Advanced Rhodium-Catalyzed Synthesis of Functionalized Naphthalene-Sulfoxide Ylide Hybrids for Commercial Scale-Up
Introduction to Novel Naphthalene-Sulfoxide Hybrid Synthesis
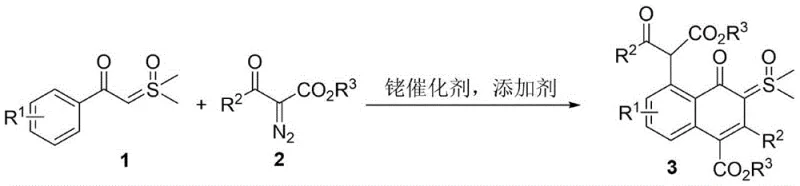
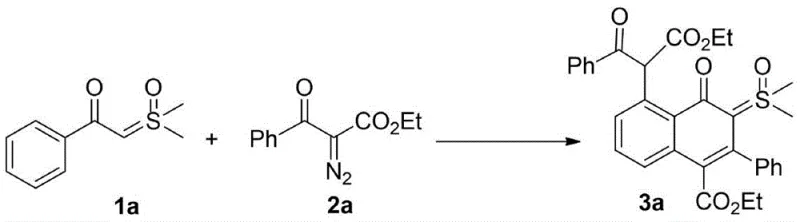
The pharmaceutical and fine chemical industries are constantly seeking efficient pathways to construct complex polycyclic architectures that serve as critical building blocks for drug discovery and material science. A significant breakthrough in this domain is documented in patent CN109810036B, which discloses a sophisticated synthesis method for 4-oxo-5-(arylformyl acetate-2-yl) naphthalene-sulfoxide ylide hybrids. This technology represents a paradigm shift in how chemists approach the functionalization of naphthalene cores, specifically by integrating sulfoxide ylide moieties directly onto the ring system through a streamlined catalytic process. The innovation lies in the ability to merge two distinct structural units—the 4-oxo-naphthalene framework and the versatile sulfoxide ylide—into a single molecular entity via a tandem reaction sequence. This approach not only addresses the historical scarcity of such hybrid structures in literature but also opens new avenues for developing luminescent materials and bioactive compounds. For R&D directors and procurement specialists, understanding the mechanistic elegance and operational simplicity of this patent is crucial for evaluating its potential impact on supply chain resilience and cost structures in API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of multifunctionalized naphthalene derivatives bearing both ketone and ylide functionalities has been a formidable synthetic challenge, often plagued by inefficiencies and environmental concerns. Conventional routes typically rely on multi-step sequences that require the isolation of unstable intermediates, leading to significant material loss and extended production timelines. Furthermore, many established protocols necessitate the use of stoichiometric amounts of strong acids or bases to drive cyclization or substitution reactions, which generates substantial quantities of hazardous waste and complicates downstream purification processes. The lack of atom economy in these older methods means that a large portion of the starting material ends up as byproduct rather than valuable product, driving up the cost of goods sold. Additionally, the harsh reaction conditions often limit the tolerance of sensitive functional groups, thereby restricting the chemical diversity that can be achieved. These factors collectively create bottlenecks in the supply of high-purity intermediates, forcing manufacturers to rely on expensive custom synthesis or accept lower yields that erode profit margins.
The Novel Approach
In stark contrast, the methodology described in CN109810036B utilizes a rhodium-catalyzed tandem reaction that elegantly bypasses these traditional hurdles by merging bond-forming events into a single operational step. By employing arylformyl sulfoxide ylide and alpha-diazocarbonyl compounds as raw materials, the process leverages the unique reactivity of rhodium carbenoids to initiate a cascade of transformations that construct the naphthalene core and install the ylide functionality simultaneously. This strategy exhibits exceptional atom economy, as the majority of atoms from the starting materials are incorporated into the final product structure, minimizing waste generation. Crucially, the reaction proceeds smoothly without the need for exogenous acids or bases, creating a greener and more environmentally benign profile that aligns with modern sustainability goals. The operational simplicity is further enhanced by the use of common organic solvents like toluene, which facilitates easy workup and solvent recovery. This novel approach not only accelerates the synthesis timeline but also expands the accessible chemical space, allowing for the introduction of diverse substituents on the aromatic rings without compromising reaction efficiency.
Mechanistic Insights into Rhodium-Catalyzed Tandem Cyclization
The success of this synthesis hinges on the precise orchestration of organometallic steps mediated by the rhodium catalyst, typically a pentamethylcyclopentadienyl rhodium dichloride dimer paired with a silver hexafluoroantimonate additive. The mechanism initiates with the decomposition of the alpha-diazocarbonyl compound by the rhodium species to generate a highly reactive metal-carbenoid intermediate. This electrophilic carbenoid then engages in a selective insertion or addition reaction with the electron-rich double bond of the arylformyl sulfoxide ylide. Following this initial coupling, the system undergoes an intramolecular cyclization, likely driven by the nucleophilic attack of the aromatic ring or the ylide carbon onto the activated carbonyl or carbenoid center, ultimately forging the fused naphthalene skeleton. The presence of the silver salt is critical for abstracting chloride ligands from the rhodium precursor, thereby generating the cationic active catalytic species that enhances the electrophilicity of the carbenoid. This delicate balance of electronic effects ensures that the reaction proceeds with high regioselectivity, favoring the formation of the desired 4-oxo-5-substituted naphthalene isomer over potential side products. Understanding this catalytic cycle allows process chemists to fine-tune parameters such as temperature and catalyst loading to maximize turnover numbers and minimize the formation of trace impurities that could affect downstream biological testing.
From an impurity control perspective, the mechanism inherently suppresses common side reactions associated with diazo compounds, such as dimerization or non-selective C-H insertion, due to the rapid trapping of the carbenoid by the sulfoxide ylide partner. The patent data highlights that when optimal solvents like toluene are used, TLC analysis reveals a clean reaction profile with minimal byproduct formation, indicating that the tandem pathway is kinetically favored over competing decomposition routes. The stability of the sulfoxide ylide moiety under these thermal conditions (60-100°C) is also noteworthy, as it survives the rigorous environment required for cyclization without undergoing premature hydrolysis or rearrangement. This robustness is essential for maintaining high purity specifications, as it reduces the burden on purification teams who would otherwise need to employ extensive chromatography to remove structurally similar impurities. For quality assurance teams, this mechanistic clarity provides confidence that the process is robust and reproducible, key factors for validating a manufacturing process for regulatory submission.
How to Synthesize 4-oxo-5-(arylformyl acetate-2-yl) naphthalene-sulfoxide ylide hybrid Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires strict adherence to the optimized conditions outlined in the patent to ensure consistent yields and product quality. The process begins with the preparation of the reaction vessel under an inert atmosphere, typically nitrogen or argon, to prevent the premature quenching of the sensitive diazo reagent by moisture or oxygen. Precise weighing of the arylformyl sulfoxide ylide and the alpha-diazocarbonyl compound is essential, with the patent suggesting a molar ratio ranging from 1:1 to 1:2.5 to drive the reaction to completion while minimizing excess reagent waste. The choice of solvent plays a pivotal role, with toluene and chlorobenzene identified as superior media that support high conversion rates and clean product profiles compared to polar alternatives. Once the reagents and the rhodium catalyst system are combined, the mixture is heated to a controlled temperature, ideally around 80°C, and maintained for a duration of approximately 24 hours to allow the tandem sequence to reach equilibrium. Monitoring the reaction progress via thin-layer chromatography ensures that the endpoint is accurately determined before proceeding to the quenching and extraction phases.
- Prepare the reaction mixture by combining arylformyl sulfoxide ylide and alpha-diazocarbonyl compound in an organic solvent such as toluene or chlorobenzene under inert gas protection.
- Add the rhodium catalyst, such as [RhCp*Cl2]2, and the silver hexafluoroantimonate additive to the reaction vessel in precise molar ratios.
- Heat the reaction mixture to a temperature between 60°C and 100°C, preferably 80°C, and stir for approximately 24 hours to complete the tandem cyclization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this rhodium-catalyzed technology offers tangible strategic benefits that extend beyond mere chemical novelty. The primary advantage lies in the significant simplification of the manufacturing workflow, which translates directly into reduced operational expenditures and enhanced supply reliability. By consolidating multiple synthetic steps into a single tandem reaction, the process eliminates the need for intermediate isolation, drying, and purification stages, which are often the most time-consuming and resource-intensive parts of chemical production. This streamlining drastically reduces the overall lead time required to produce batch quantities of the target intermediate, allowing suppliers to respond more agilely to fluctuating market demands. Furthermore, the elimination of harsh acidic or basic reagents simplifies waste management protocols, lowering the costs associated with hazardous waste disposal and environmental compliance. The use of standard solvents like toluene also ensures that raw material sourcing remains stable and cost-effective, avoiding reliance on exotic or supply-constrained chemicals. These factors collectively contribute to a more resilient supply chain capable of delivering high-purity pharmaceutical intermediates with greater consistency and lower total cost of ownership.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by its high atom economy and the avoidance of expensive stoichiometric reagents. Since the reaction does not require strong acids or bases, the costs associated with neutralization, salt disposal, and corrosion-resistant equipment are substantially mitigated. Additionally, the high selectivity of the rhodium catalyst minimizes the formation of difficult-to-remove impurities, reducing the consumption of silica gel and solvents during the purification phase. Although rhodium is a precious metal, the catalyst loading is low (typically 0.03-0.05 equivalents), and the potential for catalyst recovery or recycling in larger scale operations further amortizes this cost. The net result is a manufacturing route that offers a favorable cost profile compared to traditional multi-step syntheses, enabling competitive pricing for the final API or intermediate without sacrificing quality standards.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes significantly to supply chain stability. The tolerance of the method to various substituents on the aromatic rings means that a single platform technology can be used to produce a wide library of analogues, reducing the need for developing entirely new processes for each derivative. This flexibility allows manufacturers to pivot quickly between different product SKUs based on customer demand without retooling entire production lines. Moreover, the use of commercially available starting materials and common solvents reduces the risk of raw material shortages that often plague specialized chemical syntheses. The inert atmosphere requirement is standard in modern chemical plants, posing no significant barrier to implementation. Consequently, partners adopting this technology can expect a more predictable and continuous supply of critical intermediates, safeguarding their own production schedules against upstream disruptions.
- Scalability and Environmental Compliance: Scaling this synthesis from gram to kilogram or tonne levels is facilitated by the homogeneous nature of the catalytic system and the moderate thermal requirements. The reaction temperature of 60-100°C is easily achievable with standard heating jackets and does not require specialized high-pressure reactors, simplifying the engineering controls needed for scale-up. From an environmental standpoint, the green chemistry attributes of the process, such as high atom economy and reduced waste generation, align perfectly with increasingly stringent global regulations on chemical manufacturing. The absence of heavy metal waste streams (other than the trace rhodium which can be managed) and corrosive effluents makes the permitting process for new production facilities smoother. This compliance advantage not only future-proofs the supply chain against regulatory tightening but also enhances the corporate sustainability profile of companies utilizing this method, a factor of growing importance to end-users in the pharmaceutical sector.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating the feasibility of this synthesis for their specific applications, we have compiled answers to common inquiries regarding the process parameters and scope. These insights are derived directly from the experimental data provided in the patent documentation, ensuring accuracy and relevance for process development teams. Understanding the nuances of catalyst selection, solvent effects, and substrate compatibility is essential for successfully transferring this technology from the lab bench to commercial production. The following questions address the practical aspects of implementing the rhodium-catalyzed tandem reaction, providing a clear roadmap for overcoming potential technical hurdles.
Q: What are the key advantages of this rhodium-catalyzed method over traditional synthesis?
A: This method offers high atom economy through a tandem reaction sequence, eliminates the need for harsh acidic or basic conditions, and provides a broad substrate scope for generating diverse functionalized naphthalene derivatives.
Q: Which solvents are optimal for this transformation?
A: Research indicates that toluene and chlorobenzene are the preferred solvents, as they minimize impurity formation compared to other solvents like 1,2-dichloroethane or tetrahydrofuran.
Q: Is this process suitable for large-scale pharmaceutical manufacturing?
A: Yes, the process utilizes commercially available catalysts and operates under relatively mild thermal conditions without requiring extreme pressures, making it amenable to scale-up for industrial production of complex intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-oxo-5-(arylformyl acetate-2-yl) naphthalene-sulfoxide ylide hybrid Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the rhodium-catalyzed synthesis described in CN109810036B for advancing the production of complex pharmaceutical intermediates. As a leading CDMO partner, we possess the technical expertise and infrastructure necessary to translate this innovative academic research into robust, commercial-scale manufacturing processes. Our team of experienced process chemists is adept at optimizing catalytic systems, ensuring that the transition from milligram to multi-tonne production maintains the high yields and purity profiles demonstrated in the patent. We offer extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, leveraging our state-of-the-art facilities to meet the rigorous demands of the global pharmaceutical market. Our commitment to quality is reinforced by stringent purity specifications and rigorous QC labs that utilize advanced analytical techniques to verify the identity and integrity of every batch, ensuring that your supply chain remains uninterrupted and compliant.
We invite you to collaborate with us to explore how this advanced synthesis method can optimize your current manufacturing workflows and reduce overall production costs. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Our technical procurement team is ready to provide specific COA data and route feasibility assessments to demonstrate the viability of this technology for your portfolio. Whether you require small quantities for clinical trials or large-scale volumes for commercial launch, we are equipped to deliver reliable solutions that drive value and efficiency. Contact us today to discuss your project requirements and discover how our expertise in rhodium catalysis can accelerate your path to market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
