Advanced One-Pot Synthesis of 5-Amino-2-Nitrobenzoic Acid for High-Purity Diagnostic Reagents
The pharmaceutical and diagnostic industries are constantly seeking more efficient and safer pathways for synthesizing critical intermediates like 5-amino-2-nitrobenzoic acid. Patent CN116283629A introduces a groundbreaking preparation method that fundamentally shifts the synthetic paradigm from hazardous oxidation processes to a streamlined, one-pot acetylation and nitration strategy. This innovation addresses long-standing challenges in the production of Glupa-C substrates, which are essential for the quantitative detection of gamma-glutamyl transpeptidase in human serum. By utilizing m-aminobenzoic acid as a direct starting material, the process bypasses the need for risky oxidation steps involving potassium permanganate, thereby eliminating heavy metal contamination issues that have plagued previous manufacturing protocols. The technical breakthrough lies in the precise control of reaction conditions, specifically maintaining temperatures below 35°C during the addition of acetic anhydride and concentrated sulfuric acid, ensuring high selectivity and yield without compromising safety.
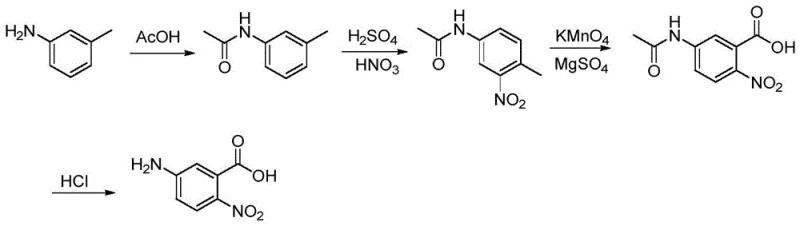
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 5-amino-2-nitrobenzoic acid relied heavily on routes starting from m-toluidine, as illustrated in the conventional pathway. This traditional approach necessitates a multi-step sequence involving acetylation, nitration, and a critical oxidation step where the methyl group is converted to a carboxylic acid using potassium permanganate. This oxidation stage presents significant industrial hazards, including the potential for runaway exothermic reactions and explosion risks associated with handling large quantities of strong oxidizers. Furthermore, the use of manganese-based reagents introduces severe downstream processing burdens, requiring complex filtration and purification procedures to remove heavy metal residues that could otherwise contaminate the final pharmaceutical grade product. The lengthy reaction sequence also results in cumulative yield losses and increased solvent consumption, making the process economically inefficient and environmentally unsustainable for modern large-scale manufacturing facilities.
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent utilizes a direct functionalization strategy starting from m-aminobenzoic acid, effectively circumventing the need for side-chain oxidation. As depicted in the reaction scheme, the process employs a clever one-pot technique where acetylation and nitration occur sequentially in the same reactor vessel without isolating the intermediate m-acetamidobenzoic acid. By selecting glacial acetic acid as the solvent, the system ensures mutual solubility during the nitration phase, facilitating smooth reaction kinetics and excellent regioselectivity for the 5-position. This approach not only drastically reduces the number of unit operations but also inherently improves the safety profile by removing the most dangerous step of the legacy process. The result is a robust, scalable protocol that delivers high-purity intermediates suitable for sensitive diagnostic applications while minimizing waste generation and operational complexity.
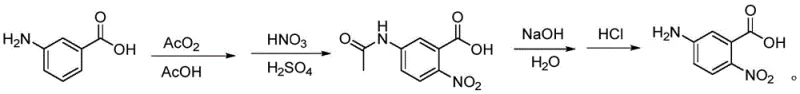
Mechanistic Insights into Regioselective Nitration and Acetylation
The core chemical innovation of this process lies in the precise manipulation of electronic effects to achieve high regioselectivity during the nitration of the acetamido-protected intermediate. When m-aminobenzoic acid is treated with acetic anhydride, the amino group is rapidly converted into an acetamido group, which serves as a moderate activator and an ortho/para director. Simultaneously, the carboxylic acid group acts as a meta-directing deactivator. The interplay between these directing groups, combined with the steric environment created by the solvent system, favors electrophilic attack at the position para to the acetamido group, which corresponds to the 5-position relative to the carboxyl group in the final numbering system. The use of fuming nitric acid in the presence of concentrated sulfuric acid generates the potent nitronium ion electrophile necessary for this transformation, while the low temperature control (20-35°C) suppresses the formation of dinitro by-products and prevents hydrolysis of the acetamido protecting group during the nitration phase.
Impurity control is rigorously managed through a combination of reaction condition optimization and a targeted recrystallization step. The patent specifies the use of absolute ethyl alcohol for recrystallizing the crude 5-acetamido-2-nitrobenzoic acid, a solvent choice that exploits the differential solubility of the desired product versus unreacted starting materials or isomeric by-products. Heating the crude solid to reflux in absolute ethanol ensures complete dissolution, followed by controlled cooling to induce the formation of high-quality crystals. This purification step is critical for removing trace amounts of ortho-nitrated isomers or residual acids that could interfere with the subsequent hydrolysis. The final hydrolysis step, conducted under mild alkaline conditions followed by careful pH adjustment to 2.0 using hydrochloric acid, ensures the quantitative recovery of the free amine without degrading the sensitive nitro functionality, resulting in a final product with purity levels exceeding 99.8%.
How to Synthesize 5-Amino-2-Nitrobenzoic Acid Efficiently
The synthesis protocol outlined in the patent offers a practical guide for laboratory and pilot-scale production, emphasizing the importance of thermal management and stoichiometric precision. The process begins with the suspension of m-aminobenzoic acid in glacial acetic acid, followed by the controlled addition of acetic anhydride to effect amidation. Without isolation, the reaction mixture is treated with concentrated sulfuric acid and fuming nitric acid to introduce the nitro group, leveraging the solubility of the intermediate in the acidic medium to maintain homogeneity. Detailed standardized synthesis steps, including specific mass ratios and temperature profiles for each addition, are provided in the technical documentation to ensure reproducibility and safety during scale-up operations.
- Perform one-pot acetylation and nitration of m-aminobenzoic acid using acetic anhydride, glacial acetic acid, sulfuric acid, and fuming nitric acid below 35°C.
- Isolate the crude 5-acetamido-2-nitrobenzoic acid and purify via recrystallization in absolute ethyl alcohol.
- Hydrolyze the purified intermediate using sodium hydroxide followed by acidification with hydrochloric acid to precipitate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route represents a significant opportunity to optimize cost structures and mitigate supply risks associated with 5-amino-2-nitrobenzoic acid. The elimination of the potassium permanganate oxidation step removes a major bottleneck in the production workflow, as it obviates the need for specialized equipment designed to handle explosive oxidizers and the costly disposal of manganese-containing waste streams. This simplification of the chemical process translates directly into reduced operational expenditures, as fewer reaction vessels and shorter cycle times are required to produce the same quantity of finished goods. Furthermore, the reliance on commodity chemicals such as acetic acid, sulfuric acid, and nitric acid ensures a stable and resilient supply chain, insulating manufacturers from the volatility often seen in the market for specialized oxidizing agents or transition metal catalysts.
- Cost Reduction in Manufacturing: The primary driver for cost savings in this new method is the drastic reduction in downstream processing requirements. By avoiding the generation of heavy metal sludge, manufacturers save substantially on waste treatment fees and the capital investment required for effluent treatment plants capable of removing manganese residues. Additionally, the one-pot nature of the acetylation and nitration steps minimizes solvent usage and energy consumption associated with heating, cooling, and transferring materials between multiple reactors. The high yield reported in the patent examples suggests that raw material utilization is maximized, further lowering the cost per kilogram of the active pharmaceutical ingredient precursor and enhancing overall profit margins for producers of diagnostic reagents.
- Enhanced Supply Chain Reliability: Switching to a synthesis route based on m-aminobenzoic acid enhances supply security because the starting material is a widely available bulk chemical with a mature global supply network. Unlike processes dependent on specific grades of oxidizers that may face regulatory shipping restrictions or seasonal availability issues, the reagents used in this protocol are standard industrial commodities. This stability allows for more accurate demand forecasting and inventory planning, reducing the likelihood of production stoppages due to raw material shortages. The robustness of the reaction conditions, which tolerate slight variations in temperature and addition rates without significant loss of quality, also contributes to consistent batch-to-batch reliability, ensuring that downstream customers receive their orders on time and to specification.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process offers a clear path to sustainable manufacturing. The absence of heavy metals simplifies the regulatory compliance landscape, making it easier to obtain necessary permits for expansion or operation in regions with strict environmental laws. The simplified workup procedure, involving straightforward filtration and crystallization, is inherently easier to scale from kilogram to tonnage quantities compared to complex oxidation workflows that require careful monitoring of exotherms. This scalability ensures that suppliers can rapidly ramp up production to meet surging demand for diagnostic tests without compromising on safety or environmental standards, positioning companies that adopt this technology as leaders in green chemistry and responsible sourcing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this technology outperforms legacy methods in terms of purity, safety, and efficiency. Understanding these nuances is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Why is the one-pot method superior to the traditional oxidation route?
A: The traditional route involves oxidizing a methyl group with potassium permanganate, which generates heavy metal waste and poses explosion risks. The new one-pot method avoids oxidation entirely, significantly improving safety and environmental compliance.
Q: What is the purity level achievable with this new process?
A: According to patent data, the process yields 5-amino-2-nitrobenzoic acid with a purity of up to 99.85% after recrystallization and hydrolysis, meeting stringent requirements for diagnostic applications.
Q: Does this method require expensive transition metal catalysts?
A: No, the synthesis relies on standard mineral acids (sulfuric and nitric) and acetic anhydride. This eliminates the need for costly transition metal catalysts and the subsequent purification steps required to remove metal residues.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Amino-2-Nitrobenzoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity intermediates play in the reliability of diagnostic assays and pharmaceutical formulations. Our team of expert chemists has extensively analyzed the technical merits of the one-pot nitration process described in CN116283629A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are committed to delivering 5-amino-2-nitrobenzoic acid that meets stringent purity specifications, utilizing rigorous QC labs to verify that every batch is free from heavy metal contaminants and isomeric impurities. Our state-of-the-art facilities are equipped to handle the specific thermal and corrosive requirements of this synthesis, ensuring a consistent supply of this vital building block for the global healthcare industry.
We invite procurement leaders and R&D directors to collaborate with us to leverage these advanced manufacturing capabilities for your specific project needs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential efficiencies of switching to this greener synthesis route. We encourage you to contact us today to obtain specific COA data for our current stock and to discuss detailed route feasibility assessments tailored to your volume requirements, ensuring a seamless integration of this high-performance intermediate into your supply chain.
