Advanced Silver-Mediated Cyclization For Commercial Scale-Up Of Complex Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex nitrogen-containing heterocycles, which serve as critical scaffolds in a vast array of bioactive molecules. Patent CN115197144A introduces a groundbreaking approach for preparing nitro-containing 7-membered nitrogen compounds, specifically benzo[b]azepine derivatives, through a metal-catalyzed nitro radical-induced addition cyclization process. This technology addresses a significant gap in organic synthesis, as the efficient construction of 7-membered N-heterocycles via nitro radical pathways has historically been underreported despite their prevalence in top-selling drug molecules. By leveraging a silver-mediated system, this method enables intermolecular regioselective addition of NO2 radicals to o-alkenylacrylamide substrates, followed by a precise 7-exo-dig cyclization. For R&D directors and procurement specialists, this represents a pivotal advancement in accessing high-purity pharmaceutical intermediates with enhanced structural diversity and synthetic efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional strategies for synthesizing 7-membered nitrogen heterocycles often suffer from severe limitations that hinder their application in commercial manufacturing. Conventional routes frequently rely on harsh reaction conditions, expensive transition metal catalysts, or multi-step sequences that result in poor overall atom economy and low yields. Furthermore, the introduction of nitro groups into these sensitive frameworks typically requires hazardous reagents that pose significant safety risks, such as explosion hazards during scale-up. The lack of regioselectivity in many existing methods leads to complex mixtures of isomers, necessitating costly and time-consuming purification processes that erode profit margins. Additionally, the removal of residual heavy metals from the final product to meet stringent pharmaceutical purity specifications often requires additional chelating agents and processing steps, further complicating the supply chain and increasing the environmental footprint of the manufacturing process.
The Novel Approach
In stark contrast, the novel methodology described in patent CN115197144A utilizes a controllable silver-catalyzed system that generates nitro radicals in situ from safe and commercially available precursors like AgNO3 and K2S2O8. This approach facilitates a direct and efficient transformation of o-alkenylacrylamides into valuable 7-nitro-containing 7-membered nitrogen compounds under relatively mild thermal conditions. The reaction proceeds through a well-defined mechanism involving single-electron oxidation and deprotonation, ensuring high selectivity and minimizing the formation of unwanted by-products. 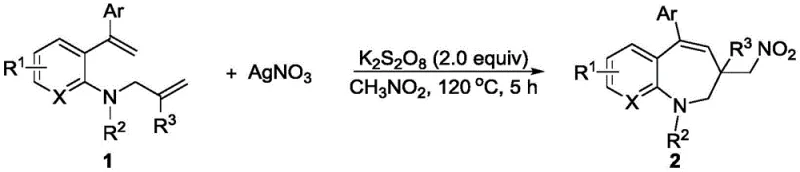 This streamlined process not only simplifies the operational workflow but also significantly enhances the safety profile of the synthesis by eliminating the need for unstable nitro sources. The broad substrate tolerance allows for the incorporation of various functional groups, including halogens and alkyl substituents, making it a versatile platform for generating diverse libraries of intermediates for drug discovery and development programs.
This streamlined process not only simplifies the operational workflow but also significantly enhances the safety profile of the synthesis by eliminating the need for unstable nitro sources. The broad substrate tolerance allows for the incorporation of various functional groups, including halogens and alkyl substituents, making it a versatile platform for generating diverse libraries of intermediates for drug discovery and development programs.
Mechanistic Insights into AgNO3-K2S2O8 Catalyzed Cyclization
The core of this technological breakthrough lies in the intricate radical mechanism that drives the formation of the 7-membered ring structure. The process initiates with the oxidation of silver nitrate by potassium persulfate, which generates the reactive NO2 radical species essential for the transformation. This radical then undergoes intermolecular regioselective addition to the double bond of the o-alkenylacrylamide substrate, forming a key carbon-centered radical intermediate. Unlike competing 5-exo cyclization pathways which are often kinetically favored in similar systems, this method successfully directs the reaction through a 7-exo-dig cyclization process. 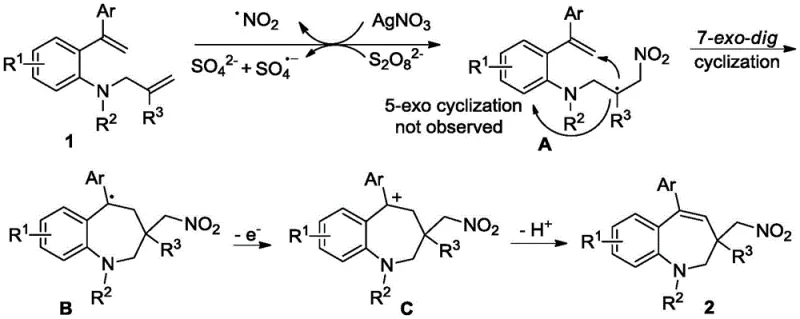 This selectivity is crucial for accessing the thermodynamically less favorable 7-membered ring architecture without forming smaller, undesired cyclic by-products. Following cyclization, the intermediate undergoes a single-electron oxidation and subsequent deprotonation to restore aromaticity and finalize the heterocyclic core. Understanding this mechanistic pathway is vital for process chemists aiming to optimize reaction parameters and ensure consistent quality in large-scale production runs.
This selectivity is crucial for accessing the thermodynamically less favorable 7-membered ring architecture without forming smaller, undesired cyclic by-products. Following cyclization, the intermediate undergoes a single-electron oxidation and subsequent deprotonation to restore aromaticity and finalize the heterocyclic core. Understanding this mechanistic pathway is vital for process chemists aiming to optimize reaction parameters and ensure consistent quality in large-scale production runs.
Furthermore, the mechanism inherently supports superior impurity control, a critical factor for meeting the rigorous standards of the pharmaceutical industry. The use of stoichiometric oxidants and silver salts ensures that the radical generation is controlled and sustained throughout the reaction duration, preventing runaway exotherms that could lead to decomposition or safety incidents. The resulting inorganic salts, such as silver sulfate and potassium bisulfate, are water-soluble and can be easily removed during the aqueous workup phase, leaving the organic product free from metal contamination. This clean reaction profile reduces the burden on downstream purification units and minimizes the generation of hazardous waste streams. For supply chain managers, this translates to a more reliable and compliant manufacturing process that aligns with modern green chemistry principles and regulatory requirements for residual solvents and metals in active pharmaceutical ingredients.
How to Synthesize Nitro-Containing 7-Membered Nitrogen Compounds Efficiently
To implement this synthesis effectively, operators must adhere to precise procedural guidelines that maximize yield and safety while maintaining reproducibility across different batch sizes. The standard protocol involves charging a reaction vessel with the o-alkenylacrylamide substrate, silver nitrate, and potassium persulfate in a nitromethane solvent system. Detailed standardized synthesis steps are provided in the guide below to ensure technical teams can replicate the patented success reliably.
- Prepare the reaction mixture by combining o-alkenylacrylamide substrate, AgNO3 catalyst, and K2S2O8 oxidant in nitromethane solvent.
- Heat the mixture to 120°C in an oil bath to initiate the nitro radical generation and subsequent 7-exo-dig cyclization process.
- Quench the reaction with saturated sodium bicarbonate, extract with dichloromethane, and purify the residue via flash column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented technology offers substantial strategic benefits for procurement managers and supply chain heads looking to optimize their sourcing strategies for complex heterocyclic intermediates. The elimination of expensive and specialized catalysts in favor of commodity chemicals like silver nitrate and potassium persulfate drastically simplifies the raw material supply chain. This shift reduces dependency on niche suppliers and mitigates the risk of procurement bottlenecks that can delay production schedules. Moreover, the simplified workup procedure, which relies on standard extraction and chromatography techniques, lowers the operational complexity and equipment requirements for manufacturing partners. 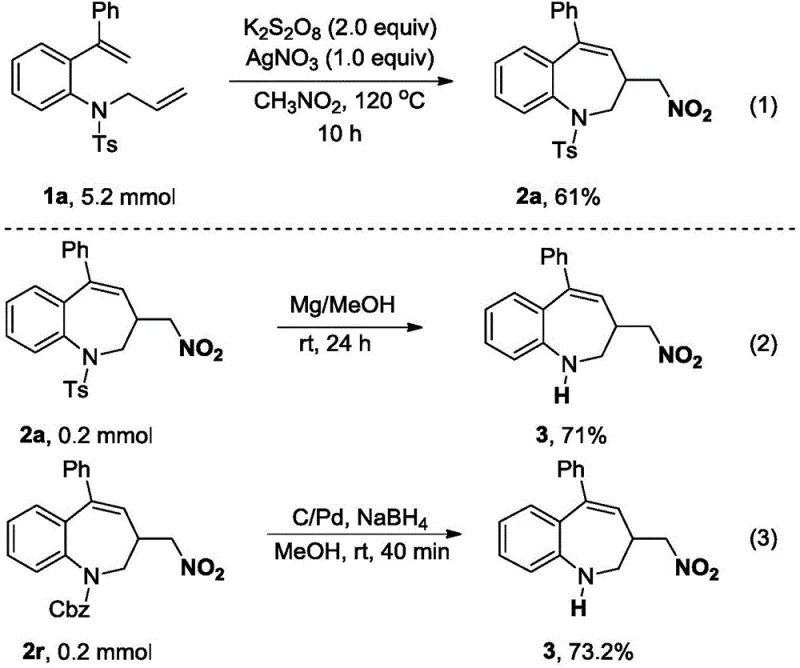 These factors collectively contribute to a more resilient supply chain capable of responding quickly to fluctuating market demands without compromising on product quality or delivery timelines.
These factors collectively contribute to a more resilient supply chain capable of responding quickly to fluctuating market demands without compromising on product quality or delivery timelines.
- Cost Reduction in Manufacturing: The economic advantages of this method are derived primarily from the use of inexpensive, readily available reagents and the reduction of processing steps. By avoiding the need for costly ligand systems or precious metal catalysts that require extensive recovery processes, the overall cost of goods sold is significantly lowered. The high efficiency of the reaction minimizes raw material waste, further enhancing the cost-effectiveness of the process. Additionally, the ability to perform the reaction in a single pot without intermediate isolation steps reduces labor costs and energy consumption associated with multiple unit operations. These cumulative savings allow for more competitive pricing structures in the global marketplace while maintaining healthy profit margins for manufacturers.
- Enhanced Supply Chain Reliability: The robustness of the chemical process ensures consistent output quality, which is paramount for maintaining trust with downstream pharmaceutical clients. The wide substrate scope demonstrated in the patent indicates that the method is tolerant to various electronic and steric environments, meaning it can be applied to a diverse range of target molecules without extensive re-optimization. This flexibility allows suppliers to pivot quickly between different product lines based on customer needs. Furthermore, the stability of the reagents ensures long shelf-life and ease of storage, reducing the logistical challenges associated with handling sensitive or hazardous materials. This reliability translates into shorter lead times and more predictable delivery schedules for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to commercial production often introduces unforeseen challenges, but this methodology is explicitly designed with scalability in mind. The use of standard heating methods and common solvents facilitates a smooth transition from gram-scale experiments to kilogram or ton-scale manufacturing. The absence of explosive reagents significantly lowers the safety barriers for scale-up, allowing for larger batch sizes in standard reactor vessels. From an environmental standpoint, the generation of benign inorganic by-products simplifies waste treatment and disposal, helping companies meet increasingly strict environmental regulations. This compliance reduces the risk of regulatory fines and shutdowns, ensuring continuous operation and long-term business sustainability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation to provide accurate guidance for potential partners. Understanding these details is essential for evaluating the feasibility of integrating this method into existing production workflows.
Q: What are the primary advantages of this silver-mediated cyclization method?
A: This method offers economy, simple raw material preparation, wide substrate scope, and ease of scale-up. It avoids explosive risks associated with traditional nitro radical sources and simplifies post-processing by using easily removable inorganic salts.
Q: Can the resulting 7-membered nitrogen heterocycles be further derivatized?
A: Yes, the products contain removable protecting groups such as N-Ts and N-Cbz. These can be selectively deprotected under mild conditions using magnesium in methanol or palladium-catalyzed reduction, allowing for the synthesis of diverse bioactive molecules.
Q: Is this process suitable for large-scale commercial production?
A: The patent explicitly highlights the scalability of the method. The use of stable reagents like AgNO3 and K2S2O8, combined with standard heating and workup procedures, makes it highly adaptable for industrial manufacturing environments ranging from pilot to commercial scale.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nitro-Containing 7-Membered Nitrogen Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing advanced synthetic technologies to drive innovation in the pharmaceutical sector. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries like patent CN115197144A can be successfully translated into industrial reality. We are committed to delivering products with stringent purity specifications and supporting our clients with rigorous QC labs that verify every batch against the highest international standards. Our infrastructure is designed to handle complex chemistries safely and efficiently, providing a secure foundation for your supply chain needs.
We invite you to collaborate with us to leverage this cutting-edge synthesis method for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can enhance your product portfolio and reduce your time to market. Let us be your partner in turning complex chemical challenges into commercial successes.
