Advanced Chiral Resolution Technology for High-Purity Pharmaceutical Intermediates and Commercial Scale-Up
Advanced Chiral Resolution Technology for High-Purity Pharmaceutical Intermediates and Commercial Scale-Up
The pharmaceutical industry's relentless pursuit of enantiomerically pure compounds has driven significant innovation in chiral separation technologies, particularly for complex scaffolds like sulfonamide derivatives. Patent CN106660949B introduces a groundbreaking method for the chiral resolution of N-[4-(1-aminoethyl)-phenyl]-sulfonamide derivatives, which serve as critical intermediates in the synthesis of potent TRPV1 antagonists. This technology addresses the longstanding challenges of safety, cost, and scalability associated with traditional asymmetric synthesis routes. By leveraging a dual-auxiliary system comprising O,O'-diacyltartaric acid derivatives and soluble salt-forming compounds, manufacturers can achieve optical purities exceeding 96% ee without the need for hazardous cryogenic conditions. This report analyzes the technical merits of this resolution strategy and its profound implications for supply chain optimization and cost reduction in API manufacturing.
![General Formula I of N-[4-(1-aminoethyl)-phenyl]-sulfonamide derivatives subject to chiral resolution](/insights/img/chiral-sulfonamide-resolution-pharma-supplier-20260305195601-01.png)
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
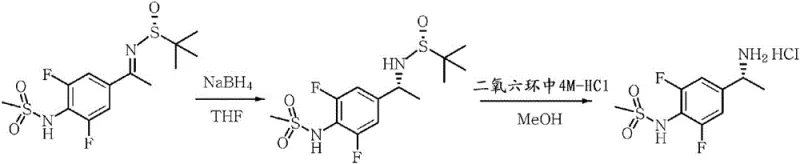
Historically, the preparation of single-isomer sulfonamide derivatives relied heavily on asymmetric synthesis utilizing Ellman's reagent (tert-butanesulfinamide). While chemically effective, this approach presents severe operational drawbacks for industrial applications. The process mandates strict low-temperature reaction conditions, often below -40°C, to maintain high stereocontrol, necessitating expensive cryogenic infrastructure and substantial energy consumption. Furthermore, the termination of the reduction step typically involves sodium borohydride, which generates excessive amounts of explosive hydrogen gas and heat, posing significant safety risks in large-scale reactors. The disposal of the resulting organic and inorganic waste streams adds another layer of economic and environmental burden, rendering the conventional route less attractive for commercial production of high-volume pharmaceutical intermediates.
The Novel Approach
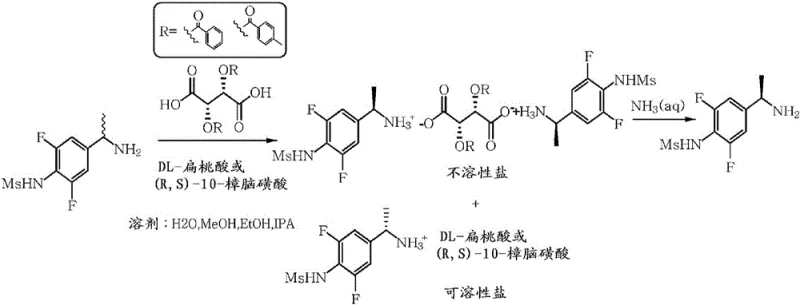
In stark contrast, the methodology disclosed in CN106660949B employs a classical yet optimized diastereomeric salt formation technique that circumvents these hazards. Instead of constructing the chiral center through dangerous reduction, this method resolves a pre-existing racemic or enriched mixture of N-[4-(1-aminoethyl)-phenyl]-sulfonamide derivatives. The core innovation lies in the specific combination of a chiral auxiliary, such as 2,3-dibenzoyl-tartaric acid, with a soluble salt-forming auxiliary like mandelic acid or camphorsulfonic acid. This dual-system creates a differential solubility environment where the desired enantiomer precipitates as an insoluble salt while the undesired isomer remains in solution. This shift from asymmetric synthesis to resolution allows the process to operate at mild temperatures between 30°C and 70°C, drastically simplifying the engineering requirements and enhancing overall process safety.
Mechanistic Insights into Diastereomeric Salt Formation
The efficacy of this resolution process is rooted in the precise thermodynamic control of crystal lattice formation. The mechanism involves the interaction of the amine group on the asymmetric carbon of the sulfonamide derivative with the carboxylic acid groups of the chiral auxiliaries. Specifically, the patent elucidates that two molecules of the sulfonamide derivative interact with one molecule of the O,O'-diacyltartaric acid derivative to form a stable, insoluble diastereomeric salt complex. This 2:1 stoichiometry is critical; using a 1:1 ratio results in inferior resolution efficiency. The presence of the second auxiliary, such as mandelic acid, acts as a solubility modifier, ensuring that the non-target enantiomer remains dissolved in the polar protic solvent matrix, thereby preventing co-precipitation and contamination of the solid phase.
Impurity control is inherently managed through the crystallization dynamics of this system. The patent data indicates that the optical purity of the precipitated salt is highly dependent on the molar ratio of the auxiliaries. Optimal results, achieving greater than 96% ee, are obtained when the chiral auxiliary is used in sub-stoichiometric amounts (0.25 to 0.5 equivalents) relative to the substrate, while the salt-forming auxiliary is used in near-stoichiometric or slight excess amounts (0.75 to 1.5 equivalents). This specific ratio minimizes the inclusion of the wrong enantiomer in the crystal lattice. Furthermore, the choice of solvent plays a pivotal role; polar protic solvents like isopropanol, methanol, and ethanol facilitate the necessary solvation shells around the ions, allowing for the selective nucleation of the target diastereomer while keeping impurities in the mother liquor.
How to Synthesize N-[4-(1-Aminoethyl)-phenyl]-sulfonamide Efficiently
The synthesis of these high-value intermediates begins with the preparation of the racemic sulfonamide scaffold, followed by the critical resolution step detailed in the patent. The process is designed to be robust and forgiving, accommodating variations in the initial enantiomeric ratio of the feed material. Operators mix the stereoisomer mixture with the defined chiral and salt-forming auxiliaries in a polar protic solvent, typically isopropanol, which offers an excellent balance of solubility and boiling point. The mixture is then heated to reflux, generally between 50°C and 70°C, and stirred for a period of 1 to 4 hours to ensure equilibrium is reached and the thermodynamically stable salt forms. Following the heating cycle, the solution is cooled to ambient temperature (15°C to 30°C) to induce crystallization. The resulting solid is filtered, washed, and subsequently treated with an aqueous ammonia solution to liberate the free base of the optically pure amine. Detailed standardized synthetic steps for this procedure are provided in the guide below.
- Mix the stereoisomer mixture of the sulfonamide derivative with an optically active O,O'-diacyltartaric acid derivative (chiral auxiliary) and a soluble salt-forming compound (e.g., mandelic acid) in a polar protic solvent.
- Stir the mixture under reflux at temperatures between 30°C to 70°C for 1 to 4 hours to facilitate the formation of diastereomeric salts.
- Cool the mixture to precipitate the insoluble diastereomeric salt, filter the solid, and treat with aqueous ammonia to liberate the high-purity optical isomer.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from asymmetric synthesis to this resolution methodology represents a strategic opportunity to de-risk the supply of critical TRPV1 antagonist intermediates. The elimination of cryogenic processing removes a major bottleneck in manufacturing capacity, allowing existing standard reactor trains to be utilized without specialized low-temperature modifications. This flexibility significantly enhances supply chain reliability, as production is no longer constrained by the availability of specialized cooling infrastructure or the safety protocols required for handling large volumes of hydrogen gas. Furthermore, the reagents employed—tartaric acid derivatives and mandelic acid—are commodity chemicals available from multiple global suppliers, reducing the risk of single-source dependency and price volatility associated with specialized chiral ligands.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived primarily from the drastic simplification of the operational parameters. By removing the need for energy-intensive cryogenic cooling and hazardous reducing agents, the utility costs per kilogram of product are substantially lowered. Additionally, the ability to recover and potentially recycle the chiral acids from the mother liquor contributes to a more sustainable cost structure. The higher throughput enabled by safer, faster reaction cycles at ambient pressures further drives down the fixed cost allocation per unit, making the final API more competitive in the global market without compromising on quality standards.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the robustness of the chemical process. Traditional methods involving sensitive reagents often suffer from batch-to-batch variability due to minor fluctuations in temperature or moisture, leading to yield losses and delivery delays. The resolution method described herein is inherently more stable, tolerating a wider range of operating conditions while consistently delivering high optical purity. This reliability ensures that downstream drug substance manufacturing schedules are met without interruption. Moreover, the use of common solvents like isopropanol simplifies logistics and waste management, as these materials are easily sourced and handled within standard chemical supply chains.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to multi-ton production is often fraught with heat transfer and safety challenges, particularly for exothermic reductions. This resolution technology sidesteps these issues entirely, as the crystallization process is endothermic or thermally neutral during the cooling phase, making scale-up predictable and linear. From an environmental perspective, the avoidance of boron-containing waste streams and the reduction of solvent usage through efficient crystallization align with modern green chemistry principles. This facilitates easier regulatory approval and reduces the environmental compliance burden on manufacturing sites, ensuring long-term operational sustainability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral resolution technology. These insights are derived directly from the experimental data and embodiments within the patent documentation, providing a clear understanding of the process capabilities and limitations for potential partners and licensees.
Q: What are the advantages of this chiral resolution method over asymmetric synthesis using Ellman's reagent?
A: Unlike asymmetric synthesis which requires hazardous cryogenic conditions (-40°C) and generates explosive hydrogen gas, this resolution method operates at mild temperatures (30-70°C) using stable reagents, significantly improving safety and reducing energy costs for commercial scale-up.
Q: Which chiral auxiliaries provide the highest optical purity for these sulfonamide derivatives?
A: The patent identifies O,O'-diacyltartaric acid derivatives, specifically 2,3-dibenzoyl-D-tartaric acid or O,O'-di-p-toluoyl-D-tartaric acid, used in combination with mandelic acid or camphorsulfonic acid, as the most effective system for achieving >96% ee.
Q: Is this process suitable for large-scale manufacturing of TRPV1 antagonists?
A: Yes, the process eliminates the need for expensive low-temperature equipment and hazardous reducing agents, making it highly scalable. The use of common polar protic solvents like isopropanol further enhances its viability for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-[4-(1-Aminoethyl)-phenyl]-sulfonamide Supplier
The technological advancements detailed in patent CN106660949B underscore the potential for producing high-purity chiral intermediates with unprecedented efficiency and safety. NINGBO INNO PHARMCHEM stands at the forefront of translating such innovative laboratory methodologies into robust commercial realities. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from development to market. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of verifying enantiomeric excess and impurity profiles to meet the most demanding global regulatory standards.
We invite you to collaborate with us to leverage this advanced resolution technology for your TRPV1 antagonist programs. Our technical team is prepared to conduct a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this process can optimize your bill of materials. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific excellence and commercial reliability.
