Advanced Synthesis of Di-tert-butyloxycarbonylamino Oxyacetic Acid for Commercial Scale-Up
The chemical industry is constantly seeking more efficient pathways for synthesizing complex peptide coupling agents, and the recent disclosure in patent CN112375015A offers a compelling solution for the production of di-tert-butyloxycarbonylamino oxyacetic acid. This novel preparation method addresses critical bottlenecks in traditional manufacturing by introducing a streamlined three-step sequence that prioritizes safety, yield, and operational simplicity. Unlike conventional routes that often suffer from lengthy process flows and difficult purification challenges, this technology leverages a unique byproduct conversion strategy to maximize material efficiency. For R&D directors and procurement specialists alike, understanding this mechanism is vital for securing a reliable supply of high-purity pharmaceutical intermediates. The patent outlines a robust protocol that transforms readily available raw materials into a valuable topological template molecule used for connecting peptide chains, signaling a shift towards more sustainable and cost-effective chemical manufacturing practices.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of bis-tert-butoxycarbonylamino-oxyacetic acid has been plagued by significant technical hurdles that hinder large-scale adoption. Traditional methods often involve convoluted reaction sequences that require multiple isolation steps, leading to substantial material loss and increased operational costs. The background art highlights that existing processes are characterized by long process routes and complex operations, which inevitably result in low overall yields that are economically unsustainable for industrial applications. Furthermore, the difficulty in post-treatment operations often necessitates the use of specialized equipment or hazardous reagents, posing safety risks to personnel and complicating regulatory compliance. These inefficiencies create a fragile supply chain where minor deviations in reaction conditions can lead to batch failures, making it challenging for manufacturers to guarantee consistent delivery schedules to downstream pharmaceutical clients.
The Novel Approach
In stark contrast, the methodology described in CN112375015A introduces a paradigm shift by simplifying the synthetic route into three distinct, manageable stages that are highly amenable to scale-up. The core innovation lies in the strategic handling of reaction byproducts; rather than treating them as waste, the process converts them into the desired final product through a targeted acid hydrolysis step. This approach not only drastically improves the overall atom economy but also simplifies the purification workflow, allowing for the use of common solvents like ethanol and dichloromethane. By operating under mild conditions, specifically maintaining temperatures between 15°C and 25°C, the method eliminates the need for energy-intensive heating or cryogenic cooling, thereby reducing the carbon footprint of the manufacturing process. This streamlined architecture ensures that the production of di-tert-butyloxycarbonylamino oxyacetic acid is both technically feasible and commercially viable for meeting global demand.
Mechanistic Insights into the Three-Step Synthesis Strategy
The first stage of the synthesis involves a nucleophilic substitution reaction where tert-butoxycarbonyl-hydroxylamine reacts with bromoacetic acid in the presence of sodium hydroxide. This step is critical as it establishes the foundational amino-oxyacetic acid skeleton with a single Boc protecting group. The addition of sodium hydroxide serves a dual purpose: it neutralizes the acidic byproducts and activates the nucleophile, driving the reaction forward efficiently at ambient temperatures. The choice of ethanol as the primary solvent facilitates the dissolution of reactants while allowing for easy removal via concentration, yielding a solid mixture that can be further purified through aqueous workup and extraction. This careful control of stoichiometry and pH ensures that the mono-protected intermediate is obtained with high purity, setting the stage for the subsequent protection steps without carrying over significant impurities.

Following the initial substitution, the process advances to the second step where the mono-protected intermediate undergoes further Boc-protection using di-tert-butyl dicarbonate (Boc anhydride) catalyzed by DMAP. This reaction is designed to install the second protecting group on the nitrogen atom, but it inherently produces a mixture containing a specific byproduct where the acyloxy position is also protected. The presence of DMAP as a nucleophilic catalyst accelerates the acylation rate, ensuring high conversion within a short timeframe of 3 to 6 hours at room temperature. While the formation of the O-Boc protected byproduct might seem detrimental in other contexts, this specific pathway anticipates its formation, integrating it into the overall yield strategy rather than viewing it as a failure. The reaction mixture thus contains both the desired bis-protected species and the O-protected variant, ready for the final transformation.
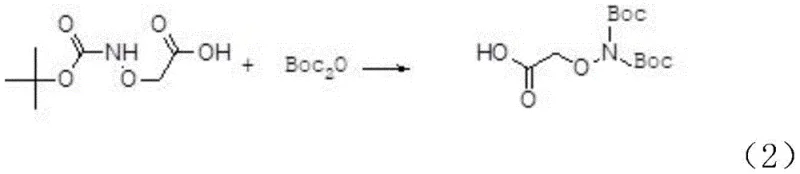
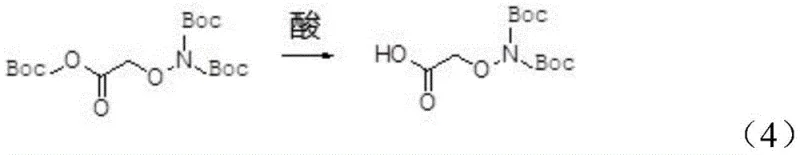
The final and perhaps most ingenious step involves the treatment of the reaction mixture with a dilute acid, specifically 1mol/L hydrochloric acid, to selectively cleave the O-Boc group from the byproduct. This acidolysis converts the unwanted O-protected species back into the target di-tert-butoxycarbonylamino-oxy-acetic acid, effectively recycling what would otherwise be waste into valuable product. The stability of the N-Boc groups under these specific acidic conditions allows for this selective deprotection without compromising the integrity of the final molecule. After the conversion is complete, a simple workup involving phase separation, washing, and drying yields a crude product that is easily purified by recrystallization from petroleum ether. This mechanistic elegance ensures that the final product achieves a purity level exceeding 99%, meeting the stringent requirements for pharmaceutical intermediate applications.
How to Synthesize Di-tert-butyloxycarbonylamino Oxyacetic Acid Efficiently
Implementing this synthesis requires precise adherence to the molar ratios and temperature controls specified in the patent to ensure optimal results. The process begins with the careful addition of reagents in the first step to manage exotherms, followed by a controlled protection phase where catalyst loading is critical for reaction kinetics. The final acid treatment must be monitored to ensure complete conversion of the byproduct without over-exposing the product to acidic conditions that could lead to degradation. Operators should focus on the recrystallization step as the final gatekeeper for purity, utilizing petroleum ether to remove any remaining organic impurities effectively. For a detailed breakdown of the specific operational parameters and safety precautions, please refer to the standardized synthesis guide below.
- React Boc-hydroxylamine with bromoacetic acid and sodium hydroxide in ethanol at 15-25°C to form the mono-protected intermediate.
- Treat the intermediate with di-tert-butyl dicarbonate and DMAP catalyst in dichloromethane to generate the bis-protected mixture.
- Add dilute hydrochloric acid to convert byproducts into the final product, followed by recrystallization from petroleum ether.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this patented method offers substantial advantages by relying on commodity chemicals that are readily available in the global market, mitigating the risk of raw material shortages. The use of common solvents such as ethanol and dichloromethane means that facilities do not require specialized infrastructure or exotic reagents, which significantly lowers the barrier to entry for contract manufacturing organizations. Furthermore, the simplified post-treatment operations reduce the labor hours required per batch, allowing production teams to increase throughput without proportional increases in overhead costs. The ability to convert byproducts into the final product directly impacts the cost of goods sold by maximizing the utility of every kilogram of input material, creating a more resilient economic model for long-term supply contracts.
- Cost Reduction in Manufacturing: The elimination of complex purification sequences and the recycling of byproducts into the main product stream lead to a drastic reduction in material waste and associated disposal costs. By avoiding the need for expensive chromatography or multi-step recrystallizations typically required in older methods, the overall operational expenditure is significantly lowered. The mild reaction conditions also translate to lower energy consumption for heating and cooling, further contributing to a leaner manufacturing budget. This efficiency allows suppliers to offer more competitive pricing structures while maintaining healthy margins, benefiting the entire value chain from manufacturer to end-user.
- Enhanced Supply Chain Reliability: The robustness of this three-step process ensures consistent batch-to-batch quality, which is paramount for maintaining uninterrupted supply lines to pharmaceutical clients. Since the raw materials are stable and widely sourced, the risk of supply disruption due to vendor-specific issues is minimized, providing greater security for long-term procurement planning. The scalability of the reaction, demonstrated from gram to kilogram scales in the patent examples, indicates that production can be ramped up quickly to meet surges in demand without compromising product integrity. This reliability makes the supplier a strategic partner rather than just a vendor, capable of supporting clinical trial phases through to commercial launch.
- Scalability and Environmental Compliance: The process is designed with environmental sustainability in mind, effectively reducing the generation of three wastes (waste water, waste gas, and solid waste) compared to traditional routes. The use of recyclable solvents and the minimization of hazardous byproducts simplify the compliance burden for environmental health and safety teams. As regulatory pressures on chemical manufacturing intensify globally, adopting a cleaner synthesis route future-proofs the supply chain against stricter emission standards. This alignment with green chemistry principles not only reduces environmental impact but also enhances the corporate social responsibility profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis route. Understanding these details helps technical teams assess the feasibility of integrating this method into their existing production workflows. The answers are derived directly from the experimental data and claims presented in the patent documentation, ensuring accuracy and relevance for process chemists.
Q: What catalyst is used in the second step of the synthesis?
A: The process utilizes 4-Dimethylaminopyridine (DMAP) as a highly efficient catalyst in the second step to promote the reaction between the intermediate and di-tert-butyl dicarbonate.
Q: How is the byproduct managed in this preparation method?
A: Instead of discarding the byproduct formed during protection, the method employs an acid treatment step (using dilute HCl) to convert the acyloxy-protected byproduct back into the desired final product, thereby maximizing yield.
Q: What are the optimal reaction conditions for the first step?
A: The first step is conducted at a mild temperature range of 15-25°C using ethanol as the solvent, with a specific molar ratio of reactants to ensure high conversion and safety.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Di-tert-butyloxycarbonylamino Oxyacetic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of novel therapeutics and protein models. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with precision and speed. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of di-tert-butyloxycarbonylamino oxyacetic acid meets the highest industry standards. Our commitment to technical excellence allows us to navigate complex synthesis challenges, delivering products that facilitate your research and manufacturing goals without compromise.
We invite you to collaborate with us to leverage this advanced preparation method for your specific applications. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your project needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can enhance your supply chain efficiency and reduce your overall time to market.
