Advanced Catalytic Synthesis of D-Biotin Intermediates for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust pathways for synthesizing essential vitamins, and the production of d-biotin remains a critical focus due to its widespread application in nutritional supplements and pharmaceutical formulations. Patent CN1473832A introduces a groundbreaking synthesis method for (3aS, 6aR)-1,3-dibenzyl-tetrahydro-4H-furo[3,4-d]imidazole-2,4(1H)-dione, a pivotal intermediate in the biosynthesis of d-biotin. This technology represents a significant departure from legacy resolution-based processes, utilizing a sophisticated chiral amine-catalyzed asymmetric monoesterification followed by a streamlined reductive cyclization. By leveraging specific (1S, 2S)-amino alcohol catalysts, the process achieves exceptional stereocontrol, delivering the target lactone with optical purity exceeding 98.5% ee and total chemical yields surpassing 88%. For R&D directors and process chemists, this patent offers a compelling blueprint for modernizing vitamin manufacturing lines, replacing inefficient stoichiometric resolutions with a catalytic cycle that maximizes atom economy and minimizes waste generation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of d-biotin intermediates relied heavily on classical resolution techniques that were inherently inefficient and costly. Early methodologies, such as those reported by Gerecke et al., involved the monoesterification of cyclic anhydrides followed by diastereomeric crystallization using resolving agents like pseudoephedrine or dehydroabietamine. These traditional approaches suffered from severe limitations, primarily characterized by low single-pass resolution rates, often capping at approximately 37%, which necessitated extensive recycling of the unwanted enantiomer or resulted in substantial material loss. Furthermore, the reliance on stoichiometric amounts of expensive chiral auxiliaries like cholesterol or optically active secondary alcohols introduced significant cost burdens and complicated downstream purification processes. The operational complexity was further exacerbated by the need for multiple recrystallization steps to achieve acceptable optical purity, leading to prolonged production cycles and increased solvent consumption, which are detrimental to both economic viability and environmental compliance in modern chemical manufacturing.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent employs a direct asymmetric catalytic strategy that fundamentally transforms the efficiency profile of the synthesis. By utilizing chiral amine catalysts to mediate the enantioselective ring-opening of the cyclic anhydride precursor with simple aliphatic alcohols, the process bypasses the need for stoichiometric resolving agents entirely. This catalytic system operates under remarkably mild conditions, typically between -15°C and -10°C, allowing for precise control over the stereochemical outcome without the harsh thermal stresses often associated with older protocols. The subsequent reduction and cyclization steps utilize common reducing agents like potassium borohydride in the presence of inexpensive Lewis acids, facilitating a seamless one-pot or telescoped transformation into the final lactone structure. This shift from resolution to asymmetric catalysis not only drastically improves the overall yield to above 88% but also simplifies the workflow, reducing the number of unit operations and enabling a more continuous and scalable manufacturing process suitable for high-volume demand.
Mechanistic Insights into Chiral Amine-Catalyzed Asymmetric Monoesterification
The core innovation of this synthesis lies in the mechanistic elegance of the chiral amine-catalyzed asymmetric monoesterification, which dictates the stereochemical fate of the molecule. The reaction initiates with the nucleophilic attack of the alcohol on the cyclic anhydride, activated and directed by the chiral environment provided by the (1S, 2S)-amino alcohol catalyst. The catalyst, featuring a bulky trityl group and a nitrophenyl moiety, creates a sterically constrained transition state that favors the formation of one enantiomer over the other through specific hydrogen bonding and steric repulsion interactions. This precise molecular recognition ensures that the ring-opening occurs selectively at one of the carbonyl groups, establishing the critical (4S, 5R) configuration of the intermediate carboxylic acid with high fidelity. The use of methanol or other simple alcohols as nucleophiles is particularly advantageous, as their small size allows for rapid kinetics while the chiral catalyst maintains strict control over the facial selectivity of the addition, effectively converting a prochiral anhydride into a chiral monoester with minimal formation of the undesired enantiomer.
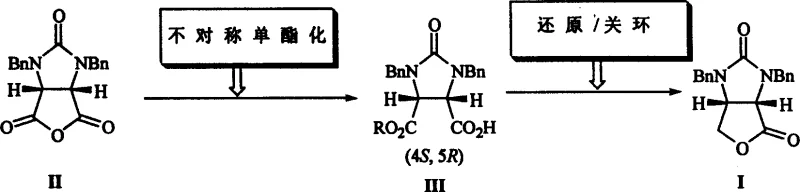
Following the establishment of chirality, the subsequent reductive cyclization mechanism is equally critical for maintaining optical integrity while forming the fused ring system. In the presence of Lewis acids such as anhydrous calcium chloride or lithium chloride, the borohydride species selectively reduces the ester or carboxylic acid functionality to the corresponding alcohol or aldehyde intermediate in situ. This reduction is immediately followed by an acid-catalyzed intramolecular cyclization, where the newly formed hydroxyl group attacks the adjacent urea carbonyl to close the furan ring. The choice of Lewis acid is paramount here, as it coordinates with the carbonyl oxygens to enhance electrophilicity without promoting racemization of the sensitive chiral centers established in the previous step. The entire sequence is designed to proceed with retention of configuration, ensuring that the high enantiomeric excess achieved in the first step is preserved in the final (3aS, 6aR)-lactone product, thereby eliminating the need for further chiral purification.
How to Synthesize (3aS, 6aR)-1,3-dibenzyl-tetrahydro-4H-furo[3,4-d]imidazole-2,4(1H)-dione Efficiently
The practical execution of this synthesis requires careful attention to reaction parameters to maximize the benefits of the catalytic system described in the patent. The process begins with the preparation of the reaction mixture containing the cyclic anhydride, the chiral amino alcohol catalyst, and the alcohol nucleophile in a dry organic solvent such as tetrahydrofuran. Maintaining the temperature strictly within the range of -15°C to -10°C during the monoesterification phase is crucial for achieving the highest enantioselectivity, as deviations can lead to background non-catalyzed reactions that erode optical purity. Once the intermediate acid is formed, the workup involves acidification and extraction to isolate the solid product, which is then subjected to the reduction conditions using potassium borohydride and a Lewis acid in ethanol at room temperature.
- Perform asymmetric monoesterification of the cyclic anhydride precursor using a chiral amino alcohol catalyst and methanol at low temperatures.
- Isolate the chiral monoester intermediate (Compound III) through acidification and crystallization.
- Execute reductive cyclization using potassium borohydride and a Lewis acid in ethanol to form the final lactone product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this catalytic synthesis route offers profound strategic advantages over traditional resolution-based manufacturing. The elimination of stoichiometric chiral resolving agents, which are often expensive and difficult to recover, translates directly into a significant reduction in raw material costs and waste disposal expenses. Furthermore, the dramatic increase in overall yield from typical resolution levels to over 88% means that less starting material is required to produce the same amount of final product, effectively lowering the cost of goods sold and improving margin potential. The use of commodity chemicals such as methanol, ethanol, and common Lewis acids ensures a stable and resilient supply chain, reducing dependency on specialized or scarce reagents that could introduce bottlenecks or price volatility. Additionally, the simplified operational workflow with fewer purification steps reduces energy consumption and solvent usage, aligning with sustainability goals and reducing the environmental footprint of the manufacturing process.
- Cost Reduction in Manufacturing: The transition from stoichiometric resolution to catalytic asymmetric synthesis removes the inherent 50% yield ceiling associated with racemic resolutions, effectively doubling the theoretical output from the same amount of starting anhydride. By avoiding the purchase and subsequent recovery of expensive resolving agents like dehydroabietamine or pseudoephedrine, manufacturers can realize substantial savings in material costs. The high chemical yield also minimizes the volume of waste streams requiring treatment, further lowering operational expenditures related to environmental compliance and waste management infrastructure.
- Enhanced Supply Chain Reliability: The reliance on widely available bulk chemicals such as methanol, potassium borohydride, and calcium chloride ensures that production is not vulnerable to the supply disruptions often associated with specialized chiral auxiliaries. The robustness of the reaction conditions, which tolerate standard industrial solvents and operate at mild temperatures, allows for flexible manufacturing scheduling and reduces the risk of batch failures due to sensitive parameter deviations. This reliability is critical for maintaining consistent inventory levels of high-purity pharmaceutical intermediates, ensuring that downstream customers receive their orders on time without interruption.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing standard reactor configurations and avoiding hazardous reagents or extreme conditions that would require specialized containment. The high atom economy of the catalytic route results in less chemical waste per kilogram of product, facilitating easier compliance with increasingly stringent environmental regulations. The ability to recycle solvents like THF and ethanol further enhances the green chemistry profile of the process, making it an attractive option for companies looking to improve their sustainability metrics while expanding production capacity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. Understanding these details is essential for evaluating the feasibility of integrating this route into existing production facilities.
Q: What is the primary advantage of this catalytic method over traditional resolution?
A: Unlike traditional resolution methods which often suffer from low single-pass yields (around 37%) and require stoichiometric resolving agents, this catalytic asymmetric route achieves total yields exceeding 88% with ee values greater than 98.5%, significantly improving material efficiency.
Q: Which chiral catalysts are most effective for this transformation?
A: The patent identifies (1S, 2S)-amino alcohols, specifically (1S, 2S)-1-(4-nitrophenyl)-2-N,N-dimethylamino-3-trityloxy-1-propanol, as highly effective catalysts that provide excellent enantioselectivity under mild reaction conditions.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes readily available raw materials like methanol and common solvents such as THF and ethanol, operates under mild temperatures, and avoids expensive or difficult-to-recover resolving agents, making it highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (3aS, 6aR)-1,3-dibenzyl-tetrahydro-4H-furo[3,4-d]imidazole-2,4(1H)-dione Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the production of life-saving vitamins and pharmaceuticals. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated catalytic chemistry described in patent CN1473832A can be translated into robust industrial reality. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of (3aS, 6aR)-1,3-dibenzyl-tetrahydro-4H-furo[3,4-d]imidazole-2,4(1H)-dione meets the exacting standards required for d-biotin synthesis. Our commitment to quality assurance means that clients can rely on us for consistent supply of this key building block, supporting their own regulatory filings and market commitments with confidence.
We invite global partners to engage with our technical procurement team to discuss how this advanced synthesis route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this catalytic method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your production needs, ensuring a seamless transition to a more efficient and cost-effective manufacturing strategy.
