Revolutionizing Indole Functionalization: A Scalable One-Pot Route for High-Value Pharmaceutical Intermediates
Revolutionizing Indole Functionalization: A Scalable One-Pot Route for High-Value Pharmaceutical Intermediates
The pharmaceutical and agrochemical industries are constantly seeking more efficient pathways to access complex heterocyclic scaffolds, particularly those based on the privileged indole structure. Patent CN116332912A introduces a groundbreaking synthetic methodology for producing 2-amino-5-thioindole derivatives, a class of compounds with profound biological significance. This innovation addresses critical bottlenecks in current manufacturing by enabling a direct, one-pot transformation of readily available indoline compounds, azoles, and disulfides. By leveraging a robust copper-catalyzed oxidative coupling strategy, this technology bypasses the need for multi-step sequences and harsh electrochemical conditions that have historically plagued this chemical space. For R&D directors and procurement specialists alike, this represents a pivotal shift towards greener, more cost-effective, and scalable production of high-value intermediates used in antiviral, anticancer, and cardiovascular therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of polysubstituted indoles, specifically those bearing thio-groups at the 5-position and amino-functionality at the 2-position, has been a formidable challenge for process chemists. Existing literature predominantly relies on multi-step synthetic routes that require the pre-functionalization of substrates, often involving hazardous reagents and protecting group strategies that drastically reduce overall atom economy. Furthermore, many established methods depend on specialized electrochemical setups or expensive transition metal catalysts that are difficult to remove to the stringent ppm levels required for API manufacturing. These conventional approaches not only inflate the cost of goods sold (COGS) due to low yields and high waste generation but also introduce significant supply chain risks associated with the sourcing of exotic starting materials. The complexity of purification in these legacy methods often results in prolonged lead times and inconsistent batch-to-batch quality, making them unsuitable for the rapid scale-up demands of modern drug development pipelines.
The Novel Approach
In stark contrast, the methodology disclosed in CN116332912A offers a streamlined, direct difunctionalization strategy that constructs the target scaffold in a single operational step. This novel approach utilizes simple indoline precursors which undergo oxidative aromatization and concurrent C-H functionalization in the presence of azoles and disulfides. The reaction is driven by abundant molecular oxygen or mild chemical oxidants, eliminating the need for stoichiometric amounts of toxic heavy metal oxidants. As illustrated in the reaction scheme below, the process achieves high chemical selectivity and excellent functional group tolerance, allowing for the direct installation of diverse aryl-thio and azole motifs without compromising the integrity of sensitive substituents.
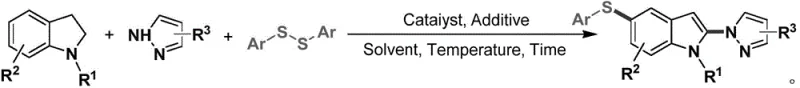
This simplification of the synthetic route translates directly into operational excellence, reducing the number of unit operations and minimizing solvent consumption. The ability to perform this transformation under relatively mild thermal conditions (25-130°C) further enhances the safety profile of the process, making it highly attractive for commercial scale-up in standard stainless steel reactors without the need for specialized corrosion-resistant equipment.
Mechanistic Insights into Copper-Catalyzed Oxidative Coupling
The core of this technological breakthrough lies in the intricate interplay between the copper catalyst and the oxidative environment, which facilitates the simultaneous formation of C-N and C-S bonds. Mechanistically, the reaction likely proceeds through a radical-mediated pathway where the copper species activates the disulfide bond to generate thiyl radicals, which subsequently attack the electron-rich indoline ring. Concurrently, the azole component acts as a nucleophile, attacking the activated intermediate to establish the C-N linkage at the 2-position. The use of molecular oxygen serves a dual purpose: it acts as the terminal oxidant to regenerate the active copper catalyst and drives the dehydrogenative aromatization of the indoline core to the indole system. This elegant cascade ensures high atom economy, as the only by-products are typically water or benign salts, aligning perfectly with the principles of green chemistry.
From an impurity control perspective, this mechanism offers distinct advantages over electrophilic substitution methods which often suffer from regioisomer formation. The specific coordination environment provided by the additives and the inherent reactivity of the indoline substrate guide the functionalization to the desired 2 and 5 positions with high fidelity. For instance, the structural integrity of the product obtained in Example 1, shown below, confirms the precise regioselectivity of the process, yielding a clean product profile that simplifies downstream purification.
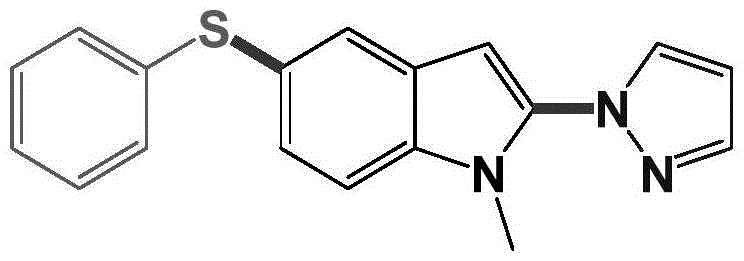
Understanding this mechanistic nuance is crucial for R&D teams aiming to further optimize the process or adapt it to novel substrates. The robustness of the catalytic cycle suggests that minor adjustments in ligand environment or oxidant pressure could unlock even broader substrate scopes, providing a versatile platform for generating diverse chemical libraries for drug discovery programs.
How to Synthesize 2-Amino-5-Thioindole Derivatives Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to the stoichiometry of the three key components and the management of the oxidative atmosphere. The patent details a straightforward protocol where indoline, azole, and disulfide are combined with a copper salt catalyst and a base additive in a polar or non-polar solvent. The reaction mixture is then heated under an oxygen balloon or pressurized oxygen line, allowing the oxidative coupling to proceed to completion. While the general procedure is robust, optimizing parameters such as temperature and reaction time is essential to maximize yield for specific substrate combinations. The detailed standardized synthesis steps see the guide below.
- Charge a reactor with indoline compound, azole compound, disulfide compound, copper catalyst, additive, and solvent.
- Stir the mixture under an oxygen atmosphere at temperatures between 25°C and 130°C for 1 to 24 hours.
- Cool the reaction, remove solvent via rotary evaporation, and purify the crude product using column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible strategic benefits that extend beyond mere technical feasibility. The shift from multi-step, protection-group-heavy syntheses to a direct one-pot coupling fundamentally alters the cost structure of producing these valuable intermediates. By collapsing several synthetic steps into a single operation, manufacturers can significantly reduce labor costs, energy consumption, and solvent waste disposal fees. Moreover, the reliance on commodity chemicals like indolines and disulfides, rather than custom-synthesized building blocks, mitigates supply risk and ensures a stable flow of raw materials even during market fluctuations.
- Cost Reduction in Manufacturing: The elimination of expensive precious metal catalysts in favor of abundant copper salts represents a major driver for cost optimization. Traditional methods often require palladium or rhodium complexes which are not only costly to purchase but also expensive to recover and validate for residual metals in the final API. By utilizing copper iodide or copper chloride, this process drastically lowers the catalyst cost burden. Additionally, the use of molecular oxygen as the oxidant avoids the purchase of hazardous stoichiometric oxidants, further reducing raw material expenses and waste treatment costs associated with heavy metal by-products.
- Enhanced Supply Chain Reliability: The starting materials for this reaction, including various substituted indolines and disulfides, are widely available from global chemical suppliers, ensuring a resilient supply chain. Unlike proprietary intermediates that may be sourced from a single vendor, the commoditized nature of these feedstocks allows procurement teams to negotiate better pricing and secure multiple sources of supply. This diversification reduces the risk of production stoppages due to raw material shortages, ensuring consistent delivery schedules for downstream pharmaceutical clients who rely on just-in-time inventory models.
- Scalability and Environmental Compliance: The simplicity of the workup procedure, which involves basic solvent removal and column chromatography, facilitates easy scale-up from gram to ton quantities. The process generates minimal hazardous waste, aligning with increasingly stringent environmental regulations regarding solvent emissions and heavy metal discharge. This environmental compliance not only avoids potential regulatory fines but also enhances the corporate sustainability profile of the manufacturing entity, a factor that is becoming increasingly important for partnerships with major multinational pharmaceutical companies committed to green supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical application of this technology for industrial partners.
Q: What are the primary advantages of this new synthesis method over traditional routes?
A: This method eliminates the need for pre-prepared substrates and harsh electrochemical conditions, utilizing a simple one-pot copper-catalyzed process with high atom economy and functional group tolerance.
Q: Is the catalyst system cost-effective for large-scale production?
A: Yes, the process employs inexpensive and readily available copper salts (such as copper iodide or copper chloride) and molecular oxygen as a green oxidant, significantly reducing raw material costs compared to precious metal catalysts.
Q: What is the substrate scope for this reaction?
A: The method demonstrates excellent compatibility with various substituents on the indoline, azole, and disulfide components, including alkyl, alkoxy, nitro, halogen, and trifluoromethyl groups, allowing for diverse derivative synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Amino-5-Thioindole Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this copper-catalyzed oxidative coupling technology for the production of next-generation pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to commercial manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 2-amino-5-thioindole derivatives meets the highest quality standards required for clinical and commercial applications.
We invite you to collaborate with us to leverage this innovative synthetic route for your specific drug development projects. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your target molecule, demonstrating how this method can optimize your overall production budget. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you accelerate your path to market with a reliable, cost-effective, and sustainable supply solution.
