Advanced Copper-Catalyzed Synthesis of 5-Sulfonylpentane-2,3-dienenitrile Intermediates for Scalable Pharma Production
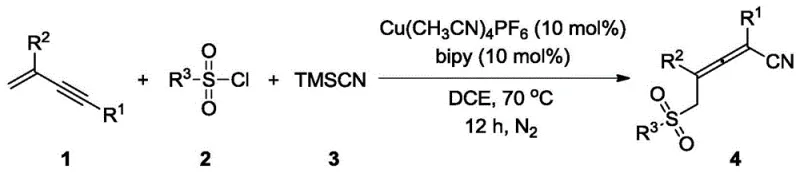
The landscape of organic synthesis for complex pharmaceutical intermediates is constantly evolving, driven by the need for more efficient, atom-economical, and cost-effective pathways. A significant breakthrough in this domain is detailed in patent CN114751800A, which discloses a novel method for synthesizing 5-sulfonylpentane-2,3-dienenitrile compounds. These molecules are critical building blocks in medicinal chemistry, possessing a unique allene core flanked by versatile sulfonyl and cyano functional groups. The patent introduces a transformative three-component coupling reaction that merges conjugated enynes, sulfonyl chlorides, and trimethylsilyl cyanide (TMSCN) in a single operational step. This approach leverages a copper-based catalytic system, specifically utilizing Cu(MeCN)₄PF₆ and 2,2'-bipyridine, to achieve high regioselectivity. For R&D directors and procurement specialists seeking reliable pharmaceutical intermediate suppliers, this technology represents a paradigm shift from laborious multi-step syntheses to streamlined, one-pot processes that enhance both purity profiles and supply chain resilience.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of polysubstituted allene frameworks has been a formidable challenge in organic synthesis, often plagued by inefficient methodologies that hinder large-scale production. Traditional routes typically rely on the functionalization of propargylic substrates through substitution, addition, or elimination reactions, which frequently suffer from poor atom economy and harsh reaction conditions. A major bottleneck in these conventional strategies is the difficulty in controlling regioselectivity during the introduction of multiple functional groups; achieving the precise 1,4-difunctionalization pattern required for bioactive allene nitriles often necessitates protecting group strategies or expensive transition metal catalysts like palladium or rhodium. Furthermore, the separate installation of sulfonyl and cyano moieties usually requires distinct synthetic stages, leading to increased waste generation, lower overall yields, and extended lead times. For supply chain heads, these inefficiencies translate into higher costs of goods sold (COGS) and vulnerability to raw material shortages, as each additional step introduces potential points of failure and purification losses.
The Novel Approach
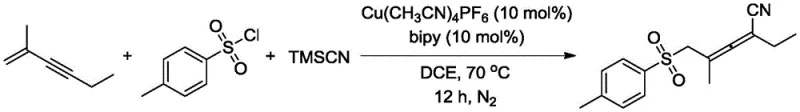
In stark contrast, the methodology outlined in CN114751800A offers a direct and elegant solution by enabling the simultaneous introduction of sulfonyl and cyano groups via a regioselective 1,4-sulfocyanation reaction. This innovative route utilizes inexpensive and readily available starting materials—conjugated enynes, sulfonyl chlorides, and TMSCN—to construct the target 5-sulfonylpentane-2,3-dienenitrile skeleton in a single pot. The reaction proceeds under relatively mild thermal conditions (70°C) in 1,2-dichloroethane (DCE), utilizing a cost-effective copper(I) catalyst system. As demonstrated in the specific example below, the reaction tolerates various substituents, including aromatic and aliphatic groups, delivering the desired products with yields consistently above 55%. This drastic simplification of the synthetic route not only accelerates the timeline for cost reduction in API manufacturing but also minimizes the environmental footprint by reducing solvent usage and waste byproducts associated with intermediate isolations.
Mechanistic Insights into Copper-Catalyzed 1,4-Sulfocyanation
The success of this transformation hinges on the precise interplay between the copper catalyst, the bipyridine ligand, and the radical or ionic species generated in situ. The proposed mechanism involves the activation of the sulfonyl chloride by the copper center, likely generating a sulfonyl radical or a copper-sulfonyl species that adds regioselectively to the terminal position of the conjugated enyne system. This initial addition creates a reactive vinyl-copper intermediate or a stabilized radical species at the propargylic position, which is subsequently trapped by the cyanide source derived from TMSCN. The use of 2,2'-bipyridine as a ligand is critical; it stabilizes the copper oxidation state and modulates the electronic environment to favor the 1,4-addition pathway over competing 1,2-addition or polymerization side reactions. This level of mechanistic control ensures that the resulting allene possesses the correct axial chirality and functional group orientation required for downstream biological activity.
From an impurity control perspective, this catalytic system offers significant advantages over stoichiometric methods. The high regioselectivity minimizes the formation of structural isomers, which are often difficult to separate and can compromise the safety profile of the final drug substance. Furthermore, the mild reaction temperature of 70°C helps prevent the decomposition of sensitive functional groups that might be present on the enyne or sulfonyl chloride substrates. The protocol specifies a nitrogen atmosphere, which is essential to prevent the oxidation of the copper catalyst and the quenching of radical intermediates by atmospheric oxygen. By strictly adhering to these parameters, manufacturers can achieve high-purity allene nitriles with simplified downstream processing, as the crude reaction mixtures are cleaner and more amenable to standard silica gel chromatography or crystallization techniques.
How to Synthesize 5-Sulfonylpentane-2,3-dienenitrile Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reagent quality and atmospheric control to maximize yield and reproducibility. The process begins with the preparation of a sealed reaction vessel equipped with magnetic stirring, into which the sulfonyl chloride, conjugated enyne, and trimethylsilyl cyanide are introduced in a 1:1.5:1.5 molar ratio using 1,2-dichloroethane as the solvent. The catalyst system, consisting of Cu(MeCN)₄PF₆ and 2,2'-bipyridine (each at 10 mol% relative to the enyne), is added sequentially to ensure proper mixing before the vessel is purged with nitrogen gas for approximately one minute to remove dissolved oxygen. Following the purge, the reaction mixture is heated to 70°C and stirred for 12 hours to allow full conversion. Upon completion, the system is cooled to room temperature, and the target molecule is isolated via standard silica gel column chromatography. For detailed standardized operating procedures and safety data sheets regarding this specific protocol, please refer to the technical guide below.
- Prepare the reaction vessel by adding sulfonyl chloride, conjugated enyne, and trimethylsilyl cyanide (TMSCN) in 1,2-dichloroethane (DCE) solvent.
- Add the catalyst system comprising Cu(MeCN)4PF6 (10 mol%) and 2,2'-bipyridine (10 mol%) under a nitrogen atmosphere.
- Heat the mixture to 70°C for 12 hours, then cool and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this copper-catalyzed methodology offers tangible strategic benefits that extend beyond mere technical novelty. The primary advantage lies in the substantial simplification of the supply chain; by consolidating what was previously a multi-step sequence into a single reaction, the number of required raw materials is reduced, and the dependency on complex, custom-synthesized intermediates is eliminated. The starting materials—sulfonyl chlorides and conjugated enynes—are commodity chemicals available from multiple global vendors, ensuring robust supply continuity and mitigating the risk of single-source bottlenecks. Moreover, the use of a base metal catalyst (copper) instead of precious metals like palladium or platinum drastically reduces the raw material cost and eliminates the need for expensive heavy metal scavenging steps during purification, which is a significant driver of cost reduction in pharmaceutical intermediate manufacturing.
- Cost Reduction in Manufacturing: The economic impact of this process is profound due to the elimination of multiple isolation and purification stages. In traditional synthesis, each step incurs costs related to solvent consumption, energy for heating/cooling, labor for operation, and yield loss during workup. By achieving the target structure in one pot with yields exceeding 55%, the overall material throughput is significantly improved. Additionally, the catalyst loading is low (10 mol%), and the ligand (bipyridine) is inexpensive, further driving down the variable cost per kilogram. The removal of precious metal catalysts also reduces the capital expenditure associated with metal recovery systems and lowers the regulatory burden related to residual metal limits in the final API.
- Enhanced Supply Chain Reliability: The robustness of this reaction conditions contributes directly to supply chain stability. The reaction tolerates a wide range of substrates, including those with electron-withdrawing or electron-donating groups, meaning that slight variations in raw material specifications from different suppliers are less likely to cause batch failures. The use of standard solvents like DCE and common reagents like TMSCN ensures that procurement teams can source materials locally or regionally without relying on specialized import channels. This flexibility allows for faster response times to market demand fluctuations and reduces the lead time for high-purity pharmaceutical intermediates, enabling just-in-time manufacturing strategies.
- Scalability and Environmental Compliance: From an EHS (Environment, Health, and Safety) perspective, this process is designed for scalability. The reaction operates at a moderate temperature of 70°C, which is easily manageable in standard glass-lined or stainless steel reactors without requiring cryogenic cooling or high-pressure equipment. The atom economy of the three-component coupling is superior to stepwise approaches, resulting in less chemical waste per unit of product. While DCE is a regulated solvent, its use in a closed loop system with efficient recovery is standard industry practice. The simplicity of the workup (silica gel chromatography) suggests that for larger scales, alternative purification methods like crystallization could be developed, further enhancing the green chemistry profile and facilitating the commercial scale-up of complex intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and scope defined in the patent literature, providing a factual basis for feasibility assessments. Understanding these details is crucial for R&D teams evaluating process transfer and for procurement officers negotiating supply contracts. The answers reflect the current state of the art as described in the intellectual property, highlighting both the capabilities and the specific operational parameters required for success.
Q: What are the key advantages of this copper-catalyzed method over traditional allene synthesis?
A: This method offers a direct one-step synthesis from readily available starting materials, avoiding multi-step sequences. It utilizes a cheap copper catalyst system and achieves high regioselectivity (1,4-addition) with yields exceeding 55%, significantly simplifying the process compared to traditional substitution or elimination methods.
Q: Is this process scalable for industrial production of pharmaceutical intermediates?
A: Yes, the process uses standard organic solvents like 1,2-dichloroethane and operates at moderate temperatures (70°C). The catalyst loading is low (10 mol%), and the substrates (sulfonyl chlorides, enynes) are commercially accessible, making it highly suitable for scale-up from grams to tons.
Q: What is the substrate scope for the sulfonyl chloride component?
A: The method demonstrates broad substrate tolerance. It works effectively with aromatic sulfonyl chlorides (e.g., p-toluenesulfonyl chloride), heteroaromatic variants (e.g., thiophene-2-sulfonyl chloride), and aliphatic sulfonyl chlorides (e.g., propanesulfonyl chloride), allowing for diverse functionalization of the allene core.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Sulfonylpentane-2,3-dienenitrile Supplier
The technological potential of this copper-catalyzed sulfocyanation route is immense, offering a clear pathway to more affordable and accessible allene-based therapeutics. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate this academic innovation into commercial reality. As a seasoned CDMO partner, we have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves seamlessly from gram-scale optimization to tonnage manufacturing. Our facilities are equipped with rigorous QC labs capable of monitoring reaction progress and verifying stringent purity specifications, guaranteeing that every batch of 5-sulfonylpentane-2,3-dienenitrile meets the exacting standards required for global pharmaceutical registration.
We invite you to collaborate with us to leverage this efficient synthesis for your next drug development program. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this one-pot method can improve your margins. Please contact our technical procurement team today to request specific COA data for similar allene intermediates and to discuss route feasibility assessments for your target molecules. Let us help you secure a competitive advantage through superior chemistry and reliable supply.
