity of the sensitive heterocyclic system.
Following the construction of the core, the introduction of the indazole fragment is achieved via a Stille coupling reaction between a 2-iodo-azaindole intermediate and an indazole stannane. This palladium-catalyzed cross-coupling is highly chemoselective, tolerating the various protecting groups (such as Boc) present on the molecule. The mechanism involves the oxidative addition of the aryl iodide to the Pd(0) catalyst, followed by transmetallation with the organotin species and reductive elimination to form the new carbon-carbon bond. This step is the linchpin of the synthesis, connecting the two pharmacophores. Understanding this mechanism allows process chemists to optimize catalyst loading and ligand selection, directly impacting the commercial scale-up of complex pharmaceutical intermediates by minimizing residual metal impurities and maximizing yield.
How to Synthesize 2-Indazole-4-Azaindole Derivatives Efficiently
The preparation of these N5-substituted derivatives follows a logical, step-wise progression that balances yield with purity. The process initiates with the alkylation of a nitropyridine precursor, followed by condensation and the aforementioned reductive cyclization to establish the bicyclic core. Subsequent protection, iodination, and coupling steps build molecular complexity. The final deprotection and acylation steps install the diverse side chains responsible for biological activity. For detailed operational parameters, solvent choices, and workup procedures, refer to the standardized guide below.
- Alkylation of 2-(tert-butoxycarbonyl(methyl)amino)-5-nitropyridine followed by condensation with DMF-DMA.
- Reductive ring closure using tin chloride or palladium on carbon to form the pyrrolo[3,2-b]pyridine core.
- Regioselective iodination at the 2-position followed by Stille coupling with indazole stannanes.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the synthetic route outlined in this patent offers distinct advantages over legacy methodologies. The starting materials, such as substituted nitropyridines and indazoles, are commodity chemicals available from multiple global sources, mitigating the risk of single-source dependency. This availability ensures enhanced supply chain reliability, allowing manufacturers to secure raw materials at competitive prices without long lead times. Furthermore, the reliance on well-established reaction types like nucleophilic substitution and palladium coupling means that the process can be executed in standard stainless steel reactors without requiring specialized exotic equipment.
- Cost Reduction in Manufacturing: The synthetic pathway is designed to minimize the number of purification steps required. By utilizing recrystallization as a primary purification method for several intermediates, the process significantly reduces the consumption of silica gel and organic solvents associated with extensive column chromatography. This simplification translates to substantial cost savings in waste disposal and solvent recovery. Additionally, the high atom economy of the coupling reactions ensures that expensive reagents are efficiently incorporated into the final product, reducing the overall cost of goods sold (COGS) for the active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: The modular nature of the synthesis allows for the decoupling of production streams. The azaindole core and the indazole fragment can be manufactured independently and stored as stable intermediates. This strategy provides flexibility in production scheduling, enabling manufacturers to respond rapidly to fluctuations in demand. By maintaining inventory of key building blocks, suppliers can drastically reduce lead times for the final API, ensuring uninterrupted supply for clinical trials and commercial launch phases.
- Scalability and Environmental Compliance: The process avoids the use of highly toxic reagents wherever possible, favoring safer alternatives for reduction and coupling. The waste streams generated are primarily aqueous and organic solvents that can be treated using standard effluent treatment protocols. This alignment with green chemistry principles facilitates regulatory approval and minimizes environmental compliance costs. The robustness of the reaction conditions, particularly the tolerance to moisture and oxygen in later stages, supports seamless technology transfer from laboratory to pilot plant and finally to multi-ton commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these novel kinase inhibitors. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity for stakeholders evaluating this technology for their pipeline.
Q: What is the primary biological target of these 2-indazole-4-azaindole derivatives?
A: These compounds are designed as potent inhibitors of CDK7 (Cyclin-Dependent Kinase 7), a key regulator of the cell cycle and transcription, showing significant activity against tumor cell lines like HL60 and KB.
Q: How does the azaindole scaffold improve upon traditional indole-based inhibitors?
A: By replacing a carbon atom with nitrogen (bioisosteric replacement), the azaindole scaffold offers improved metabolic stability, better solubility profiles, and enhanced binding affinity within the ATP pocket of the kinase.
Q: Is the synthetic route described in CN101440092B suitable for large-scale production?
A: Yes, the route utilizes standard unit operations such as recrystallization and column chromatography with commercially available reagents, making it adaptable for kilogram-to-ton scale manufacturing with proper process optimization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Indazole-4-Azaindole Supplier
As the demand for targeted oncology therapies grows, the ability to produce complex heterocyclic intermediates at scale becomes a critical differentiator. NINGBO INNO PHARMCHEM stands ready to support your drug development programs with our expertise in process chemistry and manufacturing. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from preclinical to clinical supply is seamless. Our facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 2-indazole-4-azaindole derivative meets the highest industry standards.
We invite you to collaborate with us to optimize this synthetic route for your specific needs. Our technical team can provide a Customized Cost-Saving Analysis to identify further efficiencies in the manufacturing process. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us be your partner in bringing these promising antitumor agents from the laboratory to the patients who need them most.
