Advanced Synthesis of 6-Hydroxy-2(1H)-Quinolinone for Cardiovascular Drug Manufacturing
Advanced Synthesis of 6-Hydroxy-2(1H)-Quinolinone for Cardiovascular Drug Manufacturing
The pharmaceutical landscape for cardiovascular therapeutics is constantly evolving, driven by the need for more efficient and sustainable manufacturing processes for critical intermediates. Patent CN102816116B introduces a groundbreaking methodology for the synthesis of 6-hydroxy-2(1H)-quinolinone, a pivotal scaffold known for its potent activity as a platelet cAMP specific phosphodiesterase (PDE) inhibitor. This compound is essential in the development of anticoagulants and agents capable of strengthening myocardial contraction while inducing vasodilation. The disclosed technology leverages a unique Gemini surfactant system to facilitate the cyclization of 4-hydroxyaniline and cinnamoyl chloride, offering a robust alternative to legacy synthetic routes that have long plagued the industry with environmental and efficiency challenges.
For R&D directors and procurement specialists alike, the implications of this patent extend far beyond the laboratory bench. The shift towards green chemistry methodologies is no longer just a regulatory preference but a commercial imperative. By replacing hazardous reagents with a recyclable surfactant medium, this process addresses the dual mandates of cost reduction in pharmaceutical intermediate manufacturing and environmental compliance. The ability to produce high-purity 6-hydroxy-2(1H)-quinolinone under mild thermal conditions (50~150°C) without the need for toxic heavy metal catalysts represents a significant leap forward in process chemistry, ensuring a reliable supply chain for downstream drug developers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 6-hydroxy-2(1H)-quinolinone has been fraught with inefficiencies and safety hazards that complicate commercial scale-up. Early methods, such as the electroreduction of 2-nitro-5-hydroxycinnamic acid reported by Gattermann, involve complex setup requirements and poor atom economy. Subsequent approaches by researchers like Richard R. Holmes relied on the oxidation of N-methoxy quinolines followed by chlorination and acidification, a multi-step sequence that inherently lowers overall yield and increases waste generation. Perhaps most critically, the method reported by Kobayashi utilizing MCPBA (3-chloroperoxybenzoic acid) for oxidation achieved a total recovery of only 17%, rendering it economically unviable for large-scale production.
Furthermore, the Friedel-Crafts acylation route described by Manabe and Wang, which utilizes anhydrous aluminum trichloride (AlCl3) in chlorobenzene, presents severe operational risks. The use of stoichiometric Lewis acids generates massive quantities of acidic wastewater upon quenching, creating a substantial burden on waste treatment facilities. Additionally, the requirement for anhydrous conditions and corrosive reagents demands specialized reactor materials and rigorous safety protocols, driving up capital expenditure and operational costs. These conventional pathways often result in products contaminated with metal residues, necessitating expensive purification steps to meet the stringent purity specifications required for active pharmaceutical ingredients (APIs).
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a Gemini surfactant system to create a micro-environment that promotes efficient molecular interaction between 4-hydroxyaniline and cinnamoyl chloride. This method eliminates the need for volatile organic solvents like chlorobenzene and hazardous Lewis acids, replacing them with an aqueous-based surfactant medium that is both safer and more environmentally friendly. The reaction proceeds smoothly at temperatures ranging from 50°C to 150°C, significantly reducing energy consumption compared to high-temperature fusion methods or cryogenic conditions required by other pathways.
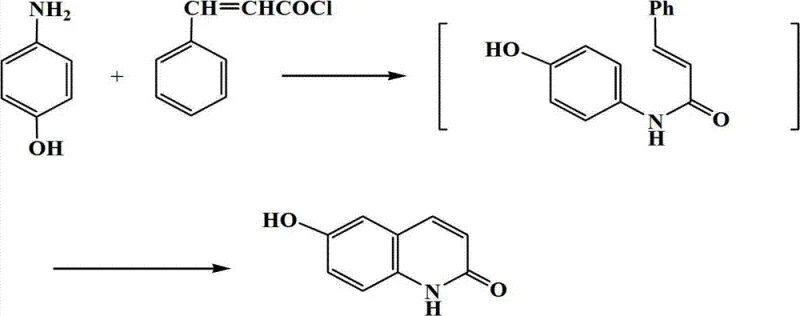
The reaction mechanism involves the initial formation of an amide intermediate followed by an intramolecular cyclization, all facilitated within the hydrophobic cores of the surfactant micelles. This "on-water" effect concentrates the organic reactants, accelerating the reaction rate without the need for aggressive catalysts. Post-reaction workup is remarkably simple, involving a straightforward toluene extraction followed by methanol recrystallization, which yields the target compound as a white solid with a melting point greater than 300°C. This streamlined process not only improves the yield to over 60% in optimized embodiments but also ensures a cleaner impurity profile, directly addressing the quality concerns of pharmaceutical manufacturers.
Mechanistic Insights into Gemini Surfactant-Mediated Cyclization
The core innovation of this technology lies in the specific architecture of the Gemini surfactant used, which features two hydrophilic head groups and two lipophilic tails connected by a spacer. Unlike conventional single-chain surfactants, Gemini surfactants exhibit significantly lower critical micelle concentrations (CMC) and superior solubilization capabilities. In the context of this synthesis, the surfactant molecules self-assemble into micelles that act as nanoreactors. The hydrophobic tails create a non-polar interior that solubilizes the organic reactants (4-hydroxyaniline and cinnamoyl chloride), effectively increasing their local concentration and collision frequency.
This micellar catalysis effect lowers the activation energy for the nucleophilic attack of the amine on the acid chloride, facilitating the formation of the amide bond. Subsequently, the acidic protons on the phenolic ring are activated within the micellar interface, promoting the intramolecular electrophilic substitution that closes the quinolinone ring. The presence of the surfactant also stabilizes the transition states, preventing the formation of polymeric byproducts that often plague free-radical or harsh acid-catalyzed reactions. This controlled environment is crucial for maintaining the structural integrity of the sensitive hydroxy-quinolinone scaffold.
From an impurity control perspective, the mild nature of the surfactant system prevents the degradation of the product or the starting materials. Traditional strong acid conditions can lead to sulfonation of the aromatic rings or hydrolysis of the lactam ring, generating difficult-to-remove impurities. The neutral to slightly basic environment maintained by the surfactant system (especially after neutralization to pH 7-8) ensures that the final product remains stable throughout the reaction duration of 1 to 20 hours. This stability translates directly to a simpler purification process, as the crude product obtained after extraction requires only a single recrystallization step to achieve pharmaceutical-grade purity.
How to Synthesize 6-Hydroxy-2(1H)-Quinolinone Efficiently
The operational simplicity of this synthetic route makes it highly attractive for process chemists aiming to transfer technology from the lab to the pilot plant. The procedure begins with the precise mixing of 4-hydroxyaniline and cinnamoyl chloride in a molar ratio optimized between 1:1 and 1:1.5 to ensure complete conversion of the limiting reagent. The addition of the pre-prepared Gemini surfactant is critical, with a weight ratio relative to the aniline of approximately 1:3 to 1:10, providing the necessary micellar density for effective catalysis.
- Mix 4-hydroxyaniline and cinnamoyl chloride in a molar ratio of 1: 1 to 1:1.5 within a reactor containing the specific Gemini surfactant system.
- Heat the reaction mixture to a temperature range of 50°C to 150°C and maintain stirring for a duration of 1 to 20 hours to ensure complete cyclization.
- Upon completion, cool the mixture, extract the product using toluene, concentrate the organic layer, and recrystallize the residue from methanol to obtain high-purity solids.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this surfactant-mediated synthesis offers tangible benefits that extend well beyond the chemical yield. The elimination of expensive and hazardous reagents like aluminum trichloride and MCPBA fundamentally alters the cost structure of the manufacturing process. By removing the need for specialized corrosion-resistant equipment and complex waste neutralization systems, the capital investment required for production facilities is significantly reduced. Furthermore, the use of cheap, commodity-grade raw materials ensures that the supply chain remains resilient against market fluctuations associated with specialty reagents.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the replacement of stoichiometric Lewis acids with a catalytic amount of recyclable surfactant. Traditional Friedel-Crafts reactions generate equimolar amounts of aluminum waste, the disposal of which is costly and regulated. By shifting to a micellar system, the expense of waste treatment is drastically simplified, and the consumption of auxiliary chemicals is minimized. Additionally, the high selectivity of the reaction reduces the loss of valuable starting materials to side products, improving the overall material efficiency and lowering the cost per kilogram of the final API intermediate.
- Enhanced Supply Chain Reliability: The reliance on widely available feedstocks such as 4-hydroxyaniline and cinnamoyl chloride mitigates the risk of supply disruptions. Unlike specialized catalysts that may have long lead times or single-source dependencies, these bulk chemicals are produced globally by numerous suppliers. The robustness of the reaction conditions, which tolerate a broad temperature range and do not require strict anhydrous environments, further enhances operational reliability. This flexibility allows manufacturers to maintain continuous production schedules even under varying utility conditions, ensuring consistent delivery to downstream pharmaceutical clients.
- Scalability and Environmental Compliance: Scaling this process from gram to ton scale is straightforward due to the absence of exothermic hazards associated with strong Lewis acid additions. The reaction can be safely conducted in standard stainless steel reactors without the need for glass-lined vessels typically required for corrosive acid handling. From an environmental standpoint, the process aligns with green chemistry principles by reducing solvent usage and eliminating heavy metal discharge. This compliance facilitates easier regulatory approval and reduces the carbon footprint of the manufacturing site, a key metric for modern sustainable supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this surfactant-based synthesis. Understanding these details is crucial for evaluating the feasibility of integrating this technology into existing production lines. The answers are derived directly from the experimental data and mechanistic insights provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers.
Q: What are the primary advantages of using Gemini surfactants over traditional Lewis acids like AlCl3?
A: Traditional methods often rely on stoichiometric amounts of aluminum trichloride (AlCl3) or hazardous oxidants like MCPBA, generating significant acidic waste and requiring complex quenching procedures. The Gemini surfactant system acts as a reusable, environmentally benign medium that eliminates heavy metal contamination, simplifies downstream purification, and drastically reduces the environmental footprint of the manufacturing process.
Q: How does this synthetic route impact the purity profile of the final quinolinone intermediate?
A: The mild reaction conditions provided by the micellar environment minimize side reactions such as polymerization or over-chlorination, which are common in harsh Friedel-Crafts conditions. The subsequent recrystallization from methanol effectively removes unreacted starting materials and surfactant residues, yielding a product with a melting point exceeding 300°C and a clean spectral profile suitable for sensitive pharmaceutical applications.
Q: Is this process suitable for large-scale industrial production of cardiovascular intermediates?
A: Yes, the process is explicitly designed for industrial scalability. It utilizes cheap, commercially available raw materials like 4-hydroxyaniline and cinnamoyl chloride, avoids expensive catalysts, and employs standard unit operations like liquid-liquid extraction and recrystallization, making it highly adaptable for multi-ton production campaigns without requiring specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6-Hydroxy-2(1H)-Quinolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving cardiovascular medications. Our team of expert process chemists has extensively analyzed the surfactant-mediated pathway described in CN102816116B and possesses the technical capability to optimize this route for commercial production. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 6-hydroxy-2(1H)-quinolinone meets the highest industry standards.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential partners to contact us to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions about your supply chain strategy. Let us help you navigate the complexities of fine chemical manufacturing with a solution that balances performance, cost, and sustainability.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
