Advanced Organocatalytic Synthesis of Benzosultam Quaternary Amino Acid Derivatives for Pharmaceutical Applications
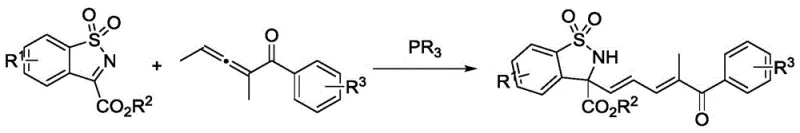
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for complex heterocyclic scaffolds, particularly those containing quaternary stereocenters which are prevalent in bioactive molecules. Patent CN111763181A, published on October 13, 2020, introduces a significant advancement in this domain by detailing a novel synthetic method for preparing benzosultam quaternary carbon amino acid derivatives containing conjugated dienone fragments. This technology leverages a tertiary phosphine organocatalytic system to facilitate a Mannich reaction between trisubstituted allenone compounds and benzosulfonimide compounds. Unlike traditional methods that often rely on harsh conditions or expensive transition metals, this approach operates under remarkably mild parameters, typically at ambient temperatures around 25°C, and achieves high yields without necessitating strict anhydrous or oxygen-free environments. For R&D directors and process chemists, this represents a pivotal shift towards more sustainable and operationally simple methodologies for constructing these valuable pharmacophores, ensuring that the resulting intermediates meet the stringent purity profiles required for downstream drug development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of benzosultam quaternary amino acid derivatives has been fraught with synthetic challenges that hinder efficient large-scale production. Previous literature, such as the work by Tagliabue's group in 2008, relied on solid-liquid phase transfer catalysis (SL-PTC) involving intramolecular nucleophilic displacement of aromatic fluorine atoms, a process that can be limited by substrate specificity and harsh reaction conditions. Furthermore, methodologies developed by Chi's group in 2016 utilized N-heterocyclic carbene (NHC) catalysis to generate radical intermediates from nitrobenzyl bromides, which introduces complexity regarding radical stability and safety concerns associated with nitro compounds. Other approaches, including those by Zhang and Jia, employed transition metal catalysts like copper acetate or nickel perchlorate with chiral ligands to achieve asymmetric alkynylation or cyclization. While effective for specific stereochemical outcomes, these metal-catalyzed routes invariably introduce the risk of heavy metal contamination, necessitating costly and time-consuming purification steps to meet regulatory limits for pharmaceutical ingredients. Additionally, photo-redox catalytic methods reported by Gong require specialized equipment and precise control of light sources, adding another layer of operational complexity and capital expenditure that is often undesirable for standard manufacturing facilities.
The Novel Approach
In stark contrast to these legacy technologies, the method disclosed in CN111763181A utilizes a metal-free organocatalytic strategy that dramatically simplifies the synthetic workflow. By employing a tertiary phosphine organic small molecule, specifically diphenylmethylphosphine, as the catalyst, the reaction activates the trisubstituted allenone towards nucleophilic attack by the benzosulfonimide. This Mannich-type transformation proceeds efficiently in common solvents like dichloromethane at temperatures ranging from 0°C to 40°C, with a preference for 25°C. The elimination of transition metals not only reduces the raw material cost but also removes the burden of metal scavenging during workup. The protocol is robust enough to tolerate a wide array of functional groups, including halogens, alkyls, and alkoxy substituents on the aromatic rings, demonstrating exceptional substrate versatility. This operational simplicity, combined with the ability to achieve high yields without rigorous exclusion of moisture or air, positions this technology as a superior alternative for the commercial manufacture of high-purity pharmaceutical intermediates, directly addressing the pain points of both cost and scalability.
Mechanistic Insights into Phosphine-Catalyzed Mannich Reaction
The core of this technological breakthrough lies in the unique activation mode provided by the tertiary phosphine catalyst. Mechanistically, the phosphine acts as a nucleophile to attack the electron-deficient central carbon of the trisubstituted allenone, generating a reactive zwitterionic intermediate. This species effectively increases the nucleophilicity of the adjacent carbon chain, allowing it to engage in a concerted or stepwise addition to the electrophilic imine carbon of the benzosulfonimide. The presence of the anhydrous sodium sulfate additive plays a crucial role in sequestering trace water that might otherwise hydrolyze sensitive intermediates or deactivate the catalyst, thereby maintaining the integrity of the catalytic cycle. Unlike radical pathways that can lead to unpredictable side reactions and polymerization, this polar ionic mechanism offers a high degree of chemoselectivity, ensuring that the conjugated dienone fragment is preserved intact within the final structure. For process chemists, understanding this mechanism is vital for troubleshooting; for instance, maintaining the optimal catalyst loading of 20 mol% ensures that the equilibrium favors the formation of the desired quaternary center without promoting oligomerization of the allenone substrate.
Impurity control is another critical aspect where this mechanism offers distinct advantages. In metal-catalyzed processes, side products often arise from beta-hydride elimination or homocoupling of organometallic species, leading to complex impurity profiles that are difficult to separate. In this phosphine-catalyzed system, the primary byproducts are typically unreacted starting materials or simple hydrolysis products, which are far easier to remove via standard silica gel column chromatography. The mild reaction temperature of 25°C further suppresses thermal degradation pathways that could compromise the stability of the conjugated dienone moiety. This results in a cleaner crude reaction profile, which translates directly to higher isolated yields and reduced solvent consumption during purification. For quality assurance teams, this means a more consistent impurity spectrum across different batches, facilitating smoother regulatory filings and reducing the risk of batch rejection due to unknown impurities exceeding identification thresholds.
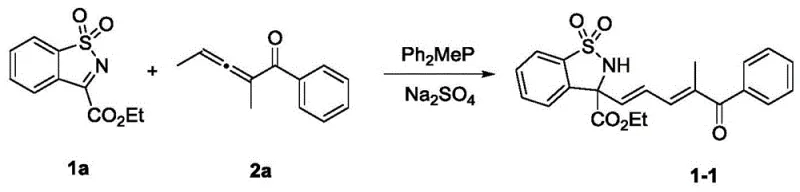
How to Synthesize Benzosultam Derivatives Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific operational parameters to maximize efficiency and yield. The general procedure involves charging a reaction vessel with the benzosulfonimide substrate and anhydrous sodium sulfate, followed by the addition of the solvent, typically dichloromethane. Once the mixture is stirred to ensure homogeneity, the tertiary phosphine catalyst and the trisubstituted allenone are introduced. The reaction is allowed to proceed at ambient temperature with monitoring via thin-layer chromatography (TLC) until the starting benzosulfonimide is fully consumed. Following completion, the reaction mixture is concentrated, and the crude product is purified using standard chromatographic techniques. Detailed standardized synthesis steps for specific derivatives are provided in the guide below to ensure reproducibility and safety during scale-up operations.
- Charge a reaction vessel with benzosulfonimide substrate and anhydrous sodium sulfate additive in a suitable solvent such as dichloromethane.
- Add the tertiary phosphine organic small molecule catalyst, specifically diphenylmethylphosphine, followed by the trisubstituted allenone substrate.
- Stir the mixture at ambient temperature (25°C) monitoring by TLC, then concentrate and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this organocatalytic methodology offers substantial strategic benefits that extend beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the supply chain for raw materials; tertiary phosphines and benzosulfonimides are generally more stable and commercially accessible than the specialized chiral ligands or air-sensitive metal complexes required by competing technologies. This accessibility mitigates the risk of supply disruptions caused by the geopolitical volatility often associated with rare earth or precious metal sourcing. Furthermore, the elimination of transition metals from the process flow significantly reduces the environmental footprint and waste disposal costs, as there is no need for specialized treatment of heavy metal-contaminated effluent. This aligns perfectly with modern green chemistry initiatives and corporate sustainability goals, potentially qualifying the manufacturing process for various environmental incentives.
- Cost Reduction in Manufacturing: The economic impact of switching to this metal-free protocol is profound, primarily driven by the removal of expensive catalytic systems and the associated purification burdens. Transition metal catalysts, especially those involving palladium, rhodium, or specialized copper complexes, represent a significant line item in the bill of materials, and their removal instantly lowers the direct material cost. Moreover, the absence of metal residues negates the need for expensive scavenger resins or additional recrystallization steps designed to lower metal content to ppm levels, which often result in yield loss. The mild reaction conditions also imply lower energy consumption for heating or cooling, contributing to reduced utility costs over the lifecycle of the product. These cumulative savings allow for a more competitive pricing structure for the final pharmaceutical intermediate, enhancing margin potential for downstream drug manufacturers.
- Enhanced Supply Chain Reliability: Reliability in the supply of critical intermediates is paramount for maintaining continuous drug production schedules, and this synthesis method bolsters security of supply through its robustness. The tolerance for non-anhydrous and non-oxygen-free conditions means that the reaction is less susceptible to failure due to minor fluctuations in plant operating conditions or solvent quality. This resilience reduces the frequency of batch failures and reworks, ensuring a steadier output of material. Additionally, the broad substrate scope allows for the use of diverse starting materials; if one specific substituted benzosulfonimide becomes scarce, the process flexibility may allow for alternative analogs or sourcing strategies without requiring a complete re-validation of the synthetic route. This adaptability is a crucial asset for supply chain managers navigating the complexities of global chemical logistics.
- Scalability and Environmental Compliance: Scaling chemical processes from the gram scale to multi-ton production often exposes hidden inefficiencies, but this protocol is inherently designed for scalability. The use of common solvents like dichloromethane and the absence of hazardous reagents simplify the engineering controls required for large reactors. The exothermic profile of the reaction is manageable at 25°C, reducing the risk of thermal runaways that plague many exothermic metal-catalyzed reactions. From an environmental compliance standpoint, the generation of waste is minimized not only by the lack of metal salts but also by the high atom economy of the Mannich addition. This facilitates easier permitting for new manufacturing lines and ensures long-term compliance with increasingly stringent environmental regulations regarding industrial emissions and waste discharge, securing the license to operate for years to come.
Frequently Asked Questions (FAQ)
To further clarify the technical and commercial implications of this patent, we have compiled a set of frequently asked questions based on the detailed experimental data and claims provided in the documentation. These answers address common concerns regarding reaction specificity, catalyst recovery, and product stability, providing a clear picture of what partners can expect when integrating this technology into their portfolio. Understanding these nuances is essential for making informed decisions about process adoption and investment.
Q: What are the advantages of this phosphine-catalyzed method over transition metal catalysis?
A: This method eliminates the need for expensive and potentially toxic transition metals like copper or nickel, simplifying purification and reducing heavy metal residue risks in pharmaceutical intermediates.
Q: Does this synthesis require strict anhydrous or oxygen-free conditions?
A: No, the patent highlights that the reaction proceeds efficiently under mild conditions without the need for rigorous anhydrous or oxygen-free operations, facilitating easier scale-up.
Q: What is the substrate scope for this Mannich reaction?
A: The method demonstrates broad applicability, tolerating various substituents on the benzosulfonimide ring including halogens, alkyl groups, and alkoxy groups, as well as different aryl groups on the allenone.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzosultam Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic methodology described in CN111763181A for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. Our state-of-the-art facilities are equipped to handle organocatalytic processes with precision, maintaining stringent purity specifications and utilizing rigorous QC labs to guarantee that every batch of benzosultam derivatives meets the highest international standards. We understand that consistency is key in the pharmaceutical supply chain, and our commitment to quality assurance ensures that the complex quaternary stereocenters are constructed with fidelity and reliability.
We invite forward-thinking pharmaceutical companies and research institutions to collaborate with us to leverage this advanced synthetic route for their drug development programs. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. We encourage you to contact our technical procurement team today to request specific COA data for our catalog of benzosultam intermediates and to discuss route feasibility assessments for your proprietary targets. Together, we can accelerate the delivery of life-saving medicines to the market by optimizing the chemistry that makes them possible.
