Advanced Photocatalytic Synthesis of ((Alkylamino)methyl)diphenylphosphine Oxides for Commercial Scale-up
The landscape of organophosphorus chemistry is undergoing a significant transformation driven by the demand for greener, more efficient synthetic methodologies. Patent CN115322222A introduces a groundbreaking photocatalytic preparation method for ((alkylamino)methyl)diphenylphosphine oxide compounds, addressing critical limitations in traditional synthesis routes. This innovative approach utilizes isocyanide compounds and diphenylphosphine oxide compounds as starting materials, leveraging the power of visible light photocatalysis to construct carbon-phosphorus bonds under remarkably mild conditions. By employing an organic photocatalyst system in a mixture of acetonitrile and water, the process achieves high yields without the need for hazardous reagents or extreme thermal energy. This technological leap represents a pivotal shift towards sustainable manufacturing practices in the fine chemical industry, offering a robust pathway for producing high-purity pharmaceutical intermediates and functional materials.
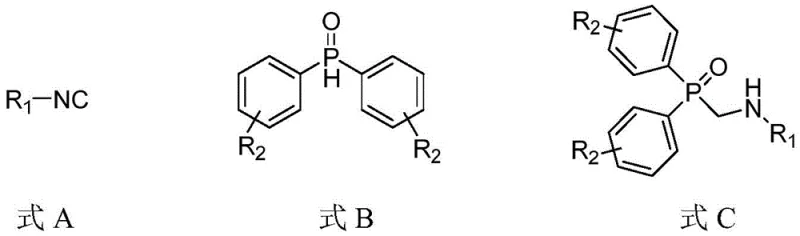
Organophosphorus compounds serve as indispensable building blocks in the pharmaceutical and agrochemical sectors, particularly as protease inhibitors and functional material precursors. Historically, the construction of the alpha-aminomethylphosphoryl scaffold has relied heavily on the Kabachnik-Fields reaction, a three-component condensation involving amines, paraformaldehyde, and phosphoryl compounds. While effective, this conventional methodology necessitates high-temperature heating, which often leads to thermal degradation of sensitive functional groups and generates significant energy consumption. Furthermore, the use of formaldehyde poses safety and handling challenges in large-scale operations. The reliance on thermal activation limits the substrate scope, particularly for thermally labile isocyanides or complex phosphine oxides that might decompose under harsh conditions. These inherent drawbacks create bottlenecks in supply chains, increasing production costs and extending lead times for critical active pharmaceutical ingredients (APIs).
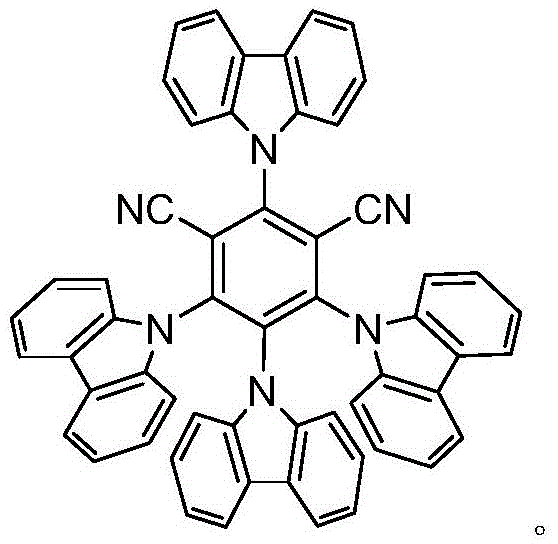
In stark contrast, the novel photocatalytic approach detailed in the patent data circumvents these thermal barriers by harnessing visible light energy to drive the reaction at room temperature. This method employs 2,4,5,6-tetra(9-carbazolyl)-isophthalonitrile, commonly known as 4-CzIPN, as a highly efficient organic photocatalyst. The reaction proceeds smoothly in a mixed solvent system of acetonitrile and water under air conditions, eliminating the need for expensive and cumbersome inert gas protection systems. The use of blue LED irradiation at 405nm provides a controlled energy input that activates the catalyst without inducing unwanted side reactions associated with thermal heating. This shift from thermal to photochemical activation not only enhances the safety profile of the manufacturing process but also significantly expands the chemical space accessible for derivatization, enabling the synthesis of complex structures that were previously difficult to access.
Mechanistic Insights into 4-CzIPN Catalyzed Radical Addition
The core of this technological advancement lies in the unique photophysical properties of the 4-CzIPN catalyst, which facilitates a single-electron transfer (SET) mechanism under visible light irradiation. Upon absorption of blue light photons, the 4-CzIPN molecule transitions to an excited state capable of oxidizing the diphenylphosphine oxide substrate to generate a phosphinoyl radical species. This radical intermediate then undergoes addition to the isocyanide functionality, forming a new carbon-phosphorus bond through a radical addition pathway. The presence of an amine additive, such as DIPEA or DBU, plays a crucial role in stabilizing intermediates and facilitating proton transfer steps necessary for the formation of the final ((alkylamino)methyl)diphenylphosphine oxide product. The mechanistic elegance of this cycle ensures high atom economy and minimizes the formation of byproducts, which is critical for maintaining high purity standards in pharmaceutical manufacturing.
From an impurity control perspective, the mild reaction conditions inherent to this photocatalytic protocol offer substantial advantages over thermal methods. High-temperature processes often promote oligomerization, decomposition, or rearrangement of reactive intermediates, leading to complex impurity profiles that are difficult to separate. By operating at room temperature, the kinetic energy of the system is carefully regulated, favoring the desired radical addition pathway while suppressing competing thermal degradation routes. The use of water as a co-solvent further enhances the green chemistry profile of the reaction, potentially aiding in the solubility of polar intermediates and simplifying the workup procedure. This precise control over the reaction environment ensures that the resulting ((alkylamino)methyl)diphenylphosphine oxides meet stringent purity specifications required for downstream applications in drug discovery and development, reducing the burden on quality control laboratories.
How to Synthesize ((Alkylamino)methyl)diphenylphosphine Oxides Efficiently
The implementation of this photocatalytic strategy requires careful attention to reaction parameters to maximize yield and reproducibility. The standard protocol involves mixing the isocyanide and diphenylphosphine oxide substrates in a specific molar ratio, typically ranging from 1:1 to 1:3, in the presence of the 4-CzIPN catalyst and an amine base. The solvent system, comprising acetonitrile and water in a volume ratio optimized around 100:1, provides the ideal medium for both substrate solubility and catalyst activity. Following the reaction period under 405nm blue LED irradiation, the workup involves simple extraction with ethyl acetate and purification via column chromatography. Detailed standardized synthesis steps see the guide below.
- Combine isocyanide compounds and diphenylphosphine oxide compounds in a reaction vessel with acetonitrile and water as mixed solvents.
- Add the organic photocatalyst 4-CzIPN and an amine additive such as DIPEA to the mixture under air conditions.
- Irradiate the reaction mixture with 405nm blue LED light at room temperature for approximately 12 hours, followed by extraction and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this photocatalytic technology translates into tangible strategic benefits regarding cost structure and operational resilience. The elimination of high-temperature heating requirements drastically reduces energy consumption, directly lowering the utility costs associated with manufacturing. Furthermore, the replacement of transition metal catalysts with an organic photocatalyst like 4-CzIPN removes the necessity for expensive heavy metal scavenging steps, which are often mandatory to meet regulatory limits for residual metals in pharmaceutical products. This simplification of the downstream processing workflow results in significant cost reduction in fine chemical manufacturing, allowing for more competitive pricing models without compromising on quality or compliance standards.
- Cost Reduction in Manufacturing: The transition to a metal-free catalytic system fundamentally alters the cost dynamics of production. Traditional methods often rely on precious metal catalysts that require rigorous removal protocols to prevent contamination of the final API. By utilizing an organic photocatalyst, the process inherently avoids heavy metal contamination, thereby eliminating the capital and operational expenditures related to metal scavengers and specialized filtration equipment. Additionally, the ability to run reactions at room temperature reduces the load on heating and cooling infrastructure, leading to lower maintenance costs and extended equipment lifespan. These cumulative efficiencies contribute to a leaner manufacturing model that is better equipped to handle market fluctuations in raw material prices.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions under air atmosphere significantly de-risks the supply chain. Conventional organometallic reactions frequently demand strictly anhydrous and oxygen-free environments, necessitating complex infrastructure and specialized operator training. The tolerance of this photocatalytic method to air and moisture simplifies the operational requirements, making it easier to scale up across different manufacturing sites without extensive retrofitting. Moreover, the starting materials, including various substituted isocyanides and phosphine oxides, are commercially available or easily synthesized, ensuring a stable supply of feedstocks. This accessibility reduces the risk of production delays caused by raw material shortages, enhancing overall supply chain continuity.
- Scalability and Environmental Compliance: Scaling photochemical reactions has historically been challenging due to light penetration issues, but advancements in LED technology and flow chemistry have made this increasingly viable. The use of low-power 10W blue LEDs indicates that the process does not require high-intensity, heat-generating lamps, facilitating safer scale-up. From an environmental standpoint, the use of water as a co-solvent and the absence of toxic heavy metals align perfectly with modern green chemistry principles and increasingly strict environmental regulations. This compliance reduces the regulatory burden associated with waste disposal and emissions, positioning the manufacturing process as sustainable and future-proof against tightening environmental legislation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic synthesis method. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on feasibility and performance. Understanding these details is essential for R&D teams evaluating the technology for process development and for procurement teams assessing vendor capabilities.
Q: What are the advantages of using 4-CzIPN over traditional transition metal catalysts?
A: 4-CzIPN is an organic metal-free photocatalyst that eliminates the risk of heavy metal contamination in pharmaceutical intermediates, simplifying downstream purification and ensuring compliance with stringent regulatory limits.
Q: Does this photocatalytic method require inert atmosphere conditions?
A: No, the patented process operates efficiently under air conditions at room temperature, significantly reducing operational complexity and equipment costs compared to methods requiring rigorous nitrogen or argon protection.
Q: What is the substrate scope for the isocyanide component in this reaction?
A: The method demonstrates broad compatibility with various isocyanides including tert-butyl, benzyl, and ester-containing alkyl isocyanides, allowing for the synthesis of diverse phosphine oxide derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable ((Alkylamino)methyl)diphenylphosphine Oxide Supplier
As the global demand for high-performance organophosphorus intermediates continues to rise, partnering with a manufacturer that possesses advanced synthetic capabilities is crucial for maintaining a competitive edge. NINGBO INNO PHARMCHEM stands at the forefront of this innovation, leveraging cutting-edge photocatalytic technologies to deliver superior quality chemicals. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of ((alkylamino)methyl)diphenylphosphine oxide meets the highest industry standards for pharmaceutical and agrochemical applications.
We invite you to explore how our advanced manufacturing capabilities can optimize your supply chain and reduce your overall production costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate the viability of integrating this green photocatalytic method into your existing production workflows. Let us collaborate to drive efficiency and innovation in your chemical sourcing strategy.
