Advanced Manufacturing of Sonidegib Intermediates via Green Epoxide Ring-Opening Technology
The pharmaceutical landscape for oncology treatments continues to evolve, with Sonidegib (also known as Sonchibu or Odomzo) standing as a critical SMO receptor antagonist for treating locally advanced basal cell carcinoma. As demand for this potent Hedgehog pathway inhibitor grows, the efficiency of its supply chain becomes paramount for global health security. Patent CN109293649B introduces a transformative synthetic methodology that redefines the production of Sonidegib intermediates, specifically targeting the costly and complex morpholine-pyridine core. Unlike traditional routes that rely on prohibitively expensive chiral starting materials, this innovation leverages a strategic epoxide ring-opening strategy starting from readily available 2-amino-5-nitropyridine. This approach not only simplifies the synthetic sequence but also drastically improves the environmental profile by eliminating hazardous diazotization steps. For industry stakeholders, this represents a pivotal shift towards more sustainable and economically viable pharmaceutical intermediates manufacturing.
The implementation of this technology addresses the critical need for cost reduction in API manufacturing while maintaining the rigorous purity standards required for oncology drugs. By bypassing the need for pre-formed chiral morpholines, manufacturers can secure a more resilient supply chain less susceptible to the volatility of niche chiral reagent markets. The process flow, as detailed in the patent, outlines a robust pathway from simple precursors to the high-value amine intermediate, ready for final amidation. This report analyzes the technical merits and commercial implications of adopting this novel route for the commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Sonidegib has been hindered by reliance on inefficient and costly upstream processes. As illustrated in prior art schemes, conventional methods often commence with 2-chloro-5-nitropyridine, a substrate that itself requires a tedious three-step preparation involving nitration, diazotization, hydrolysis, and chlorination of 2-aminopyridine. This sequence is not only operationally hazardous due to the handling of diazonium salts but also generates substantial volumes of acidic wastewater, posing significant environmental compliance challenges. Furthermore, these routes typically depend on the coupling of this chloropyridine with cis-2,6-dimethylmorpholine. This chiral morpholine reagent is notoriously expensive and difficult to source in bulk quantities, creating a bottleneck for reliable API intermediate supplier networks aiming for large-scale production.
Alternative prior art strategies attempt to mitigate some risks by performing Suzuki couplings early in the sequence to build the biphenyl core before amidation. However, these methods still fundamentally rely on the same expensive chiral amine building blocks and often involve palladium-catalyzed cross-couplings that require rigorous metal scavenging to meet residual metal specifications. The cumulative effect of these inefficiencies is a high cost of goods sold (COGS) and a fragile supply chain vulnerable to raw material shortages. The complexity of purifying intermediates from these multi-step sequences further erodes overall yield, making the final high-purity pharmaceutical intermediate economically challenging to produce at the multi-ton scale required for global market penetration.
The Novel Approach
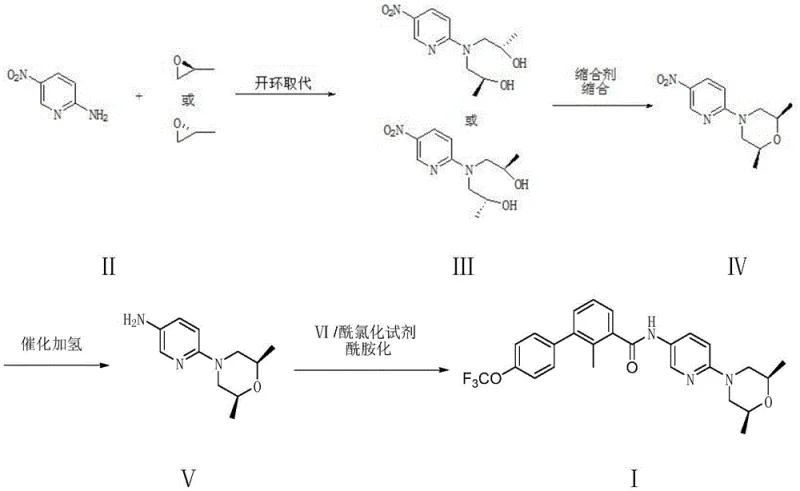
The methodology disclosed in CN109293649B offers a paradigm shift by reconstructing the morpholine ring in situ rather than purchasing it pre-formed. The novel approach initiates with the nucleophilic attack of 2-amino-5-nitropyridine on enantiomerically pure propylene oxide (either R or S configuration). This ring-opening substitution is highly selective and utilizes inexpensive, commodity-grade epoxides, effectively decoupling the production cost from the volatile chiral amine market. Following the formation of the bis-hydroxypropylamino intermediate, a sophisticated cyclization step closes the morpholine ring using a condensing agent under controlled conditions. This strategy reduces the step count for the key intermediate synthesis from three or more steps down to just two highly efficient transformations.
By avoiding the diazotization pathway entirely, the new route eliminates a major source of process wastewater and safety risk, aligning perfectly with modern green chemistry mandates. The use of 2-amino-5-nitropyridine as a direct starting material ensures that the nitrogen functionality is already correctly positioned, removing the need for complex functional group interconversions. This streamlined logic results in a process that is not only safer and cleaner but also inherently more scalable. For procurement teams, this translates to a reliable agrochemical intermediate supplier grade reliability for pharma applications, where consistency and continuity of supply are non-negotiable. The ability to generate the chiral center from cheap epoxides rather than expensive amines is the cornerstone of the economic advantage offered by this technology.
Mechanistic Insights into Lewis Acid-Catalyzed Epoxide Ring-Opening
The success of this novel route hinges on the precise control of the initial ring-opening reaction between 2-amino-5-nitropyridine and propylene oxide. The patent specifies the use of Lewis acids such as zinc chloride, ferric chloride, or aluminum trichloride to catalyze this transformation. Mechanistically, the Lewis acid coordinates with the oxygen atom of the epoxide ring, increasing the electrophilicity of the adjacent carbon atoms and facilitating the nucleophilic attack by the amino group of the pyridine. This activation allows the reaction to proceed smoothly at moderate temperatures (40-60°C) in solvents like tetrahydrofuran or dichloromethane. The stoichiometry is critical, utilizing approximately 2.0 to 2.5 equivalents of propylene oxide to ensure complete conversion to the bis-substituted product, N,N-bis(2-hydroxypropylamino)-5-nitropyridine, without significant oligomerization.
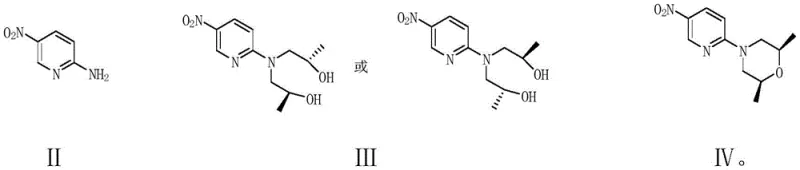
Following the ring-opening, the formation of the morpholine ring via cyclization is a masterpiece of process control. The patent highlights a "double-dropping" technique where both the bis-hydroxypropylamino intermediate and the condensing agent (such as trifluoromethanesulfonic anhydride) are added simultaneously and slowly into a basic reaction mixture. This kinetic control maintains a low instantaneous concentration of reactants, which is vital for selectivity. If the concentration were too high, intermolecular reactions or disulfonation of both hydroxyl groups could occur, leading to polymerization or inactive by-products. Instead, the low concentration favors the formation of a mono-sulfonate ester, which immediately undergoes an intramolecular SN2 substitution by the remaining hydroxyl group. This intramolecular attack closes the six-membered morpholine ring with high stereospecificity, preserving the chirality introduced by the propylene oxide and yielding the target (2S,6R)-2,6-dimethyl-4-(5-nitropyridin-2-yl)morpholine with exceptional purity.
How to Synthesize Sonidegib Intermediate Efficiently
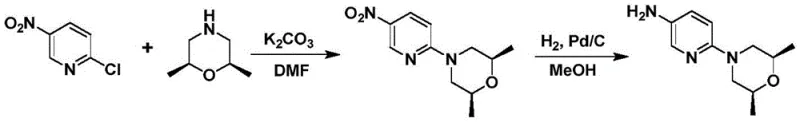
The synthesis of the key Sonidegib intermediate involves a carefully orchestrated sequence beginning with the Lewis acid-catalyzed alkylation of 2-amino-5-nitropyridine. Operators must maintain strict temperature control between 40°C and 60°C during the epoxide addition to prevent exothermic runaway while ensuring complete conversion. Following the isolation of the bis-hydroxypropylamino species, the cyclization step requires precise metering pumps to achieve the simultaneous dropwise addition described in the mechanistic section. The reaction mixture must be kept basic, typically using potassium carbonate or triethylamine, to scavenge the acid generated during sulfonate formation. Once the morpholine nitro-compound is secured, the final transformation involves a standard catalytic hydrogenation.
- Perform Lewis acid-catalyzed ring-opening substitution of 2-amino-5-nitropyridine with two equivalents of propylene oxide in a solvent like THF at 40-60°C to form the bis-hydroxypropylamino derivative.
- Execute a controlled condensation cyclization by simultaneously dropwise adding the bis-hydroxypropylamino derivative and a condensing agent (e.g., trifluoromethanesulfonic anhydride) into a basic reaction mixture to form the morpholine ring.
- Reduce the nitro group of the resulting morpholine intermediate via catalytic hydrogenation (Pd/C or Raney Ni) to yield the final amine precursor for Sonidegib.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers compelling strategic advantages that extend beyond simple unit price reductions. The primary value driver is the substitution of high-cost, low-availability chiral amines with commodity chemicals. Propylene oxide and 2-amino-5-nitropyridine are produced on a massive industrial scale for other sectors, ensuring a stable and competitive pricing structure that is immune to the supply shocks often seen in the fine chemical sector. This shift fundamentally alters the cost structure of the intermediate, allowing for significant margin improvement or more aggressive pricing strategies in the final API market. Furthermore, the simplification of the process flow reduces the number of unit operations, which directly correlates to lower capital expenditure (CAPEX) requirements for new production lines and reduced operational expenditure (OPEX) for existing facilities.
- Cost Reduction in Manufacturing: The elimination of the expensive cis-2,6-dimethylmorpholine starting material represents a direct and substantial decrease in raw material costs. Additionally, by removing the diazotization and chlorination steps required to make 2-chloro-5-nitropyridine, the process saves on reagents, energy, and waste disposal fees. The higher overall yield resulting from fewer purification steps further amplifies these savings, ensuring that the cost per kilogram of the final intermediate is drastically optimized without compromising quality.
- Enhanced Supply Chain Reliability: Relying on commodity feedstocks like propylene oxide mitigates the risk of supply interruptions associated with specialized chiral reagents. The robustness of the chemistry, which tolerates standard industrial solvents and conditions, means that production can be easily transferred between different manufacturing sites globally. This flexibility is crucial for maintaining reducing lead time for high-purity pharmaceutical intermediates, ensuring that downstream API synthesis is never delayed by upstream bottlenecks. The simplified logistics of sourcing fewer, more common ingredients also streamline inventory management.
- Scalability and Environmental Compliance: The green nature of this process, characterized by reduced wastewater generation and the absence of hazardous diazonium intermediates, facilitates easier regulatory approval and permitting for new plants. The "double-dropping" cyclization technique is readily scalable from pilot to commercial reactors using standard dosing equipment. This scalability ensures that the technology can support the transition from clinical trial materials to commercial tonnage seamlessly. Moreover, the reduced environmental footprint aligns with the increasing ESG (Environmental, Social, and Governance) criteria demanded by top-tier pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented Sonidegib synthesis route. These answers are derived directly from the experimental data and process descriptions found in CN109293649B, providing clarity on the feasibility and benefits of the technology. Understanding these details is essential for technical teams evaluating the transfer of this process to their own manufacturing infrastructure.
Q: How does this new route reduce raw material costs compared to conventional methods?
A: Conventional methods rely on expensive chiral cis-2,6-dimethylmorpholine or require multi-step synthesis of 2-chloro-5-nitropyridine involving hazardous diazotization. This novel route utilizes cheap, commercially available 2-amino-5-nitropyridine and propylene oxide, significantly lowering the Bill of Materials (BOM) cost.
Q: What are the environmental advantages of avoiding the diazotization step?
A: Traditional synthesis of the pyridine core often involves diazotization and hydrolysis, which generates large volumes of acidic wastewater and requires complex treatment. By starting directly from 2-amino-5-nitropyridine, this process eliminates those specific waste streams, aligning better with green chemistry principles and reducing E-factor.
Q: How is high stereochemical purity maintained during the morpholine ring formation?
A: The process uses enantiomerically pure propylene oxide (R or S) for the initial ring-opening. The subsequent cyclization is an intramolecular SN2 substitution that preserves the chiral centers established in the first step, ensuring the final product meets the strict (2S,6R) stereochemical requirements without needing resolution.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sonidegib Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this novel synthetic route for Sonidegib production. As a premier CDMO partner, we possess the technical expertise and infrastructure to translate this patent-protected methodology into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate kinetics of the epoxide ring-opening and cyclization steps are perfectly managed at scale. We are committed to delivering products that meet stringent purity specifications, utilizing our rigorous QC labs to verify that every batch of intermediate conforms to the highest standards required for oncology drug substance manufacturing.
We invite pharmaceutical partners to collaborate with us to leverage these cost and efficiency advantages. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments to understand how this green, economical pathway can strengthen your supply chain for Sonidegib and related Hedgehog pathway inhibitors. Let us help you secure a competitive edge through superior process chemistry.
