Advanced Chiral Catalysis for Commercial Scale-up of High-Purity Synthetic (S)-Nicotine
The global demand for high-purity synthetic nicotine has surged as the electronic cigarette industry seeks alternatives to tobacco-derived alkaloids that are free from agricultural contaminants and supply volatility. Patent CN113999084A introduces a groundbreaking methodology for the asymmetric synthesis of (S)-(-)-nicotine, addressing critical pain points in stereochemical control and process efficiency. This technology leverages a sophisticated chiral catalytic system to transform myosmine directly into the biologically active (S)-enantiomer, bypassing the inefficient resolution steps typical of older racemic syntheses. For R&D directors and procurement strategists, this represents a paradigm shift towards more sustainable and economically viable manufacturing of this high-value pharmaceutical intermediate. The protocol utilizes formic acid as a benign hydrogen donor in conjunction with specialized ruthenium or palladium catalysts modified with chiral ligands, ensuring exceptional enantioselectivity. By integrating this advanced catalytic approach, manufacturers can achieve product specifications that meet the rigorous standards required for next-generation inhalation products and pharmacological research.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the sourcing of nicotine has been dominated by extraction from tobacco leaves, a process inherently plagued by variability in alkaloid profiles and the presence of undesirable minor alkaloids that are difficult to separate. Alternatively, chemical synthesis routes often produced racemic nicotine, necessitating a subsequent resolution step using chiral resolving agents to isolate the desired (S)-isomer. This classical resolution approach is fundamentally inefficient, theoretically capping the maximum yield at 50% unless dynamic kinetic resolution is employed, which adds significant complexity and cost. Furthermore, traditional reduction methods frequently rely on stoichiometric metal hydrides like sodium borohydride, generating substantial amounts of boron-containing waste that complicates downstream purification and environmental compliance. The reliance on harsh reagents and multi-step purification sequences in conventional methods results in elevated operational expenditures and extended production lead times. These legacy processes struggle to consistently deliver the ultra-high enantiomeric excess required for premium applications without incurring prohibitive losses in material throughput.
The Novel Approach
The innovative strategy disclosed in the patent circumvents these inefficiencies by employing a direct asymmetric transfer hydrogenation of myosmine to generate the chiral pyrrolidine core with high fidelity. This route utilizes myosmine, a readily accessible precursor, and subjects it to a catalytic environment where chirality is induced during the bond-forming event rather than corrected afterwards. The use of formic acid as the reducing agent offers a safer and more atom-economical alternative to high-pressure hydrogen gas or reactive hydride salts. Following the reduction, a streamlined methylation step using paraformaldehyde completes the synthesis, delivering (S)-(-)-nicotine with minimal byproduct formation. This telescoped logic significantly shortens the critical path of manufacturing, allowing for faster batch turnover and reduced inventory holding costs. The ability to tune the stereoselectivity through ligand selection provides a robust control knob for quality assurance teams to maintain strict batch-to-batch consistency.
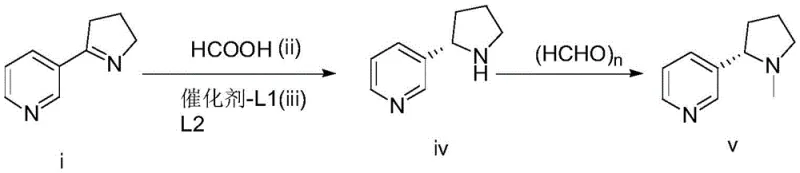
Mechanistic Insights into Ruthenium-Catalyzed Asymmetric Transfer Hydrogenation
The core of this technological advancement lies in the precise orchestration of the metal-ligand complex, which creates a chiral pocket capable of differentiating between the prochiral faces of the imine substrate. The catalyst system, typically comprising a ruthenium(II) chloride dimer paired with chiral diamine or amino-alcohol ligands, facilitates the transfer of a hydride from the formate anion to the substrate. This mechanism proceeds through a six-membered transition state where the proton and hydride are delivered in a concerted manner, locking in the stereochemistry defined by the ligand's backbone. The choice of ligand structure, ranging from TsDPEN derivatives to binaphthyl-based phosphines, critically influences the steric environment around the metal center, thereby dictating the enantiomeric ratio of the product. Understanding this interplay allows process chemists to optimize reaction parameters such as temperature and solvent polarity to maximize the ee value, often exceeding 99% in optimized examples. This level of mechanistic control ensures that the formation of the unwanted (R)-enantiomer is suppressed kinetically, simplifying the purification burden on the manufacturing team.
Impurity control is intrinsically built into this catalytic cycle, as the mild reaction conditions prevent the degradation of the sensitive pyrrolidine ring which can occur under strongly acidic or basic conditions used in older methods. The selectivity of the catalyst minimizes over-reduction or side reactions with the pyridine ring, preserving the structural integrity of the nicotine scaffold. By avoiding stoichiometric metal waste, the process reduces the load on wastewater treatment facilities and lowers the risk of heavy metal contamination in the final API intermediate. The robustness of the catalytic system against moisture and oxygen variations further enhances its suitability for large-scale operations where perfect inertness is challenging to maintain. Consequently, the impurity profile of the resulting nicotine is significantly cleaner, requiring fewer crystallization or distillation steps to reach market-ready specifications. This mechanistic elegance translates directly into higher overall process mass intensity and a greener manufacturing footprint.
How to Synthesize (S)-(-)-Nicotine Efficiently
The implementation of this synthesis requires careful attention to the preparation of the catalytic species and the control of reaction thermodynamics to ensure optimal conversion. Operators must first establish an inert atmosphere to protect the sensitive metal-ligand complex before introducing the myosmine substrate and the formic acid reducing agent. The reaction mixture is then maintained within a specific thermal window, typically between 20°C and 50°C, to balance reaction rate with stereochemical fidelity. Upon completion, the workup involves a pH swing to liberate the free amine followed by extraction into an organic phase, which is then concentrated for the subsequent methylation step.
- Perform asymmetric transfer hydrogenation of myosmine using formic acid and a chiral ruthenium catalyst system to generate (S)-3-pyrrolidinylpyridine.
- Isolate the intermediate amine through pH adjustment, solvent extraction, and vacuum distillation to ensure high optical purity.
- Conduct N-methylation of the purified intermediate using paraformaldehyde under controlled thermal conditions to yield final (S)-(-)-nicotine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this catalytic technology offers profound strategic benefits that extend beyond simple unit cost calculations. The elimination of the resolution step effectively doubles the theoretical yield from the starting material compared to racemic synthesis, drastically improving raw material utilization rates. This efficiency gain means that less feedstock is required to produce the same volume of finished goods, insulating the supply chain from fluctuations in the pricing of key precursors like myosmine. Furthermore, the use of commercially available and stable reagents such as formic acid and paraformaldehyde reduces dependency on specialized or hazardous chemicals that often face logistical bottlenecks. The simplified workflow reduces the number of unit operations, which in turn lowers energy consumption and labor hours per kilogram of output. These operational efficiencies compound to create a significantly more resilient and cost-competitive supply model for high-purity synthetic nicotine.
- Cost Reduction in Manufacturing: The primary driver of cost savings is the avoidance of the 50% yield penalty associated with resolving racemic mixtures, effectively halving the raw material cost basis for the chiral center construction. Additionally, the replacement of expensive stoichiometric reducing agents with catalytic amounts of ruthenium complexes and cheap formic acid substantially lowers the bill of materials. The reduction in waste generation also translates to lower disposal fees and reduced environmental compliance costs, contributing to a leaner overall cost structure. By streamlining the synthesis into fewer steps with higher convergence, the facility can achieve better asset utilization and lower fixed cost allocation per unit. These factors collectively enable a competitive pricing strategy that is robust against market volatility.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals and robust catalytic systems minimizes the risk of supply disruptions caused by the shortage of exotic reagents. The process operates under mild conditions that do not require specialized high-pressure reactors, allowing for production across a wider range of manufacturing sites with standard equipment. This flexibility enhances supply continuity and allows for rapid scaling in response to surging market demand without lengthy capital expenditure cycles. The high purity achieved directly from the reactor reduces the need for complex purification chains that are often vulnerable to bottlenecks. Consequently, lead times for delivering high-purity pharmaceutical intermediates can be significantly compressed, improving service levels to downstream customers.
- Scalability and Environmental Compliance: The atom economy of the transfer hydrogenation reaction aligns perfectly with green chemistry principles, generating water and carbon dioxide as the primary byproducts of the oxidant. This clean profile simplifies the permitting process for new manufacturing lines and reduces the burden on effluent treatment plants. The scalability is further supported by the exothermic nature of the reaction being manageable within standard jacketed vessels, ensuring safe operation at the 100 MT scale. The absence of heavy metal waste streams simplifies the validation process for GMP production, accelerating time-to-market for new product launches. This environmental stewardship not only mitigates regulatory risk but also enhances the brand value for clients seeking sustainable sourcing options.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral synthesis technology in an industrial setting. These insights are derived directly from the experimental data and process descriptions found in the patent literature to ensure accuracy.
Q: What is the primary advantage of this synthetic route over traditional extraction?
A: Unlike tobacco extraction which suffers from variable purity and co-extracted alkaloids, this chiral catalytic method delivers consistent >99% ee values and eliminates heavy metal contaminants often found in plant-derived sources.
Q: How does the catalyst system impact production costs?
A: By utilizing efficient transfer hydrogenation with formic acid, the process avoids expensive high-pressure hydrogenation equipment and eliminates the 50% material loss associated with resolving racemic mixtures.
Q: Is this process scalable for industrial API intermediate manufacturing?
A: Yes, the reaction operates at mild temperatures (20-50°C) and atmospheric pressure, utilizing common organic solvents, which significantly reduces engineering complexity for large-scale commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-(-)-Nicotine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a stable and high-quality supply of chiral intermediates for the rapidly evolving inhalation and pharmaceutical sectors. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of (S)-(-)-nicotine meets the highest international standards. Our infrastructure is designed to handle complex chiral catalysis with the precision required for regulated markets, providing our partners with peace of mind regarding quality and consistency.
We invite forward-thinking organizations to collaborate with us to leverage this advanced synthetic route for their product pipelines. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can optimize your supply chain and reduce total landed costs. Let us be your strategic partner in delivering high-performance chemical solutions.
