Advanced Photocatalytic Synthesis of Fluoroalkyl-Substituted Benzofuran and Indole Derivatives for Commercial Scale
The pharmaceutical and agrochemical industries are constantly seeking efficient pathways to incorporate fluorine atoms into complex molecular scaffolds, driven by the unique ability of fluoroalkyl groups to enhance metabolic stability and bioactivity. Patent CN111072605B presents a groundbreaking advancement in this domain by disclosing a highly efficient preparation method for fluoroalkyl-substituted benzofuran derivatives and indole derivatives. This technology leverages visible light photocatalysis to achieve oxygen or amino fluoroalkyl cyclization of olefins in a single step, marking a significant departure from traditional multi-step syntheses. By utilizing readily available 2-allyl phenolic or 2-allyl-aniline compounds as starting materials, the process eliminates the need for pre-functionalization, thereby streamlining the synthetic route. The invention highlights the use of specific photocatalysts and mild reaction conditions to drive these transformations, offering a green and sustainable alternative that holds immense promise for the commercial production of high-value heterocyclic intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of fluoroalkyl groups into heterocyclic systems has been fraught with synthetic challenges that hinder efficient large-scale manufacturing. Conventional strategies often rely on the pre-synthesis of functionalized heterocycles or the use of harsh reagents such as fluoroalkyl sulfonyl chlorides, which can generate significant amounts of hazardous waste. Furthermore, many established protocols necessitate the use of expensive transition metal catalysts, particularly palladium complexes, which not only inflate raw material costs but also introduce complexities regarding residual metal removal—a critical quality attribute for pharmaceutical intermediates. Traditional thermal cyclization methods frequently require elevated temperatures and high-pressure reactors to overcome activation energy barriers, leading to increased energy consumption and safety risks associated with handling volatile fluorinated reagents under extreme conditions. These factors collectively contribute to a cumbersome supply chain and higher overall production costs, limiting the accessibility of these valuable fluorinated building blocks for drug discovery programs.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent utilizes a visible light-mediated radical cyclization strategy that operates under exceptionally mild conditions. The core of this innovation lies in the direct fluoroalkyl difunctionalization of olefins, where the carbon-carbon double bond of the allyl side chain serves as the reactive handle for cyclization. This method employs accessible photocatalysts, such as iridium complexes or organic salts, which absorb visible light to initiate a single-electron transfer process. 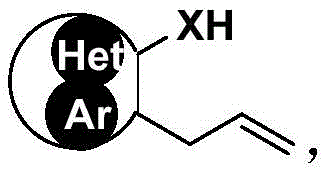 As illustrated in the general reaction scheme, the process seamlessly integrates the fluoroalkyl group and the heteroatom (oxygen or nitrogen) into the final cyclic structure in a single pot. The reaction proceeds at temperatures around 20 +/- 10 °C under nitrogen atmosphere, utilizing standard LED light sources with wavelengths between 380-500 nm. This eliminates the need for specialized high-pressure equipment, drastically simplifying the operational requirements and enhancing the safety profile of the manufacturing process.
As illustrated in the general reaction scheme, the process seamlessly integrates the fluoroalkyl group and the heteroatom (oxygen or nitrogen) into the final cyclic structure in a single pot. The reaction proceeds at temperatures around 20 +/- 10 °C under nitrogen atmosphere, utilizing standard LED light sources with wavelengths between 380-500 nm. This eliminates the need for specialized high-pressure equipment, drastically simplifying the operational requirements and enhancing the safety profile of the manufacturing process.
Mechanistic Insights into Visible Light Photocatalytic Cyclization
The mechanistic elegance of this transformation relies on the photoredox catalytic cycle initiated by the excitation of the photocatalyst upon irradiation. When the photocatalyst, such as the iridium complex shown in 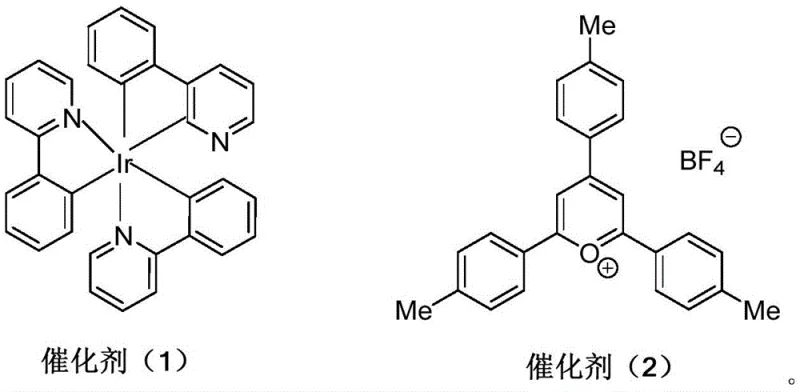 or the organic salt depicted in
or the organic salt depicted in  , absorbs a photon, it transitions to an excited state capable of engaging in single-electron transfer (SET) with the fluoroalkyl iodide reagent. This interaction generates a highly reactive fluoroalkyl radical and regenerates the ground state of the catalyst or enters a subsequent redox cycle. The generated radical adds regioselectively to the terminal alkene of the 2-allyl substrate, forming a new carbon-carbon bond and a transient carbon-centered radical intermediate. This intermediate then undergoes intramolecular cyclization by attacking the proximal heteroatom (phenolic oxygen or aniline nitrogen), followed by oxidation and deprotonation to yield the final aromatic or partially aromatic heterocyclic product. The precise control over the redox potentials of the catalysts ensures high efficiency and minimizes side reactions, which is crucial for maintaining high purity in the final product.
, absorbs a photon, it transitions to an excited state capable of engaging in single-electron transfer (SET) with the fluoroalkyl iodide reagent. This interaction generates a highly reactive fluoroalkyl radical and regenerates the ground state of the catalyst or enters a subsequent redox cycle. The generated radical adds regioselectively to the terminal alkene of the 2-allyl substrate, forming a new carbon-carbon bond and a transient carbon-centered radical intermediate. This intermediate then undergoes intramolecular cyclization by attacking the proximal heteroatom (phenolic oxygen or aniline nitrogen), followed by oxidation and deprotonation to yield the final aromatic or partially aromatic heterocyclic product. The precise control over the redox potentials of the catalysts ensures high efficiency and minimizes side reactions, which is crucial for maintaining high purity in the final product.
From an impurity control perspective, the mild nature of the photocatalytic conditions plays a pivotal role in suppressing the formation of degradation products often seen in thermal processes. The specificity of the radical addition to the unactivated olefin, guided by the electronic properties of the substrate and the catalyst, ensures excellent regioselectivity. Furthermore, the use of bases such as potassium acetate, triethylamine, or DBU helps to neutralize the hydroiodic acid byproduct formed during the reaction, preventing acid-catalyzed decomposition of sensitive functional groups on the aromatic ring. The broad substrate scope demonstrated in the patent, accommodating electron-withdrawing groups like aldehydes and electron-donating groups like methyl and methoxy, indicates a robust mechanism that tolerates diverse electronic environments. This tolerance is essential for pharmaceutical applications where late-stage functionalization is often required, allowing chemists to introduce fluorine motifs into complex molecules without compromising the integrity of other sensitive moieties.
How to Synthesize Fluoroalkyl-Substituted Benzofuran Efficiently
The practical implementation of this synthesis route is designed to be straightforward and adaptable to standard laboratory and pilot plant setups. The procedure typically involves charging a reaction vessel, such as a Schlenk tube, with the 2-allyl substrate, the chosen fluoroalkyl iodide, a suitable base, and a catalytic amount of the photocatalyst in an organic solvent like acetonitrile or dichloromethane. The mixture is then degassed and placed under a nitrogen atmosphere to prevent quenching of the excited catalyst by oxygen. Irradiation with a blue LED lamp drives the reaction to completion over a period of 8 to 24 hours, depending on the specific substrate and reagent combination. Following the reaction, standard workup procedures involving extraction with ethyl acetate and purification via silica gel column chromatography yield the target fluoroalkyl-substituted heterocycles in high yields, often exceeding 80%. For a detailed breakdown of the standardized synthesis steps and specific molar ratios, please refer to the guide below.
- Prepare the reaction mixture by combining 2-allyl phenol or 2-allyl-aniline compounds with a base, fluoroalkyl reagent, solvent, and photocatalyst in a Schlenk tube.
- Stir the solution under a nitrogen atmosphere while illuminating with a blue LED light source (380-500 nm) at a temperature of 20 +/- 10 °C for 8-24 hours.
- Upon completion, extract the reaction mixture with ethyl acetate, remove the solvent under reduced pressure, and purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this photocatalytic technology offers transformative benefits that directly impact the bottom line and operational resilience. The shift away from precious metal catalysts like palladium to more abundant and recyclable photocatalysts represents a significant opportunity for cost reduction in pharmaceutical intermediates manufacturing. By eliminating the need for expensive heavy metals, companies can avoid the costly and time-consuming steps associated with metal scavenging and validation of residual metal levels, which are strictly regulated in API production. Additionally, the use of simple LED lighting systems instead of high-energy thermal heating or high-pressure reactors leads to substantial energy savings and reduces the capital expenditure required for specialized reactor infrastructure. The mild reaction conditions also extend the lifespan of equipment and reduce maintenance downtime, further contributing to overall operational efficiency and cost effectiveness.
- Cost Reduction in Manufacturing: The economic advantages of this process are multifaceted, stemming primarily from the simplification of the synthetic route and the reduction in raw material costs. By enabling a one-step cyclization from readily available allyl phenols and anilines, the method removes the need for multiple synthetic steps and protecting group manipulations that characterize conventional routes. The avoidance of palladium catalysts not only lowers the direct cost of reagents but also mitigates the volatility associated with the pricing of precious metals in the global market. Furthermore, the high atom economy of the reaction, where the fluoroalkyl group and the cyclization occur simultaneously, minimizes waste generation and reduces the costs associated with waste disposal and environmental compliance. These factors combine to create a leaner, more cost-effective manufacturing process that enhances profit margins for high-purity fluoroalkyl-substituted benzofuran derivatives.
- Enhanced Supply Chain Reliability: Supply chain stability is significantly bolstered by the use of commodity chemicals and robust reaction conditions that are less susceptible to disruption. The starting materials, such as 2-allylphenol and various fluoroalkyl iodides, are commercially available from multiple suppliers, reducing the risk of single-source dependency. The mild operating conditions mean that production is not constrained by the availability of specialized high-pressure vessels or extreme temperature control systems, allowing for greater flexibility in manufacturing site selection and capacity expansion. Moreover, the scalability of the photochemical process from gram to kilogram scales ensures that supply can be ramped up quickly to meet fluctuating market demands without the long lead times typically associated with installing complex chemical processing units. This reliability is crucial for maintaining continuous production schedules for critical drug intermediates.
- Scalability and Environmental Compliance: The environmental footprint of this synthesis is markedly lower than traditional methods, aligning with the increasing regulatory pressure for greener chemical manufacturing. The reaction operates at near-ambient temperatures and pressures, inherently reducing the risk of thermal runaways and improving workplace safety. The use of visible light as the energy source is a hallmark of green chemistry, replacing fossil-fuel-derived thermal energy with renewable electricity. Additionally, the high selectivity of the reaction reduces the formation of byproducts, simplifying downstream purification and minimizing solvent usage. The ability to scale this process using flow chemistry techniques or large batch reactors with efficient light penetration further enhances its industrial viability. This combination of safety, sustainability, and scalability makes the technology an attractive option for companies aiming to reduce their carbon footprint while maintaining high production volumes of complex fluorinated intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic synthesis method. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these details is essential for R&D teams evaluating the feasibility of adopting this technology for their specific pipeline projects and for procurement teams assessing the long-term viability of the supply source.
Q: What are the advantages of using visible light photocatalysis over traditional thermal methods for fluoroalkylation?
A: Visible light photocatalysis allows for reactions to proceed under extremely mild conditions (room temperature, atmospheric pressure) compared to traditional methods that often require high temperatures, high pressures, or expensive transition metal catalysts like palladium. This results in better functional group tolerance and significantly reduced energy consumption.
Q: Which fluoroalkyl reagents are compatible with this synthesis method?
A: The method demonstrates broad compatibility with various fluoroalkyl iodides, including ethyl difluoroiodoacetate, perfluoroiodopropane, perfluoroiodobutane, and longer chain perfluoroalkyl iodides such as perfluorohexyliodoalkane and perfluorooctyliodoalkane.
Q: Is this process scalable for industrial production of pharmaceutical intermediates?
A: Yes, the process is highly scalable because it utilizes standard laboratory equipment like Schlenk tubes or conventional reactors without the need for specialized high-pressure autoclaves. The use of simple LED light sources and mild temperatures makes the safety profile favorable for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluoroalkyl-Substituted Benzofuran Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of advanced fluorination technologies in modern drug development and are fully equipped to leverage this patent-protected methodology for our clients. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of fluoroalkyl-substituted benzofuran or indole derivative meets the highest international standards for pharmaceutical intermediates. Our commitment to quality and consistency makes us a trusted partner for global pharmaceutical companies seeking reliable sources of complex fluorinated building blocks.
We invite you to collaborate with us to explore how this innovative photocatalytic process can optimize your supply chain and reduce costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project requirements, demonstrating the tangible economic benefits of switching to this greener synthesis route. Please contact us today to request specific COA data for our existing inventory or to discuss route feasibility assessments for your custom synthesis needs. Let us help you accelerate your drug development timeline with our cutting-edge manufacturing capabilities and unwavering dedication to excellence.
