Advanced Green Catalytic Synthesis of Benzofuran Triarylmethane Compounds for Pharmaceutical Applications
Advanced Green Catalytic Synthesis of Benzofuran Triarylmethane Compounds for Pharmaceutical Applications
The pharmaceutical and material science industries are constantly seeking efficient pathways to construct complex molecular architectures, particularly triarylmethane scaffolds which serve as critical intermediates for dyes, fluorophores, and bioactive agents. Patent CN109651344B introduces a groundbreaking green catalytic synthesis method for benzofuran triarylmethane compounds, addressing the long-standing challenges of traditional cross-coupling reactions. This innovation leverages a copper-catalyzed cross-dehydrogenative coupling (CDC) strategy, utilizing benzofuranone derivatives and aromatic heterocycles as primary feedstocks. By employing cuprous bromide as a catalyst in conjunction with potassium persulfate and potassium dihydrogen phosphate in an acetonitrile medium, this protocol achieves high yields of triaryl quaternary carbon compounds under mild conditions. The significance of this technology lies in its ability to bypass the need for pre-functionalized substrates, thereby streamlining the synthetic route and aligning with modern principles of atom economy and environmental sustainability.
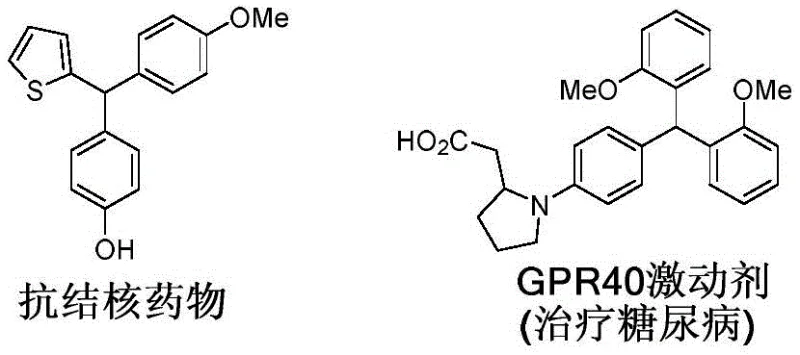
The structural versatility of triarylmethanes is exemplified by their presence in potent pharmaceutical agents, ranging from anti-tuberculosis medications to GPR40 agonists for diabetes treatment, as well as anticancer inhibitors. The ability to access these privileged structures through a direct C-H activation pathway represents a significant leap forward for reliable pharmaceutical intermediate supplier networks aiming to reduce lead times and costs. This report analyzes the technical merits and commercial implications of this novel synthesis route, providing actionable insights for R&D directors, procurement managers, and supply chain heads looking to optimize their manufacturing pipelines for high-value fine chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of triarylmethane skeletons has relied heavily on classical cross-coupling reactions such as Suzuki-Miyaura or Friedel-Crafts alkylations, which often necessitate the use of pre-functionalized starting materials like aryl halides or organometallic reagents. These conventional approaches inherently suffer from several drawbacks, including the generation of stoichiometric amounts of toxic waste, the requirement for expensive palladium or other noble metal catalysts, and the need for rigorous exclusion of moisture and oxygen. Furthermore, the synthesis of quaternary carbon centers via these methods can be sterically hindered, leading to poor conversion rates and difficult purification processes that escalate production costs. The reliance on halogenated precursors also introduces safety hazards and environmental burdens associated with halogen waste disposal, making these routes less attractive for large-scale industrial applications where green chemistry metrics are increasingly scrutinized by regulatory bodies and corporate sustainability goals.
The Novel Approach
In stark contrast, the method disclosed in CN109651344B utilizes a direct cross-dehydrogenative coupling (CDC) reaction that activates inert C-H bonds directly, eliminating the need for pre-functionalization and significantly simplifying the synthetic workflow. By employing a cost-effective copper catalyst system rather than precious metals, this approach drastically reduces raw material expenses while maintaining high catalytic efficiency. The reaction proceeds smoothly in acetonitrile at a moderate temperature of 80°C, utilizing potassium persulfate as a benign oxidant and potassium dihydrogen phosphate as a buffer to maintain optimal reaction pH. This novel pathway not only enhances atom utilization efficiency but also minimizes the formation of hazardous byproducts, offering a cleaner and more sustainable alternative for the preparation of complex triarylmethane derivatives. The operational simplicity of this method, which does not require specialized equipment or stringent inert atmosphere conditions, makes it highly amenable to commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into CuBr-Catalyzed Cross-Dehydrogenative Coupling
The core of this innovative synthesis lies in the copper-catalyzed radical mechanism that facilitates the formation of C-C bonds between the benzofuranone scaffold and various aromatic heterocycles. The catalytic cycle likely initiates with the oxidation of the cuprous bromide species by potassium persulfate to generate a reactive copper(II) or copper(III) intermediate, which subsequently activates the C-H bond at the alpha-position of the benzofuranone. Simultaneously, the aromatic heterocycle undergoes single-electron transfer or hydrogen atom abstraction to form a nucleophilic radical species. The convergence of these activated species leads to the formation of the new C-C bond, constructing the sterically crowded triaryl quaternary carbon center with high regioselectivity. The presence of potassium dihydrogen phosphate plays a crucial role in stabilizing the reaction environment, potentially acting as a proton shuttle to facilitate the deprotonation steps required to restore aromaticity and complete the catalytic turnover. This mechanistic elegance allows for the tolerance of diverse functional groups on both coupling partners, enabling the synthesis of a wide array of derivatives without compromising yield or purity.
Understanding the impurity profile is critical for R&D teams aiming to implement this technology, as the radical nature of the CDC reaction could theoretically lead to homocoupling byproducts or over-oxidation species. However, the specific combination of CuBr and the phosphate buffer appears to suppress these side reactions effectively, directing the selectivity towards the desired cross-coupled product. The reaction conditions are optimized to balance the rate of radical generation with the rate of coupling, ensuring that the concentration of reactive intermediates remains low enough to prevent uncontrolled polymerization or decomposition. For quality control purposes, monitoring the consumption of the oxidant and the formation of the characteristic quaternary carbon signal via NMR or LC-MS is recommended to ensure batch consistency. The robustness of this catalytic system against moisture and air further simplifies the process control requirements, reducing the risk of batch failures due to environmental fluctuations during manufacturing.
How to Synthesize Benzofuran Triarylmethane Efficiently
Implementing this synthesis route requires careful attention to reagent stoichiometry and thermal management to maximize yield and minimize impurity formation. The patent outlines a generalized procedure where benzofuranone derivatives and heterocyclic partners are combined in a specific molar ratio, typically with a slight excess of the benzofuranone component to drive the reaction to completion. The addition of the catalyst and oxidant must be controlled to prevent exothermic spikes, although the reaction temperature of 80°C is relatively mild and easily manageable in standard jacketed reactors. Detailed standardized synthesis steps, including specific workup procedures and purification protocols via column chromatography, are essential for reproducing the high yields reported in the patent examples, which range significantly depending on the electronic and steric properties of the substrates. For a comprehensive guide on executing this transformation in a GMP-compliant environment, please refer to the technical documentation below.
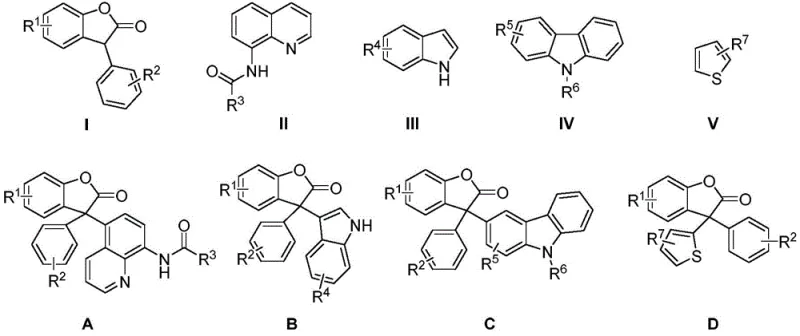
- Combine benzofuranone derivatives and aromatic heterocyclic compounds (quinoline, indole, carbazole, or thiophene) in a reaction vessel.
- Add cuprous bromide catalyst (10 mol%), potassium persulfate oxidant, and potassium dihydrogen phosphate base in acetonitrile solvent.
- Heat the mixture to 80°C for 12 hours, then isolate the triarylmethane product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the shift from precious metal catalysts to earth-abundant copper salts represents a substantial opportunity for cost reduction in pharmaceutical intermediate manufacturing. Copper bromide is significantly cheaper and more readily available than palladium or rhodium complexes, and its lower toxicity profile simplifies waste handling and disposal logistics. The elimination of pre-functionalized starting materials, such as aryl boronic acids or halides, further reduces the bill of materials cost, as these precursors often require multi-step synthesis themselves. Additionally, the use of acetonitrile, a common and recyclable industrial solvent, avoids the need for exotic or highly regulated solvents, streamlining the solvent recovery process and reducing overall operational expenditures. These factors collectively contribute to a more economical production model that can withstand market volatility in raw material pricing.
- Cost Reduction in Manufacturing: The replacement of expensive noble metal catalysts with cuprous bromide eliminates the need for costly metal scavenging steps to meet residual metal specifications in API intermediates. Since the catalyst loading is moderate at 10 mol% and the metal is non-precious, the overall catalyst cost per kilogram of product is drastically lowered compared to traditional palladium-catalyzed routes. Furthermore, the avoidance of pre-functionalized reagents reduces the number of synthetic steps required upstream, compounding the savings in labor, energy, and raw materials. This streamlined approach allows manufacturers to offer high-purity pharmaceutical intermediates at a more competitive price point without sacrificing quality or yield.
- Enhanced Supply Chain Reliability: The reagents utilized in this process, including benzofuranones, simple heterocycles, and inorganic salts, are commodity chemicals with stable and robust global supply chains. Unlike specialized organometallic reagents that may have long lead times or single-source dependencies, the inputs for this CDC reaction are widely available from multiple vendors, mitigating the risk of supply disruptions. The operational simplicity of the reaction, which tolerates ambient atmosphere and does not require cryogenic conditions or high-pressure equipment, also means that production can be easily transferred between different manufacturing sites or scaled up rapidly to meet surging demand. This flexibility ensures a continuous supply of critical intermediates for downstream drug development programs.
- Scalability and Environmental Compliance: The green chemistry attributes of this method, such as high atom economy and the use of less hazardous oxidants, align perfectly with increasingly stringent environmental regulations. The absence of halogenated waste streams and heavy metal residues simplifies the effluent treatment process, reducing the environmental footprint of the manufacturing facility. Scalability is further supported by the homogeneous nature of the reaction mixture and the moderate thermal requirements, which allow for safe operation in large-scale reactors without significant heat transfer limitations. This makes the technology suitable for commercial scale-up of complex pharmaceutical intermediates, enabling producers to transition seamlessly from pilot plant batches to multi-ton annual production capacities while maintaining compliance with ecological standards.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this copper-catalyzed synthesis method. These answers are derived directly from the experimental data and scope defined in the patent literature, providing a reliable foundation for process development teams evaluating this technology for their specific projects. Understanding these nuances is essential for successful technology transfer and risk mitigation during the early stages of route selection.
Q: What catalyst system is used for this triarylmethane synthesis?
A: The process utilizes cuprous bromide (CuBr) as the catalyst at a loading of 10 mol%, paired with potassium persulfate as the oxidant and potassium dihydrogen phosphate as the base.
Q: What are the optimal reaction conditions described in the patent?
A: The reaction is conducted in acetonitrile solvent at a temperature of 80°C for a duration of 12 hours to achieve high yields of the target quaternary carbon compounds.
Q: Which heterocyclic substrates are compatible with this method?
A: The method demonstrates broad substrate scope, successfully coupling benzofuranones with quinolines, indoles, carbazoles, and thiophenes to form diverse triarylmethane scaffolds.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzofuran Triarylmethane Supplier
The technological advancements detailed in patent CN109651344B underscore the potential for more efficient and sustainable production of high-value triarylmethane intermediates. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent quality regardless of order volume. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including residual metal analysis and chiral purity assessments where applicable. We are committed to leveraging such green catalytic technologies to deliver cost-effective solutions that accelerate our partners' drug development timelines while adhering to the highest standards of safety and environmental stewardship.
We invite procurement leaders and R&D directors to engage with our technical procurement team to discuss how this novel synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your target molecule. We encourage you to contact us today to obtain specific COA data for similar intermediates and to schedule a consultation for detailed route feasibility assessments tailored to your project's unique requirements.
