Revolutionizing Seleno-Oxazolidine-2,4-Dione Production with Visible Light Catalysis for Commercial Scale
The pharmaceutical and agrochemical industries are constantly seeking more efficient, sustainable, and cost-effective pathways to synthesize complex heterocyclic scaffolds that serve as critical building blocks for bioactive molecules. A significant breakthrough in this domain is documented in patent CN113603653A, which discloses a novel visible light-promoted synthesis method for seleno-oxazolidine-2,4-dione derivatives. This technology represents a paradigm shift from traditional thermal or transition-metal-catalyzed processes, leveraging clean photon energy to drive the cyclization of N-Boc protected acrylamides with diselenides. For R&D Directors and Procurement Managers alike, this innovation offers a compelling value proposition: it eliminates the need for harsh reaction conditions and expensive catalysts while delivering exceptional yields. As a reliable pharmaceutical intermediate supplier, understanding and adopting such cutting-edge synthetic methodologies is crucial for maintaining a competitive edge in the global market. This report delves into the technical nuances and commercial implications of this visible light-driven protocol, highlighting its potential to redefine cost reduction in pharmaceutical intermediate manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the oxazolidine-2,4-dione skeleton has relied on methodologies that present significant operational and safety challenges for large-scale production. Conventional routes often involve the condensation of alpha-hydroxyamides with carbonates or the reaction of alpha-hydroxy esters with urea or isocyanates. These traditional approaches are fraught with limitations, including the requirement for pre-prepared substrates that add steps to the synthesis tree, thereby increasing overall lead time. Furthermore, the use of isocyanates introduces severe toxicity concerns, necessitating specialized handling equipment and rigorous safety protocols that inflate operational expenditures. Alternative methods utilizing carbon dioxide as a C1 resource often demand high temperature and high pressure conditions, which not only consume substantial energy but also pose safety risks in a commercial plant setting. Additionally, many existing protocols suffer from moderate atom economy and inconsistent yields, leading to significant material loss and complicated downstream purification processes that hinder the commercial scale-up of complex polymer additives or pharmaceutical intermediates.
The Novel Approach
In stark contrast, the visible light-promoted method described in CN113603653A offers a streamlined, green chemistry solution that directly addresses the inefficiencies of prior art. By utilizing visible light as the sole energy source, this novel approach operates under remarkably mild conditions, typically at room temperature and atmospheric pressure, without the need for external heating or pressurization. The reaction employs readily available N-Boc protected acrylamides and diselenides as starting materials, which are commercially accessible and stable, thus enhancing supply chain reliability. The process is notably insensitive to air and moisture, removing the stringent requirement for inert gas protection and allowing for simpler reactor setups. This robustness translates to reduced lead time for high-purity intermediates, as the operational complexity is drastically minimized. Moreover, the method achieves high reaction efficiency with excellent functional group tolerance, enabling the synthesis of diverse derivatives without compromising yield or purity, making it an ideal candidate for modern, sustainable manufacturing pipelines.
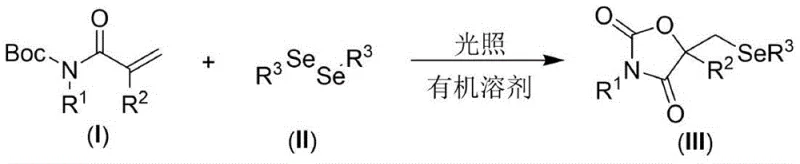
Mechanistic Insights into Visible Light-Promoted Cyclization
The core of this technological advancement lies in its unique photochemical mechanism, which facilitates the formation of the selenium-carbon bond and subsequent cyclization without the aid of transition metal catalysts. Under visible light irradiation, the diselenide bond undergoes homolytic cleavage to generate selenyl radicals. These reactive species then add across the double bond of the N-Boc protected acrylamide substrate, initiating a radical cascade sequence. The resulting carbon-centered radical undergoes intramolecular cyclization onto the carbonyl group, followed by oxidation and proton loss to furnish the seleno-oxazolidine-2,4-dione core. This metal-free pathway is particularly advantageous for R&D teams focused on purity and impurity profiles, as it inherently eliminates the risk of heavy metal contamination that often plagues palladium or copper-catalyzed reactions. Consequently, the burden on downstream purification is significantly reduced, ensuring that the final high-purity OLED material or pharmaceutical intermediate meets stringent regulatory specifications with minimal processing.
Furthermore, the mechanism demonstrates exceptional compatibility with a wide range of substituents, which is critical for developing a versatile library of bioactive compounds. The radical nature of the reaction allows it to tolerate various electronic environments on the aromatic rings of both the acrylamide and the diselenide components. Whether the substrate contains electron-withdrawing groups like halogens or electron-donating groups like alkyl or alkoxy moieties, the reaction proceeds smoothly to afford the desired products in high yields. This broad substrate scope implies that the process can be adapted for the synthesis of diverse analogs without extensive re-optimization, accelerating the drug discovery timeline. The ability to maintain high efficiency across different structural variants ensures consistent quality and reduces the variability often associated with multi-step synthetic routes, thereby strengthening the overall robustness of the manufacturing process for specialty chemical applications.
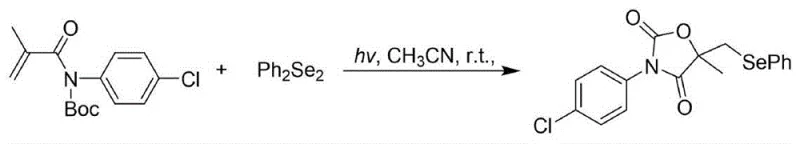
How to Synthesize Seleno-Oxazolidine-2,4-Dione Efficiently
Implementing this visible light-promoted synthesis in a laboratory or pilot plant setting involves a straightforward procedure that aligns with standard organic synthesis practices while leveraging photochemical equipment. The process begins with the preparation of the reaction mixture, where N-Boc protected acrylamide and diselenide are dissolved in a suitable organic solvent such as acetonitrile. The simplicity of the reagent setup minimizes preparation time and reduces the potential for human error during the charging phase. Once the solution is prepared, it is subjected to irradiation from a common visible light source, such as a white compact fluorescent lamp, positioned at a close distance to ensure adequate photon flux. The reaction is allowed to stir at room temperature for a period ranging from 26 to 36 hours, during which the transformation proceeds to completion without the need for temperature control or inert atmosphere. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by combining N-Boc protected acrylamide and diselenide in an organic solvent such as acetonitrile.
- Irradiate the reaction mixture with a visible light source, such as a fluorescent lamp, at room temperature for 26 to 36 hours.
- Purify the crude product using column chromatography with a petroleum ether and ethyl acetate eluent system to obtain the target compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this visible light technology offers tangible benefits that extend beyond mere technical novelty, directly impacting the bottom line and operational resilience. The elimination of expensive transition metal catalysts represents a significant cost reduction in manufacturing, as it removes the need for purchasing precious metals and the associated costs of scavenging them from the final product to meet regulatory limits. Furthermore, the mild reaction conditions reduce energy consumption substantially, as there is no requirement for heating mantles, oil baths, or high-pressure reactors, leading to lower utility bills and a smaller carbon footprint. The use of commercially available and stable starting materials enhances supply chain reliability, mitigating the risk of delays caused by the sourcing of exotic or unstable reagents. This stability ensures continuous production capabilities, which is vital for meeting the demanding delivery schedules of multinational pharmaceutical clients.
- Cost Reduction in Manufacturing: The economic advantages of this protocol are driven by the simplification of the reaction setup and the removal of costly catalytic systems. By avoiding transition metals, the process sidesteps the expensive purification steps required to reduce metal residues to ppm levels, which is a mandatory requirement for API intermediates. Additionally, the high yields reported in the patent, often exceeding 90%, mean that less raw material is wasted, improving the overall material efficiency and reducing the cost per kilogram of the final product. The simplicity of the workup, involving standard solvent removal and column chromatography, further lowers labor and processing costs, making the entire manufacturing route more economically viable for large-scale production.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes significantly to supply chain stability. Since the reaction is insensitive to air and moisture, it does not require specialized glovebox techniques or rigorous drying of solvents, which simplifies the operational workflow and reduces the likelihood of batch failures due to environmental factors. The starting materials, N-Boc acrylamides and diselenides, are either commercially available or easily synthesized from bulk chemicals, ensuring a steady supply stream. This accessibility reduces the dependency on single-source suppliers for niche reagents, thereby diversifying the supply base and minimizing the risk of disruptions. Such reliability is crucial for maintaining consistent inventory levels and fulfilling long-term contracts with key stakeholders in the fine chemical industry.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this method aligns perfectly with modern green chemistry principles. The use of visible light as a renewable energy source reduces the reliance on fossil-fuel-derived energy, supporting corporate sustainability goals. The absence of toxic reagents like isocyanates and the generation of minimal waste simplify waste management and disposal procedures, ensuring compliance with increasingly stringent environmental regulations. The mild conditions also facilitate safer scale-up, as the risks associated with exothermic runaways or high-pressure incidents are negligible. This safety profile allows for larger batch sizes without proportional increases in safety infrastructure, enabling efficient commercial scale-up of complex heterocyclic compounds to meet growing market demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this visible light-promoted synthesis method. These answers are derived directly from the experimental data and beneficial effects outlined in the patent documentation, providing clarity on the practical aspects of the technology. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The insights provided here aim to bridge the gap between academic innovation and industrial application, ensuring that all stakeholders have a clear understanding of the process capabilities and limitations.
Q: What are the advantages of using visible light for this synthesis compared to traditional methods?
A: Traditional methods often require toxic isocyanates, high temperatures, or expensive transition metal catalysts. This visible light-promoted method operates under mild conditions, is insensitive to air and water, and avoids heavy metal contamination, significantly simplifying purification and reducing environmental impact.
Q: Is this method scalable for commercial production of pharmaceutical intermediates?
A: Yes, the method utilizes readily available starting materials like N-Boc protected acrylamide and diselenides. The reaction conditions are mild (room temperature, atmospheric pressure), and the workup involves standard column chromatography, making it highly suitable for commercial scale-up of complex heterocyclic compounds.
Q: What kind of yields can be expected with this visible light protocol?
A: The patent data indicates consistently high yields, ranging from 90% to 96% across various substrates including those with electron-withdrawing and electron-donating groups. This high efficiency ensures minimal waste and maximizes material throughput for supply chain reliability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Seleno-Oxazolidine-2,4-Dione Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of visible light photocatalysis in the synthesis of high-value heterocyclic intermediates. Our team of expert chemists is well-versed in adapting such innovative protocols for commercial production, ensuring that the theoretical benefits of patents like CN113603653A are realized in practical, large-scale operations. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, guaranteeing that your supply needs are met with consistency and precision. Our facilities are equipped with state-of-the-art rigorous QC labs that enforce stringent purity specifications, ensuring that every batch of seleno-oxazolidine-2,4-dione delivered meets the highest industry standards. By partnering with us, you gain access to a supply chain that is not only reliable but also committed to continuous improvement and technological advancement.
We invite you to explore the possibilities of this advanced synthesis method for your specific project requirements. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume and purity needs, demonstrating how this green chemistry approach can optimize your budget. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on concrete performance metrics. Let us collaborate to bring this efficient, sustainable, and cost-effective technology to your production line, driving innovation and value creation in your pharmaceutical or agrochemical development pipeline.
