Revolutionizing Allyl Phosphorus Synthesis: A Green Catalytic Route for High-Purity Pharmaceutical Intermediates
Revolutionizing Allyl Phosphorus Synthesis: A Green Catalytic Route for High-Purity Pharmaceutical Intermediates
The landscape of organophosphorus chemistry is undergoing a significant transformation driven by the urgent need for greener, more efficient synthetic methodologies in the pharmaceutical and agrochemical sectors. Patent CN111943980B introduces a groundbreaking preparation method for allyl phosphorus compounds that addresses critical bottlenecks in traditional synthesis. This technology leverages the unique Lewis acidity of tris(pentafluorophenyl)borane to catalyze the direct dehydrative coupling of secondary allyl alcohols with phosphorus-hydrogen compounds. Unlike conventional routes that rely on harsh conditions or toxic heavy metals, this innovation operates under mild thermal conditions with water as the sole byproduct, representing a paradigm shift towards sustainable chemical manufacturing. For R&D directors and procurement strategists, this patent offers a compelling pathway to access high-value intermediates with improved atom economy and reduced environmental footprint.
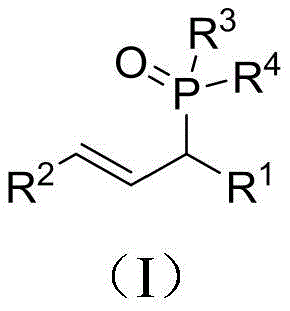
Organophosphorus compounds serve as indispensable building blocks in modern medicinal chemistry, functioning as C-H activation directing groups, versatile reagents, and key motifs in bioactive molecules such as NAALADase inhibitors and herbicides. The structural versatility of the allyl phosphorus backbone allows for extensive downstream functionalization, making it a prized scaffold in drug discovery. However, the historical reliance on transition metal catalysis has imposed significant burdens on supply chains, including high catalyst costs, difficult removal of metal residues, and sensitivity to air and moisture. The methodology disclosed in this patent circumvents these issues by utilizing a robust, metal-free catalytic system that maintains high conversion rates across a broad spectrum of substrates, ensuring consistent quality for complex pharmaceutical pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis of allyl phosphorus compounds has predominantly relied on transition metal-catalyzed cross-coupling reactions or addition reactions under strong basic conditions. These legacy methods suffer from inherent inefficiencies, including the requirement for expensive ligands and stoichiometric oxidants which drastically inflate production costs. Furthermore, the use of transition metals like palladium necessitates rigorous purification steps to meet stringent regulatory limits on heavy metal residues in active pharmaceutical ingredients (APIs). The harsh reaction conditions often associated with strong bases can lead to poor chemoselectivity and the formation of complex impurity profiles, complicating the isolation of the desired product. From a supply chain perspective, the dependency on specialized catalysts and sensitive reagents introduces volatility and potential delays in manufacturing schedules.
The Novel Approach
The innovative approach detailed in the patent utilizes tris(pentafluorophenyl)borane as a highly effective Lewis acid catalyst to activate the hydroxyl group of secondary allyl alcohols directly. This activation facilitates a nucleophilic attack by the phosphorus-hydrogen bond, resulting in the elimination of water and the formation of the C-P bond under neutral to mildly acidic conditions. This method eliminates the need for exogenous oxidants or strong bases, thereby enhancing the safety profile and operational simplicity of the process. The reaction proceeds efficiently at temperatures between 80°C and 100°C, demonstrating excellent functional group tolerance for substrates bearing halogens, alkyl, and alkoxy groups. By shifting to this catalytic system, manufacturers can achieve high yields, such as the 98% yield observed in model reactions, while significantly streamlining the workflow.
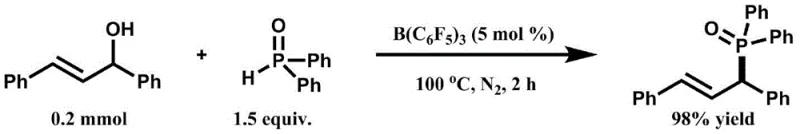
Mechanistic Insights into B(C6F5)3-Catalyzed Dehydrative Coupling
The core of this technological advancement lies in the potent Lewis acidity of the boron center in tris(pentafluorophenyl)borane, which coordinates with the oxygen atom of the allylic alcohol. This coordination weakens the C-O bond, effectively generating an allylic cation-like character or a highly activated complex that is susceptible to nucleophilic attack. The phosphorus-hydrogen compound, acting as the nucleophile, attacks the activated allylic position, leading to the displacement of the hydroxyl group as water. This mechanism avoids the formation of radical intermediates often seen in other coupling strategies, resulting in cleaner reaction profiles and higher stereochemical integrity where applicable. The catalytic cycle is sustained by the regeneration of the borane species, allowing for low catalyst loadings (as low as 5 mol%) to drive the reaction to completion without the accumulation of metal waste.
Impurity control is inherently superior in this system due to the absence of competing side reactions typically induced by strong bases or oxidative conditions. The mild nature of the Lewis acid catalyst ensures that sensitive functional groups on the aromatic rings of the substrates remain intact, preventing degradation or unwanted substitution. For instance, substrates containing bromine or fluorine atoms, which are crucial for subsequent cross-coupling modifications in drug synthesis, are fully compatible with these reaction conditions. This selectivity minimizes the formation of regioisomers and byproducts, simplifying the downstream purification process and ensuring that the final product meets the rigorous purity specifications required for clinical applications.
How to Synthesize Allyl Phosphorus Compounds Efficiently
The synthesis protocol outlined in the patent provides a robust framework for producing these valuable intermediates with minimal operational complexity. The process involves simply mixing the secondary allyl alcohol, the phosphorus source, and the borane catalyst in an inert atmosphere, followed by heating. This straightforward procedure eliminates the need for complex addition protocols or strict anhydrous conditions beyond standard inert gas protection, making it highly accessible for scale-up. Detailed standardized synthesis steps for specific derivatives are provided in the technical guide below to ensure reproducibility and quality control.
- Mix secondary allyl alcohol, phosphine compound, and tris(pentafluorophenyl)borane catalyst in a molar ratio of approximately 1: 1.5:0.05 in a reaction vessel under inert gas.
- Heat the mixture to 80-100°C and stir for 2 to 12 hours to facilitate the dehydrative coupling reaction.
- Purify the crude reaction product using thin layer chromatography with a petroleum ether/ethyl acetate solvent system to isolate the high-purity allyl phosphorus compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers tangible strategic benefits centered around cost efficiency and supply reliability. The elimination of precious metal catalysts removes a major cost driver and mitigates the risk associated with the price volatility of metals like palladium. Additionally, the simplified purification process reduces solvent consumption and processing time, leading to substantial operational expenditure savings. The use of commercially available, inexpensive starting materials further enhances the economic viability of the process, ensuring a stable supply chain that is less susceptible to raw material shortages.
- Cost Reduction in Manufacturing: The replacement of expensive transition metal catalysts and ligands with a reusable Lewis acid system drastically lowers the bill of materials. Furthermore, the atom economy of the reaction is maximized as water is the only byproduct, reducing waste disposal costs and environmental compliance burdens. The ability to run reactions without solvents in certain embodiments also contributes to significant savings in solvent procurement and recovery infrastructure.
- Enhanced Supply Chain Reliability: The reliance on widely available commodity chemicals such as allyl alcohols and phosphine oxides ensures a resilient supply chain不受制于 specialized reagent availability. The robustness of the reaction conditions means that production can be maintained consistently without frequent interruptions due to catalyst deactivation or sensitivity issues. This reliability is crucial for maintaining continuous manufacturing flows for critical pharmaceutical intermediates.
- Scalability and Environmental Compliance: The mild reaction temperatures and lack of hazardous byproducts make this process ideal for large-scale commercial production. The green chemistry attributes, specifically the generation of water as the sole byproduct, align perfectly with increasingly stringent environmental regulations, facilitating easier permitting and reducing the carbon footprint of the manufacturing facility.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on its practical application in industrial settings.
Q: What are the advantages of using B(C6F5)3 over transition metal catalysts for this synthesis?
A: The use of tris(pentafluorophenyl)borane eliminates the need for expensive transition metals like palladium and complex ligands. This significantly reduces raw material costs and simplifies the purification process by avoiding heavy metal residue removal, which is critical for pharmaceutical grade intermediates.
Q: What is the substrate scope for this allyl phosphorus synthesis method?
A: The method demonstrates broad substrate tolerance, successfully reacting with various secondary allyl alcohols containing aryl, heterocyclic, and alkyl groups. It is compatible with electron-withdrawing and electron-donating substituents such as halogens, methyl, and methoxy groups on the aromatic rings.
Q: Is this process scalable for industrial production?
A: Yes, the process is highly scalable due to its mild reaction conditions (80-100°C), simple operation, and the fact that the only byproduct is water. The absence of sensitive reagents and the use of commercially available starting materials make it suitable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Allyl Phosphorus Compounds Supplier
NINGBO INNO PHARMCHEM stands at the forefront of adopting advanced synthetic methodologies to deliver superior chemical solutions to the global market. Our expertise in scaling diverse pathways from 100 kgs to 100 MT/annual commercial production ensures that we can meet the demanding volume requirements of multinational corporations. We leverage cutting-edge technologies like the borane-catalyzed synthesis described here to maintain stringent purity specifications and rigorous QC labs, guaranteeing that every batch of allyl phosphorus compounds meets the highest industry standards for pharmaceutical and agrochemical applications.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through innovative chemistry. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your R&D and commercialization goals, ensuring a seamless partnership from gram-scale development to full-scale production.
