Scalable Manufacturing of Phenanthroindolizidine Derivatives via Iron-Catalyzed Oxidative Coupling
Introduction to Advanced Phenanthroindolizidine Manufacturing
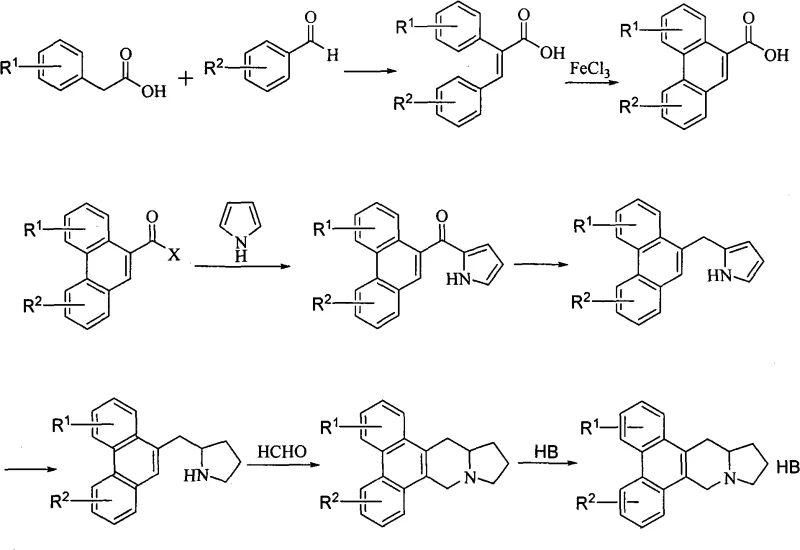
The pharmaceutical industry continuously seeks robust and cost-effective pathways for synthesizing complex alkaloid scaffolds, particularly those exhibiting potent biological activity. Patent CN101348483A introduces a groundbreaking methodology for the preparation of phenanthroindolizidine derivatives, a class of compounds renowned for their potential in oncology and antiviral applications. This technology leverages a highly efficient iron-catalyzed oxidative coupling strategy to construct the critical phenanthrene core, bypassing traditional reliance on precious metal catalysts. By utilizing readily available starting materials such as substituted phenylacetic acids and benzaldehydes, this route offers a streamlined approach to generating diverse libraries of bioactive molecules. The innovation lies not only in the chemical elegance of the FeCl3-mediated cyclization but also in its practical implications for large-scale manufacturing, where cost control and supply chain reliability are paramount. For global procurement teams and R&D directors, understanding this synthetic pathway provides a strategic advantage in sourcing high-purity intermediates for next-generation therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the phenanthrene skeleton, a prerequisite for phenanthroindolizidine synthesis, has often relied on transition metal-catalyzed cross-coupling reactions, such as Suzuki-Miyaura or Heck couplings. While effective on a laboratory scale, these methods present significant challenges when translated to industrial production. The primary bottleneck is the dependence on palladium catalysts, which are not only exorbitantly expensive but also subject to volatile market pricing and supply constraints. Furthermore, the removal of residual palladium from the final active pharmaceutical ingredient (API) requires sophisticated and costly purification steps to meet stringent regulatory limits, typically below 10 ppm. Additionally, traditional routes often involve multiple protection and deprotection steps, increasing the overall step count, reducing atom economy, and generating substantial chemical waste. These factors collectively inflate the cost of goods sold (COGS) and extend lead times, creating friction in the supply chain for critical drug candidates.
The Novel Approach
The methodology disclosed in CN101348483A represents a paradigm shift by employing Ferric Chloride (FeCl3) as the oxidant for the intramolecular cyclization of 2,3-diaryl acrylic acids. This approach fundamentally alters the economic landscape of phenanthroindolizidine manufacturing. Iron is one of the most abundant and inexpensive metals on earth, drastically reducing the raw material cost compared to palladium. The reaction conditions are relatively mild, often proceeding at room temperature or with moderate heating in common solvents like dichloromethane. This simplicity translates directly into operational efficiency, as it eliminates the need for specialized equipment required for high-pressure hydrogenation or inert atmosphere handling often associated with sensitive Pd-catalysts. Moreover, the workup procedure is straightforward, involving simple filtration or aqueous extraction, which facilitates easier isolation of the 9-phenanthrenecarboxylic acid intermediate. This novel route effectively decouples the synthesis of complex alkaloids from the volatility of the precious metal market, ensuring a more stable and predictable supply chain for downstream users.
Mechanistic Insights into FeCl3-Catalyzed Oxidative Cyclization
The core of this technological advancement is the oxidative photocyclization or chemical oxidative cyclization mediated by Lewis acids. In the specific context of this patent, the reaction involves the treatment of 2,3-diaryl acrylic acid derivatives with anhydrous ferric chloride. The mechanism likely proceeds through a single-electron transfer (SET) process where Fe(III) acts as a one-electron oxidant. Initially, the electron-rich aromatic rings of the diaryl acrylic acid coordinate with the Lewis acidic iron center. This interaction facilitates the generation of a radical cation species on the aromatic system. Subsequent intramolecular radical attack on the adjacent aryl ring leads to the formation of a new carbon-carbon bond, closing the central ring of the phenanthrene system. The loss of protons and rearomatization, driven by the oxidizing power of the iron species, yields the fully conjugated phenanthrene carboxylic acid structure. This mechanistic pathway is highly tolerant of various substituents, including methoxy and halogen groups, allowing for significant structural diversity in the final product without compromising yield.
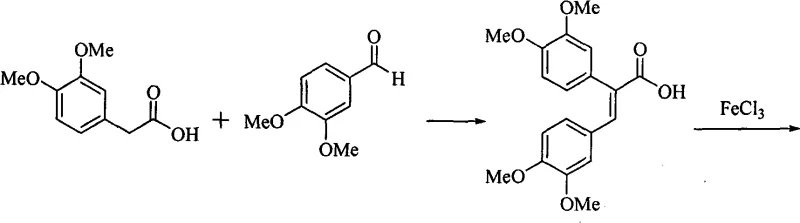
Controlling impurities in this oxidative coupling is critical for pharmaceutical applications. The use of FeCl3 minimizes the formation of homocoupling byproducts often seen in radical reactions because the intramolecular cyclization is kinetically favored over intermolecular processes due to entropy. Furthermore, the subsequent steps in the synthesis, such as the conversion to the acyl chloride using thionyl chloride and the reaction with pyrrole, are well-established transformations with predictable impurity profiles. The reduction steps, utilizing reagents like sodium borohydride or catalytic hydrogenation over Pd-C, are highly selective for the carbonyl and pyrrole functionalities, leaving the sensitive methoxy groups on the phenanthrene core intact. This chemoselectivity ensures that the final phenanthroindolizidine scaffold maintains high purity, reducing the burden on downstream purification processes like preparative HPLC or extensive recrystallization, which are major cost drivers in API manufacturing.
How to Synthesize Phenanthroindolizidine Derivatives Efficiently
The synthesis of these valuable intermediates follows a logical sequence designed for maximum efficiency and yield. The process begins with a Knoevenagel-type condensation between a substituted phenylacetic acid and a benzaldehyde to form the stilbene-like acrylic acid precursor. This is followed by the pivotal FeCl3-mediated cyclization to establish the phenanthrene core. The resulting acid is then activated, typically as an acid chloride, and coupled with pyrrole. Subsequent reduction of the carbonyl group and the pyrrole ring, followed by a final Pictet-Spengler-like cyclization with formaldehyde, constructs the indolizidine moiety fused to the phenanthrene system. Detailed standardized operating procedures for each of these steps, including precise stoichiometry, temperature controls, and safety protocols, are essential for successful technology transfer.
- Condense substituted phenylacetic acid with substituted benzaldehyde to form 2,3-diaryl acrylic acid.
- Perform oxidative coupling using Ferric Chloride (FeCl3) to generate the 9-phenanthrenecarboxylic acid core.
- Convert the acid to an acyl chloride or amide, react with pyrrole, reduce the carbonyl and pyrrole ring, and finally close the indolizidine ring using formaldehyde.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this FeCl3-based synthetic route offers compelling strategic benefits that extend beyond simple chemistry. The primary advantage is the drastic reduction in dependency on critical raw materials that are prone to supply shocks. By substituting palladium with iron, manufacturers can insulate their production costs from the fluctuations of the precious metals market. This stability allows for more accurate long-term budgeting and pricing agreements with pharmaceutical clients. Additionally, the simplified purification workflow reduces the consumption of expensive chromatography media and solvents, further driving down the variable costs associated with production. The robustness of the reaction conditions also implies a lower risk of batch failures, enhancing overall supply reliability and ensuring consistent delivery schedules for downstream partners.
- Cost Reduction in Manufacturing: The elimination of expensive palladium catalysts and the associated ligands results in a significant decrease in direct material costs. Furthermore, the simplified workup procedures reduce the requirement for specialized scavengers used to remove heavy metals, lowering both material and waste disposal expenses. The high yields reported in the patent embodiments, such as the 87% yield for the cyclization step and over 90% for subsequent reductions, indicate a highly efficient process that maximizes output per unit of input, directly improving the margin profile for commercial scale-up of complex pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Iron chloride is a commodity chemical available globally in vast quantities, ensuring that production is never halted due to reagent shortages. This contrasts sharply with specialized catalysts that may have single-source suppliers or long lead times. The use of common solvents like dichloromethane, ethanol, and methanol further simplifies logistics, as these are standard inventory items for most chemical manufacturing facilities. This accessibility translates to reduced lead time for high-purity phenanthroindolizidine derivatives, allowing manufacturers to respond more agilely to market demand spikes or urgent clinical trial material requests.
- Scalability and Environmental Compliance: The reaction conditions described are inherently scalable, moving seamlessly from gram-scale laboratory experiments to multi-kilogram pilot runs without requiring exotic high-pressure or cryogenic equipment. From an environmental perspective, replacing heavy metals with iron aligns with green chemistry principles, potentially simplifying regulatory filings related to elemental impurities (ICH Q3D). The reduced heavy metal load in the waste stream also lowers the cost and complexity of effluent treatment, making the process more sustainable and compliant with increasingly stringent environmental regulations governing chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a clear picture of the process capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What is the primary advantage of using FeCl3 for phenanthrene ring closure?
A: The use of Ferric Chloride (FeCl3) eliminates the need for expensive palladium catalysts typically required for biaryl coupling, significantly reducing raw material costs and simplifying heavy metal removal processes.
Q: Can this synthesis route be scaled for commercial production?
A: Yes, the patent demonstrates robust yields (e.g., 87% for the cyclization step) using standard reagents like thionyl chloride and sodium borohydride, indicating high feasibility for kilogram-to-ton scale manufacturing.
Q: What forms of the final product are available?
A: The process allows for the formation of various salts, including hydrochlorides, hydrobromides, and picrates, enhancing the solubility and stability profiles suitable for different pharmaceutical formulations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phenanthroindolizidine Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis and contract development, possessing the technical expertise to translate complex patent methodologies like CN101348483A into commercial reality. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop to plant floor. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of phenanthroindolizidine intermediate meets the highest quality standards required for pharmaceutical applications. Our commitment to quality assurance ensures that impurity profiles are thoroughly characterized and controlled, mitigating risks during your drug development lifecycle.
We invite you to collaborate with us to leverage this cost-effective synthetic route for your specific project needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can optimize your supply chain and accelerate your time to market. Let us be your partner in delivering high-quality chemical solutions with unmatched reliability and efficiency.
