Advanced Palladium-Catalyzed Synthesis of Chiral Tetra-Substituted Allenic Acids for Commercial Scale-Up
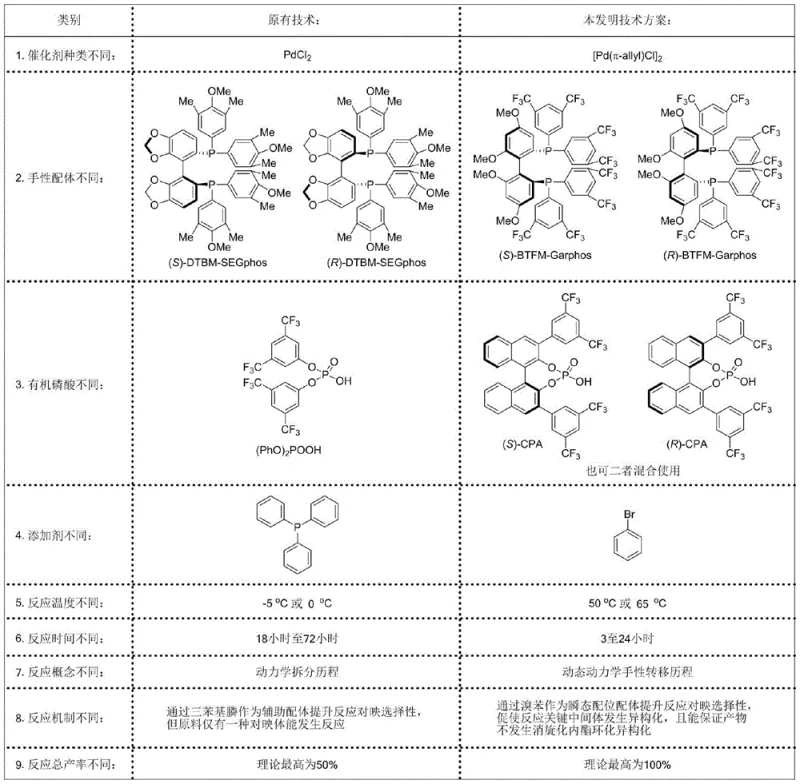
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to construct complex chiral architectures, particularly those containing axial chirality which serves as a privileged scaffold in modern drug design. Patent CN114835541A introduces a groundbreaking preparation method for chiral tetra-substituted allenoic acid compounds based on a sophisticated palladium catalytic system. This technology represents a significant leap forward by enabling the direct construction of highly optically active allenoic acids containing axial chirality in a single step from readily available tertiary propargyl alcohols, carbon monoxide, and water. The process operates under mild reaction conditions and utilizes a synergistic catalyst system comprising a palladium source, a chiral bisphosphine ligand, an organic phosphoric acid, and a specific organic additive. This innovation addresses long-standing challenges in asymmetric synthesis, offering a robust solution for generating valuable intermediates used in the construction of gamma-butyrolactones and other complex chiral molecules.
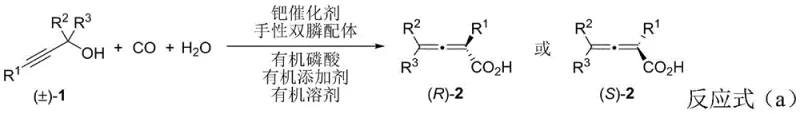
The development of this methodology marks a pivotal shift from traditional approaches that often suffer from inherent inefficiencies. Conventional methods for synthesizing chiral allenoic acids typically rely on the resolution of racemic mixtures or the hydrolysis of chiral esters, processes that are fundamentally limited by a maximum theoretical yield of 50%. This limitation not only wastes half of the valuable starting material but also necessitates additional separation steps that increase cost and environmental burden. Furthermore, existing catalytic methods often struggle with narrow substrate scopes and poor functional group tolerance, making them unsuitable for late-stage functionalization of complex drug candidates. The novel approach detailed in this patent overcomes these barriers by employing a dynamic kinetic chirality transfer strategy. By utilizing a specific combination of ligands and additives, the reaction converts both enantiomers of the racemic starting material into a single chiral product, effectively doubling the potential output compared to kinetic resolution protocols.
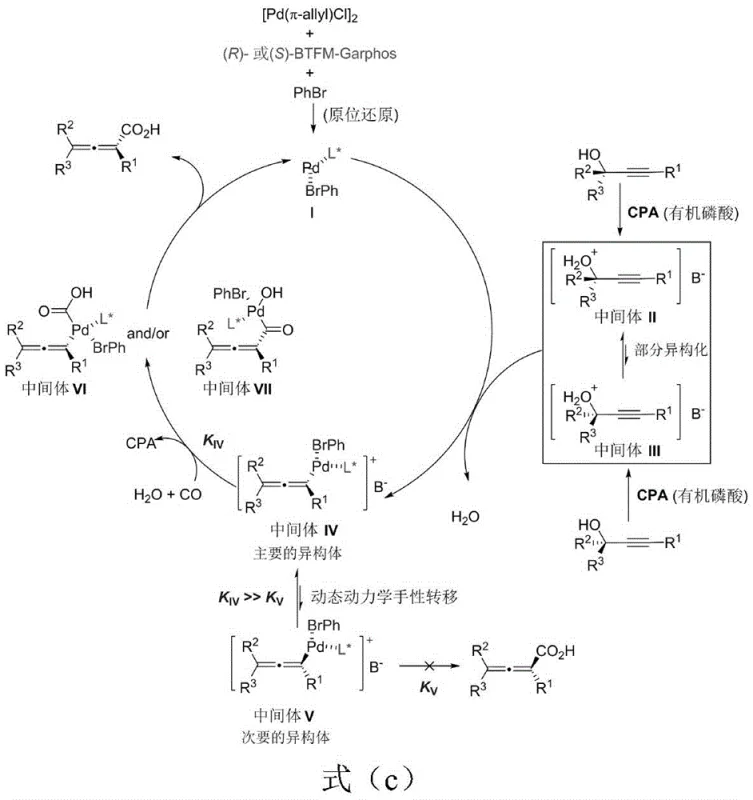
Understanding the mechanistic underpinnings of this transformation is crucial for appreciating its technical superiority and reproducibility. The reaction proceeds through a carefully orchestrated catalytic cycle where the palladium catalyst, activated by the chiral ligand and bromobenzene additive, facilitates an SN2' oxidative addition with the propargyl alcohol substrate. This generates a pair of diastereomeric allenyl palladium intermediates. A key feature of this mechanism is the dynamic equilibrium between these intermediates, driven by the unique properties of the organic additive. The additive acts as a transient ligand that significantly accelerates the isomerization of the slower-reacting intermediate into the faster-reacting species, ensuring that the entire substrate pool is funneled towards the desired product. Subsequent carbonylation and nucleophilic attack by water lead to the formation of the chiral allenoic acid, while the catalyst is regenerated to continue the cycle. This intricate balance prevents common side reactions such as elimination to enynes or intramolecular lactonization, which typically plague similar transition metal-catalyzed processes.
For research and development teams looking to implement this synthesis, the protocol is designed for operational simplicity while maintaining rigorous control over stereochemistry. The procedure involves charging a reaction vessel with the catalyst components under an inert atmosphere, followed by the addition of the substrate, solvent, and reagents. The use of common solvents like toluene and standard equipment such as Schlenk tubes makes this method highly adaptable to existing laboratory infrastructure. Critical to success is the precise control of reaction parameters, including temperature and the stoichiometric ratio of the bromobenzene additive, which must be present in significant excess to ensure optimal enantioselectivity. The following guide outlines the standardized steps derived from the patent examples to ensure consistent results.
From a commercial and supply chain perspective, this technology offers compelling advantages that extend beyond mere academic interest. The ability to achieve theoretical yields approaching 100% fundamentally alters the cost structure of manufacturing these high-value intermediates. By eliminating the 50% yield ceiling associated with kinetic resolution, manufacturers can significantly reduce the consumption of raw materials per kilogram of final product, leading to substantial cost savings in large-scale production. Additionally, the use of readily available tertiary propargyl alcohols as starting materials ensures a stable and reliable supply chain, reducing dependency on exotic or hard-to-source precursors. The mild reaction conditions, typically ranging from 50 to 65 degrees Celsius, further contribute to energy efficiency and safety, lowering the operational expenditure associated with heating and cooling requirements. These factors combined make the process highly attractive for cost reduction in pharmaceutical intermediate manufacturing.
Scalability is another critical dimension where this method excels, addressing the needs of supply chain heads focused on continuity and volume. The reaction demonstrates excellent functional group compatibility, tolerating halogens, esters, silyl groups, and even complex heterocyclic systems without the need for extensive protecting group strategies. This robustness simplifies the purification process, often requiring only standard silica gel chromatography to isolate the product with high purity. The suppression of side reactions such as lactonization ensures that the impurity profile remains clean, facilitating easier regulatory approval and quality control. Furthermore, the process avoids the use of expensive transition metal scavengers often required to remove residual palladium, as the catalyst loading is optimized for efficiency. These attributes collectively enhance supply chain reliability by minimizing batch failures and ensuring consistent delivery of high-purity chiral allenoic acids.
Frequently asked questions regarding this technology often center on its practical application and scope. Stakeholders are particularly interested in how the dynamic kinetic resolution compares to established methods in terms of enantiomeric excess and substrate versatility. The data indicates that enantioselectivity consistently ranges from 77% to 96% ee across a diverse array of substrates, demonstrating the broad applicability of the catalyst system. Questions also arise regarding the specific role of the bromobenzene additive, which is distinct from its traditional use as an electrophile in coupling reactions. In this context, it serves a purely mechanistic function to modulate the catalyst's behavior, a nuance that underscores the sophistication of the ligand design. Addressing these technical queries helps build confidence in the method's viability for industrial adoption.
- Prepare the reaction vessel under inert atmosphere and add the palladium catalyst, chiral bisphosphine ligand, and organic phosphoric acid.
- Introduce the tertiary propargyl alcohol substrate, water, organic solvent, and the critical organic additive such as bromobenzene.
- Pressurize with carbon monoxide and heat the mixture to facilitate the dynamic kinetic chirality transfer reaction.
Frequently Asked Questions (FAQ)
Q: What is the theoretical yield advantage of this new method?
A: Unlike traditional kinetic resolution methods limited to a maximum 50% yield, this dynamic kinetic chirality transfer process allows for a theoretical yield of up to 100%.
Q: Why is bromobenzene used as an additive?
A: Bromobenzene acts as a transient coordination ligand that accelerates the isomerization of slow-reacting intermediates and prevents product racemization or lactonization.
Q: Is this process suitable for complex drug molecules?
A: Yes, the method demonstrates excellent functional group tolerance and compatibility with substrates containing complex natural product or drug molecular backbones.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Tetra-Substituted Allenic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the one described in Patent CN114835541A for accelerating drug discovery and development. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes can be seamlessly translated into robust manufacturing operations. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our infrastructure is designed to handle complex chiral syntheses with the precision and reliability required by global pharmaceutical partners.
We invite you to explore how our expertise can optimize your supply chain and reduce time-to-market for your critical projects. By leveraging our technical proficiency in asymmetric catalysis and process optimization, we can provide a Customized Cost-Saving Analysis tailored to your specific molecular targets. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments for your upcoming campaigns. Let us partner with you to turn complex chemical challenges into commercial successes.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
