Advanced Catalyst-Free Synthesis of Multi-Substituted 1-Naphthols for Commercial Scale-Up
Advanced Catalyst-Free Synthesis of Multi-Substituted 1-Naphthols for Commercial Scale-Up
The chemical industry constantly seeks robust methodologies for constructing complex aromatic scaffolds, particularly naphthol derivatives which serve as critical building blocks in the synthesis of dyes, agrochemicals, and pharmaceutical active ingredients. A significant breakthrough in this domain is documented in patent CN101638355B, which discloses a highly efficient method for synthesizing multi-substituted 1-naphthols through a zinc-mediated cascade reaction. This technology represents a paradigm shift from traditional catalytic cycles, utilizing a thermal intramolecular cyclization strategy that operates effectively at temperatures between 140°C and 160°C. By leveraging the unique reactivity of 2,3-allenoates with organozinc reagents in high-boiling solvents like xylene or 1,3,5-trimethylbenzene, this process achieves remarkable structural diversity without the need for exogenous catalysts. For R&D directors and process chemists, this patent offers a compelling alternative to legacy routes, promising streamlined operations and enhanced control over impurity profiles in the production of high-value fine chemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of polysubstituted naphthol cores has been fraught with synthetic challenges that hinder scalable manufacturing and cost-effective production. Traditional approaches, such as the silica-based benzyne Diels-Alder reaction with substituted furans reported in literature, often suffer from severe limitations regarding substrate availability and regioselectivity. These conventional pathways typically require harsh conditions or specialized precursors that are difficult to prepare on a large scale, leading to bottlenecks in the supply chain for key intermediates. Furthermore, many existing methods restrict the substitution pattern, often confining functional groups to specific positions like the 4-position of the naphthol ring, which limits the chemical space accessible for drug discovery and material science applications. The reliance on transition metal catalysts in some older protocols also introduces significant downstream processing burdens, necessitating rigorous purification steps to meet stringent residual metal specifications required by regulatory bodies for pharmaceutical ingredients.
The Novel Approach
In stark contrast to these legacy techniques, the methodology outlined in CN101638355B introduces a streamlined, catalyst-free protocol that fundamentally simplifies the synthetic architecture. By employing a tandem sequence involving 1,4-addition followed by intramolecular Friedel-Crafts cyclization, this novel approach enables the simultaneous introduction of three distinct substituents onto the naphthol framework in a single operational step. The use of readily accessible 2,3-allenoate esters and versatile organozinc reagents allows for unprecedented flexibility in molecular design, permitting the installation of aryl, alkyl, and halogen groups at the 2, 3, and 4 positions with high precision. This elimination of external catalysts not only reduces the raw material costs associated with expensive metal complexes but also drastically simplifies the work-up procedure, as there is no need for complex metal scavenging processes. Consequently, this innovation provides a reliable pharmaceutical intermediates supplier with a powerful tool to enhance production efficiency and reduce the overall environmental footprint of naphthol manufacturing.
Mechanistic Insights into Zinc-Mediated Cascade Cyclization
The core of this technological advancement lies in the elegant mechanistic pathway where the organozinc species acts as a dual-function reagent, initiating the sequence and driving the cyclization without auxiliary catalysis. The reaction commences with the nucleophilic attack of the organozinc reagent on the 2,3-allenoate substrate, specifically undergoing a 1,4-addition to generate a reactive 1,3-conjugated dienyl zinc oxysalt intermediate. This transient species is pivotal, as it sets the stage for the subsequent ring-closing event by positioning the nucleophilic carbon in proximity to the electrophilic aromatic system. Upon heating in a high-boiling solvent such as xylene or mesitylene, the system gains sufficient thermal energy to overcome the activation barrier for the intramolecular Friedel-Crafts reaction. This cyclization step constructs the second aromatic ring of the naphthol core, forming a stable 1-naphthol zinc oxysalt complex that retains the structural integrity of the newly formed bonds.
Following the cyclization, the final step involves a protolysis reaction, typically achieved during the aqueous quenching phase with saturated ammonium chloride, which releases the free multi-substituted 1-naphthol product. This mechanism ensures high atom economy and excellent regiocontrol, as the substituents from the starting allenoate are faithfully transferred to the 2 and 4 positions, while the organic group from the zinc reagent is installed at the 3-position. The absence of competing side reactions, such as polymerization or non-selective addition, contributes to the high purity of the crude product, often ranging from 68% to 93% yield as demonstrated in the patent examples. Understanding this mechanistic flow is crucial for cost reduction in pharmaceutical intermediates manufacturing, as it allows process engineers to optimize reaction parameters like temperature and stoichiometry to maximize throughput while minimizing waste generation.
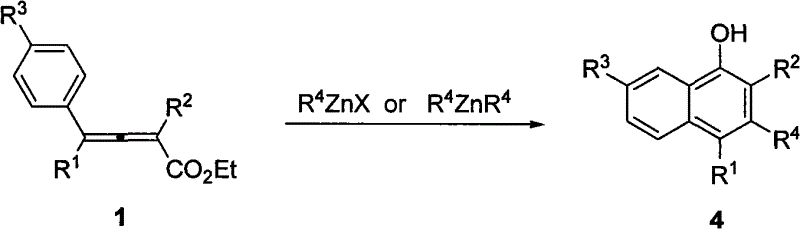
How to Synthesize Multi-Substituted 1-Naphthols Efficiently
Executing this synthesis requires careful attention to solvent selection and thermal management to ensure the cascade reaction proceeds to completion without degradation of the sensitive allenoate starting material. The protocol dictates the use of anhydrous conditions under nitrogen protection to prevent premature hydrolysis of the organozinc reagent, which is typically supplied as a solution in hexane or tetrahydrofuran. A critical operational step involves the removal of low-boiling solvents like THF prior to the addition of the substrate, ensuring that the reaction mixture reaches the necessary 140°C to 160°C range efficiently. Once the high-boiling solvent system is established, the slow addition of the 2,3-allenoate solution allows for controlled exotherm management and optimal mixing, which are vital for maintaining consistent product quality across batches. The detailed standardized synthesis steps for implementing this robust protocol are provided in the guide below.
- Prepare the reaction vessel under nitrogen protection and add the organozinc reagent solution along with a high-boiling solvent such as xylene or 1,3,5-trimethylbenzene.
- Distill off low-boiling solvents like THF under a nitrogen stream, then heat the mixture to 140°C or 160°C before adding the 2,3-allenoate substrate solution.
- Maintain the reaction temperature with agitation until completion, then cool the mixture, quench with saturated ammonium chloride, and purify the resulting 1-naphthol via extraction and column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this catalyst-free zinc-mediated route offers substantial strategic benefits that extend beyond mere chemical yield. The primary advantage lies in the drastic simplification of the supply chain for raw materials, as the process utilizes commodity chemicals like xylene and commercially available organozinc reagents rather than bespoke, high-cost catalysts. This shift significantly mitigates the risk of supply disruptions caused by the scarcity of precious metals or specialized ligands, thereby enhancing the overall reliability of the manufacturing schedule. Furthermore, the operational simplicity of the method, which avoids complex filtration steps for catalyst removal, translates directly into reduced processing time and lower utility consumption per kilogram of product produced. These factors collectively contribute to a more resilient and cost-effective production model that can better withstand market volatility and raw material price fluctuations.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts from the synthetic route removes a major cost center associated with both the purchase of expensive reagents and the downstream purification required to meet regulatory limits. Without the need for metal scavengers or extensive chromatography solely for metal removal, the production process becomes significantly leaner, leading to substantial cost savings in terms of consumables and labor. Additionally, the high yields reported in the patent examples imply that less starting material is wasted, further optimizing the cost of goods sold and improving the overall margin profile for the final API intermediate.
- Enhanced Supply Chain Reliability: By relying on robust, thermally driven chemistry rather than sensitive catalytic systems, the manufacturing process becomes less susceptible to variations in reagent quality or minor deviations in reaction conditions. The use of stable, high-boiling solvents like 1,3,5-trimethylbenzene ensures that the reaction can be run safely and consistently at scale, reducing the likelihood of batch failures that could disrupt delivery timelines. This inherent stability makes the process ideal for long-term contracts where consistent supply of high-purity pharmaceutical intermediates is critical for the customer's own production planning.
- Scalability and Environmental Compliance: The straightforward work-up procedure, involving simple extraction and washing steps, facilitates easy scale-up from laboratory to commercial production volumes without the need for specialized equipment. Moreover, the reduction in chemical waste generated by avoiding catalyst residues aligns with modern green chemistry principles, helping manufacturers meet increasingly stringent environmental regulations. This eco-friendly profile not only reduces disposal costs but also enhances the corporate sustainability credentials of the supply chain, a factor of growing importance to global pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this zinc-mediated cyclization technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the method's capabilities and limitations. By clarifying these points, we aim to assist technical teams in evaluating the feasibility of integrating this route into their existing manufacturing portfolios for complex aromatic compounds.
Q: What are the primary advantages of this zinc-mediated cyclization over traditional Diels-Alder approaches?
A: Unlike traditional methods that often rely on difficult-to-prepare benzyne precursors and limit substitution patterns, this zinc-mediated approach utilizes readily available 2,3-allenoates and organozinc reagents. It allows for the simultaneous introduction of three distinct substituents at the 2, 3, and 4 positions of the naphthol ring without requiring external catalysts, significantly simplifying the purification process.
Q: Does this synthesis require expensive transition metal catalysts?
A: No, a key feature of this patented methodology is that it proceeds without the need for additional transition metal catalysts. The organozinc reagent acts both as the nucleophile for the initial addition and facilitates the subsequent intramolecular Friedel-Crafts cyclization, thereby eliminating the costs and regulatory burdens associated with heavy metal removal in pharmaceutical manufacturing.
Q: What types of substituents can be introduced using this method?
A: This method offers exceptional versatility, allowing for the introduction of aryl, alkyl, halogen, and hydrogen groups at various positions. Specifically, the R4 group from the zinc reagent is introduced at the 3-position, while the substituents from the 2,3-allenoate precursor are incorporated at the 2 and 4 positions, enabling the synthesis of a wide library of polysubstituted derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Multi-Substituted 1-Naphthol Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the catalyst-free synthesis described in CN101638355B for producing high-value naphthol derivatives. As a dedicated CDMO partner, we possess the technical expertise and infrastructure to translate this innovative laboratory protocol into a robust commercial reality. Our facilities are equipped to handle the specific thermal requirements and solvent systems needed for this reaction, ensuring that we can deliver extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of multi-substituted 1-naphthol meets the exacting standards required for downstream pharmaceutical and agrochemical applications.
We invite you to collaborate with us to leverage this advanced synthetic route for your next project. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this catalyst-free method can optimize your budget. Please contact our technical procurement team today to request specific COA data and discuss route feasibility assessments for your target molecules, ensuring a seamless transition from development to full-scale supply.
