Revolutionizing Palladium Catalysis with Advanced Triarylmonophosphine Ligands for Commercial Scale Production
The landscape of transition metal catalysis is undergoing a significant transformation driven by the need for more robust and efficient ligand systems in the synthesis of complex organic molecules. Patent CN110240616B introduces a groundbreaking class of m-triarylmonophosphine ligands that address critical limitations found in conventional biaryl phosphine systems. This innovation is particularly relevant for the production of high-value pharmaceutical intermediates and fine chemicals where catalytic efficiency directly impacts process economics. The core discovery lies in the structural modification of the phosphine backbone to prevent detrimental conformational changes during the catalytic cycle. By ensuring that the lone pair of electrons on the phosphorus atom is consistently oriented towards an aryl ring, these new ligands maintain the palladium center in its most active configuration. This structural integrity translates to enhanced turnover numbers and broader substrate scope, offering a compelling value proposition for manufacturers seeking to optimize their cross-coupling processes.
For research and development teams, the implications of this technology extend beyond simple yield improvements. The ability to utilize less reactive substrates, such as chlorinated aromatics, without sacrificing reaction rates allows for the use of cheaper starting materials. This shift reduces the overall raw material cost burden while simplifying the purification workflow due to fewer side reactions. Furthermore, the thermal stability of these ligands under inert atmospheres permits reactions at elevated temperatures, expanding the operational window for difficult transformations. As the industry moves towards more sustainable and atom-economical processes, the adoption of such high-performance ligands becomes a strategic necessity for maintaining competitiveness in the global supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional biaryl phosphine ligands, while revolutionary in their time, suffer from an inherent structural flaw related to conformational flexibility. In standard biaryl systems, the bond connecting the phosphine-bearing ring to the adjacent aryl ring can rotate freely. This rotation can lead to a conformation where the palladium center faces away from the adjacent aryl ring, significantly diminishing the beneficial electronic interactions that stabilize the active catalytic species. 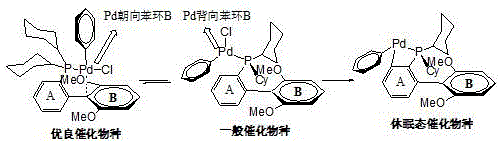 As illustrated in the structural analysis, this misalignment can result in the formation of dormant catalytic species or even the formation of stable Pd-C bonds that remove the metal from the catalytic cycle entirely. Studies have shown that in actual catalytic processes, the population of these inactive conformers can be substantial, leading to inconsistent reaction rates and the need for higher catalyst loadings to achieve acceptable conversion. This inefficiency not only drives up the cost of precious metal usage but also complicates the removal of residual metal from the final product, a critical quality parameter in pharmaceutical manufacturing.
As illustrated in the structural analysis, this misalignment can result in the formation of dormant catalytic species or even the formation of stable Pd-C bonds that remove the metal from the catalytic cycle entirely. Studies have shown that in actual catalytic processes, the population of these inactive conformers can be substantial, leading to inconsistent reaction rates and the need for higher catalyst loadings to achieve acceptable conversion. This inefficiency not only drives up the cost of precious metal usage but also complicates the removal of residual metal from the final product, a critical quality parameter in pharmaceutical manufacturing.
The Novel Approach
The novel m-triarylmonophosphine ligands described in the patent effectively lock the geometry of the ligand to prevent this deactivation pathway. By introducing a third aryl group in the meta-position relative to the phosphorus atom, the steric environment is engineered such that the lone pair of electrons on the phosphorus is always directed towards an available aryl ring. 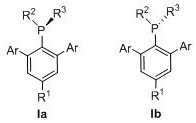 This structural constraint ensures that the palladium center remains coordinated in the optimal orientation for oxidative addition and reductive elimination steps. The result is a catalyst system that exhibits superior stability and activity compared to its biaryl counterparts. Specific embodiments such as XTPhos and ZTPhos have demonstrated remarkable performance in benchmark reactions, outperforming established ligands in both speed and yield. This design philosophy represents a paradigm shift from merely tuning electronic properties to fundamentally engineering the steric landscape to enforce catalytic competence.
This structural constraint ensures that the palladium center remains coordinated in the optimal orientation for oxidative addition and reductive elimination steps. The result is a catalyst system that exhibits superior stability and activity compared to its biaryl counterparts. Specific embodiments such as XTPhos and ZTPhos have demonstrated remarkable performance in benchmark reactions, outperforming established ligands in both speed and yield. This design philosophy represents a paradigm shift from merely tuning electronic properties to fundamentally engineering the steric landscape to enforce catalytic competence.
Mechanistic Insights into Triarylmonophosphine-Palladium Catalysis
The enhanced performance of these ligands can be attributed to a synergistic combination of electronic donation and steric protection. The triaryl backbone provides a rich electron density to the metal center, facilitating the oxidative addition of aryl halides, which is often the rate-determining step in cross-coupling reactions. Simultaneously, the bulky substituents on the peripheral aryl rings create a protective pocket around the palladium. This pocket prevents the aggregation of palladium species into inactive nanoparticles, a common cause of catalyst death in homogeneous catalysis. The patent details various palladium complexes formed with these ligands, showcasing their stability and readiness for catalytic turnover. 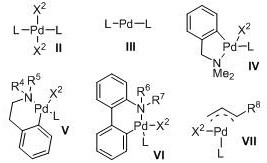 These pre-formed complexes or in-situ generated systems maintain high activity over extended periods, allowing for lower catalyst loadings without compromising reaction completion. The mechanistic robustness ensures that impurities typically associated with catalyst decomposition are minimized, leading to cleaner reaction profiles and simplified downstream processing.
These pre-formed complexes or in-situ generated systems maintain high activity over extended periods, allowing for lower catalyst loadings without compromising reaction completion. The mechanistic robustness ensures that impurities typically associated with catalyst decomposition are minimized, leading to cleaner reaction profiles and simplified downstream processing.
Furthermore, the versatility of the ligand structure allows for fine-tuning of the steric and electronic parameters to match specific substrate requirements. By varying the substituents on the aryl rings, such as introducing methoxy or isopropyl groups, chemists can modulate the cone angle and electron richness of the phosphine. This tunability is crucial for adapting the catalyst to diverse chemical environments, from electron-deficient heterocycles to sterically hindered biaryls. The ability to customize the ligand environment provides a powerful tool for process chemists to overcome specific bottlenecks in synthetic routes, ensuring that the catalytic system remains effective across a wide range of molecular architectures.
How to Synthesize Triarylmonophosphine Ligands Efficiently
The synthesis of these advanced ligands is designed with scalability and operational simplicity in mind. The patent outlines a robust methodology that leverages readily available starting materials such as m-dichlorobenzene and various aryl bromides. The process typically involves the generation of a Grignard reagent followed by a lithiation step to construct the triaryl backbone. Subsequent reaction with chlorophosphine reagents installs the phosphorus functionality in a controlled manner. This approach avoids the need for exotic reagents or extreme conditions, making it accessible for standard chemical manufacturing facilities. The detailed standardized synthesis steps are provided in the guide below to facilitate immediate implementation.
- Prepare Grignard reagent from aryl bromide and magnesium turnings in THF under inert atmosphere.
- React m-dichlorobenzene with butyllithium at low temperature, then add the Grignard reagent and reflux.
- Add chlorophosphine reagent to the lithiated intermediate, warm to room temperature, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this ligand technology offers substantial strategic benefits. The primary driver for cost optimization lies in the efficiency of the catalytic system itself. By enabling reactions to proceed with higher turnover frequencies and lower catalyst loadings, the consumption of expensive palladium metal is significantly reduced. This reduction directly lowers the bill of materials for the final active pharmaceutical ingredient or intermediate. Moreover, the improved selectivity minimizes the formation of by-products, which reduces the burden on purification units and decreases solvent consumption. These factors collectively contribute to a leaner and more cost-effective manufacturing process without the need for capital-intensive equipment upgrades.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the drastic improvement in catalytic efficiency. Traditional methods often require high loadings of palladium to compensate for catalyst deactivation, leading to significant metal costs and expensive scavenging processes to meet regulatory limits. The new triarylmonophosphine ligands maintain high activity throughout the reaction, allowing for a substantial decrease in the amount of palladium required per batch. Additionally, the ability to use cheaper chlorinated substrates instead of more expensive brominated or iodinated analogues further drives down raw material expenses. This dual saving mechanism on both catalyst and substrate creates a compelling business case for switching to this new technology.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of commodity-grade starting materials for the ligand synthesis. Unlike specialized ligands that rely on complex multi-step syntheses with scarce precursors, these m-triarylphosphines are derived from bulk chemicals like m-dichlorobenzene and simple aryl halides. This ensures a stable and continuous supply of the ligand itself, mitigating the risk of production delays due to raw material shortages. Furthermore, the robustness of the catalytic process reduces the likelihood of batch failures, ensuring consistent output volumes that are critical for meeting delivery commitments to downstream customers.
- Scalability and Environmental Compliance: The synthesis protocol is inherently scalable, utilizing standard unit operations such as Grignard formation and column chromatography which are well-understood in industrial settings. The one-pot nature of certain variations reduces the number of isolation steps, thereby minimizing waste generation and solvent usage. This aligns with increasingly stringent environmental regulations and corporate sustainability goals. The high efficiency of the catalytic reaction also means less energy is consumed per unit of product, as reactions can often be completed in shorter times or at milder temperatures. This environmental footprint reduction enhances the overall sustainability profile of the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of these ligands in industrial processes. The answers are derived directly from the experimental data and technical specifications provided in the patent documentation. Understanding these details is essential for evaluating the feasibility of integrating this technology into existing production lines.
Q: What is the primary advantage of m-triarylmonophosphine ligands over traditional biaryl phosphines?
A: The m-triaryl structure prevents conformational rotation that deactivates traditional biaryl phosphines, ensuring the palladium center remains in the active orientation for higher catalytic efficiency.
Q: Are these ligands suitable for large-scale pharmaceutical manufacturing?
A: Yes, the synthesis utilizes cheap starting materials like m-dichlorobenzene and supports one-pot procedures, making it highly scalable and cost-effective for industrial applications.
Q: What types of coupling reactions benefit most from XTPhos and ZTPhos ligands?
A: These ligands demonstrate superior performance in Suzuki-Miyaura couplings and Buchwald-Hartwig aminations, particularly with challenging substrates like chlorinated aromatic hydrocarbons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Triarylmonophosphine Ligands Supplier
NINGBO INNO PHARMCHEM stands at the forefront of delivering high-performance catalytic solutions to the global chemical industry. Our expertise in scaling diverse pathways from 100 kgs to 100 MT/annual commercial production ensures that we can meet your volume requirements with consistency and precision. We understand that the transition to new catalytic technologies requires rigorous validation, which is why our stringent purity specifications and rigorous QC labs are dedicated to providing you with materials that perform exactly as expected in your process. Our commitment to quality ensures that every batch of ligand meets the high standards necessary for sensitive pharmaceutical applications.
We invite you to engage with our technical procurement team to discuss how these advanced ligands can optimize your specific synthetic routes. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits for your operation. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your project needs. Let us partner with you to drive efficiency and innovation in your manufacturing processes.
