Advanced Synthesis of Cefamandole Nafate Derivatives via Silanization Protection for Commercial Scale-up
Introduction to Advanced Cephalosporin Derivative Synthesis
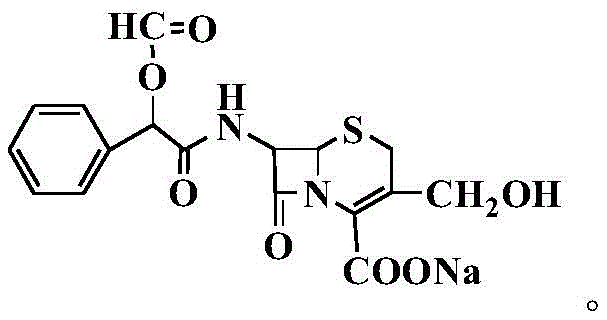
The pharmaceutical landscape for second-generation cephalosporins continues to evolve, driven by the critical need for high-purity intermediates and well-characterized impurity standards. Patent CN110204557B, published in August 2021, introduces a groundbreaking preparation method for cefamandole nafate derivatives that addresses longstanding challenges in synthetic efficiency and product quality. This technology leverages a sophisticated silanization protection strategy followed by precise acylation with D-(-)-O-formylmandelic acid chloride, resulting in derivatives with exceptional structural integrity. For R&D directors and procurement specialists alike, this patent represents a significant leap forward, offering a robust pathway to synthesize complex beta-lactam structures that were previously difficult to isolate in high purity. The ability to generate these derivatives reliably is paramount for ensuring the safety and efficacy of final antibiotic formulations, as well as for facilitating the development of next-generation cephalosporin compounds.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis and isolation of cefamandole nafate derivatives have been plagued by significant technical hurdles that hindered both research progress and commercial viability. Traditional approaches often relied heavily on liquid chromatographic methods or complex purification sequences to separate the desired derivatives from reaction byproducts and unreacted starting materials. These conventional techniques are not only operationally cumbersome and time-consuming but also suffer from inherently low yields, which drastically inflates the cost of goods sold (COGS). Furthermore, the reliance on extensive purification often leads to product degradation, compromising the stability of the sensitive beta-lactam ring. For supply chain managers, these inefficiencies translate into unpredictable lead times and difficulties in securing consistent batches of reference standards or active intermediates, creating bottlenecks in the broader antibiotic manufacturing ecosystem.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent utilizes a streamlined chemical sequence centered on silanization protection. By temporarily masking reactive functional groups on the cephalosporin nucleus using agents such as N,O-Bistrimethylsilylacetamide (BSA) or hexamethyldisilazane, the process prevents unwanted side reactions during the subsequent acylation step. This strategic protection allows for a highly selective reaction with D-(-)-O-formylmandelic acid chloride under mild thermal conditions, typically ranging from -5°C to 25°C. The result is a dramatic improvement in reaction specificity and yield, with experimental data showing purities exceeding 98% without the need for arduous chromatographic separation. This methodological shift not only simplifies the workflow but also establishes a reproducible framework for synthesizing a variety of derivatives, including those with alkyl or alkylhydroxy substituents at the R1 position.
Mechanistic Insights into Silanization-Protection and Acylation
The core of this technological breakthrough lies in the precise orchestration of the silanization and acylation mechanisms. The process initiates with the silylation of the amino and hydroxyl groups on the cephalosporin scaffold, effectively increasing the nucleophilicity of the target sites while protecting sensitive moieties from hydrolysis. This step is typically conducted in polar aprotic solvents like acetonitrile or dichloromethane at temperatures between 10°C and 55°C, ensuring complete conversion of the starting material into the silylated intermediate. Following this protection phase, the introduction of the acyl chloride electrophile proceeds with high regioselectivity. The mild reaction conditions prevent the epimerization of the chiral centers, a common pitfall in beta-lactam chemistry that can render the final product biologically inactive. The subsequent hydrolysis step, performed by adding water at 0°C to 25°C, cleanly removes the silyl protecting groups, regenerating the free functional groups and yielding the final cefamandole nafate derivative in its active form.
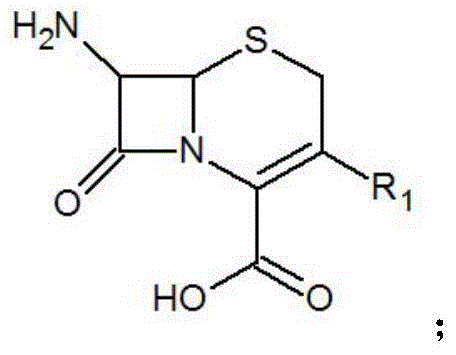
Furthermore, the mechanism includes a robust workup procedure that enhances the overall purity profile of the product. After hydrolysis and phase separation, the organic layer undergoes rigorous washing, dehydration using anhydrous magnesium sulfate, and decoloring with activated carbon. This sequence effectively removes inorganic salts, residual acids, and colored impurities that often persist in crude reaction mixtures. The final crystallization step, often induced by the addition of salt-forming reagents like sodium isooctanoate in ethanol or methanol, ensures that the product precipitates in a highly crystalline form with defined particle size distribution. This level of control over the solid-state properties is crucial for downstream processing, whether the derivative is intended for use as a reference standard in quality control labs or as a key intermediate in the synthesis of new antibiotic candidates.
How to Synthesize Cefamandole Nafate Derivatives Efficiently
Implementing this synthesis route requires careful attention to reaction parameters, particularly temperature control and reagent stoichiometry, to maximize yield and minimize impurity formation. The patent outlines a clear protocol starting with the suspension of the cephalosporin nucleus in a suitable solvent, followed by the gradual addition of the silylating agent. Once the protection is complete, the acyl chloride is added dropwise to manage the exotherm and maintain selectivity. The detailed standardized synthesis steps, including specific molar ratios and workup procedures for different derivatives like the 5-mercapto-1-methyltetrazole variant or the lactone form, are provided in the technical guide below.
- Perform silanization protection on Compound 1 (e.g., D-7ACA or 7-ADCA) using agents like BSA or HMDS/TMCS in solvents such as ethyl acetate or dichloromethane at 10-55°C.
- Conduct acylation reaction by dropwise adding D-(-)-O-formylmandelic acid chloride at controlled low temperatures between -5°C and 25°C.
- Execute hydrolysis by adding water at 0-25°C, followed by layering, washing, dehydration, decoloring, and crystallization to obtain the high-purity derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this silanization-based synthesis route offers profound advantages for procurement managers and supply chain leaders seeking to optimize their antibiotic intermediate portfolios. The elimination of complex chromatographic purification steps translates directly into significant cost reductions in pharmaceutical intermediate manufacturing, as it removes the need for expensive stationary phases and large volumes of specialized solvents. Additionally, the use of widely available and cost-effective reagents such as BSA, hexamethyldisilazane, and common organic solvents like ethyl acetate ensures that the raw material supply chain remains stable and resilient against market fluctuations. This process reliability allows for more accurate forecasting and inventory management, reducing the risk of production stoppages due to material shortages.
- Cost Reduction in Manufacturing: The streamlined nature of this process drastically simplifies the production workflow by removing unit operations associated with low-yield purification methods. By achieving high crude purity through chemical selectivity rather than physical separation, manufacturers can significantly lower energy consumption and solvent waste disposal costs. The ability to recycle solvents like dichloromethane and ethyl acetate further enhances the economic viability of the process, making it an attractive option for large-scale production where margin compression is a constant challenge.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, which tolerate a relatively wide temperature range and utilize stable reagents, ensures consistent batch-to-batch quality. This consistency is vital for maintaining the continuity of supply for critical antibiotics like cefamandole nafate. Furthermore, the versatility of the method allows for the rapid synthesis of various derivatives by simply changing the R1 group on the starting cephalosporin nucleus, enabling suppliers to respond quickly to changing market demands for specific impurity standards or novel analogues without retooling entire production lines.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively from laboratory scale up to pilot batches with yields reaching up to 75%. The mild reaction temperatures and the absence of heavy metal catalysts align well with modern green chemistry principles, reducing the environmental footprint of the manufacturing process. This compliance with stringent environmental regulations facilitates smoother regulatory approvals and minimizes the risk of shutdowns due to non-compliance, thereby securing long-term supply continuity for global pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and technical specifications outlined in patent CN110204557B, providing a clear understanding of the process capabilities and limitations for potential adopters.
Q: What are the key advantages of the silanization protection method described in CN110204557B?
A: The method overcomes prior art limitations by avoiding complex chromatographic separation. It utilizes mild reaction conditions (0-55°C) and common solvents to achieve high yields (up to 75% in examples) and purity exceeding 98%, making it ideal for producing impurity standards and new cephalosporin research materials.
Q: Which starting materials are compatible with this synthesis route?
A: The process is versatile and supports various cephalosporin nuclei. Specifically, the patent demonstrates successful application with D-7ACA (Deacetoxycephalosporanic Acid) and 7-ADCA (Deacetylcephalosporanic Acid) to produce derivatives like the 5-mercapto-1-methyltetrazole derivative and 3-methyl cefamandole nafate.
Q: How does this process impact commercial scalability and cost?
A: By eliminating the need for expensive transition metal catalysts and complex purification steps like liquid chromatography, the process significantly reduces operational costs. The use of standard industrial solvents (ethyl acetate, dichloromethane) and mild temperature profiles ensures the route is readily adaptable for multi-ton commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefamandole Nafate Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development and production of life-saving antibiotics. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated silanization techniques described in this patent can be seamlessly transferred to an industrial setting. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch against the highest international standards.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this advanced technology for their specific needs. By contacting our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your production volume and specific derivative requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific excellence and commercial reliability.
