Scalable Synthesis of 7-Substituted Tetrahydrocyclobutanocoumarin-5-one for Advanced Drug Discovery
Introduction to Novel Fused Ring Synthesis
The field of organic synthesis is constantly evolving to meet the demands of complex natural product total synthesis, particularly for scaffolds containing strained ring systems. Patent CN109535120B introduces a groundbreaking preparation method for 7-substituted-3,4,4,7-tetrahydrocyclobutanocoumarin-5-one, a critical structural motif found in biologically active natural products such as Kingianin A and Nervonin A. This technology addresses a significant gap in the literature regarding the efficient construction of [4,6,6] annulated frameworks, offering a streamlined pathway that bypasses the traditional limitations of low regioselectivity and harsh reaction conditions. By leveraging a cascade of hypervalent iodine oxidation followed by copper-catalyzed allene formation, this method provides researchers and manufacturers with a robust tool for accessing these valuable intermediates. The strategic design of this synthesis not only enhances the accessibility of these complex molecules but also opens new avenues for the development of novel pharmaceutical agents derived from the coumarin backbone.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of fused ring systems containing a cyclobutane moiety adjacent to a lactone or ketone has been a formidable challenge in synthetic chemistry. Conventional approaches often rely on photochemical [2+2] cycloadditions or complex multistep sequences that suffer from poor atom economy and unpredictable stereochemical outcomes. These traditional routes frequently require specialized equipment, such as high-pressure reactors or UV irradiation setups, which complicate process safety and scalability. Furthermore, the purification of intermediates in these legacy methods is often arduous, leading to significant material loss and reduced overall throughput. The lack of general methods for introducing diverse substituents at the 7-position further restricts the utility of these compounds in medicinal chemistry campaigns, forcing R&D teams to resort to bespoke, low-yielding syntheses for each new analog.
The Novel Approach
In stark contrast, the methodology disclosed in CN109535120B offers a modular and highly efficient alternative that transforms simple phenols into complex fused architectures in just three operational steps. The innovation lies in the use of propargyl alcohol as a bifunctional reagent that serves both as an oxygen source and a carbon backbone extender. By initially forming a spiro-dienone intermediate via oxidative dearomatization, the process sets the stage for a subsequent copper-catalyzed transformation that installs the critical allene functionality. This allene intermediate then undergoes a spontaneous thermal cyclization in protic solvents, driven by the inherent strain release and electronic activation of the system. This approach eliminates the need for exotic reagents or extreme conditions, replacing them with a sequence that is operationally simple, cost-effective, and remarkably high-yielding, thereby representing a paradigm shift in how chemists approach [4,6,6] ring construction.
Mechanistic Insights into Hypervalent Iodine Oxidation and Copper Catalysis
The success of this synthetic route is underpinned by a sophisticated interplay of oxidative dearomatization and transition metal catalysis. The first step utilizes phenyliodine(III) diacetate (PhI(OAc)2) to effect the oxidative coupling of a 4-substituted phenol with propargyl alcohol. This hypervalent iodine reagent acts as a two-electron oxidant, facilitating the formation of a C-O bond at the para-position of the phenol while simultaneously disrupting the aromaticity to generate a reactive 2,5-cyclohexadienone species. This spiro-cyclic intermediate is pivotal, as it locks the geometry required for the subsequent ring closure. The second stage involves a copper(I)-catalyzed reaction with paraformaldehyde and diisopropylamine, which functions as a formaldehyde equivalent in a Mannich-type process. This step effectively extends the carbon chain by one unit and converts the alkyne into an allene, a transformation that is crucial for the final cyclization event. The copper catalyst coordinates with the alkyne and the iminium ion generated in situ, directing the nucleophilic attack to form the hydroxymethyl allenyl ether with high precision.
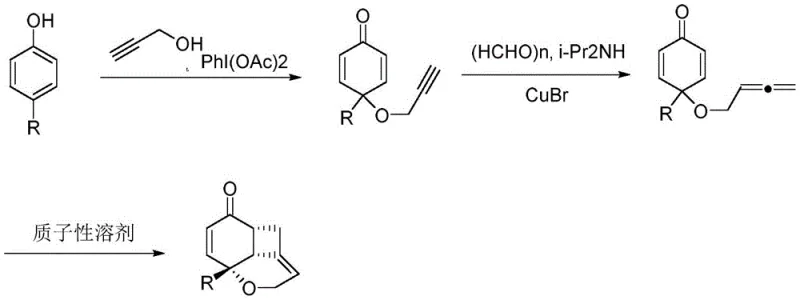
Following the formation of the allene intermediate, the final cyclization is a thermally driven pericyclic-like process that occurs readily in protic solvents. The mechanism likely involves an intramolecular attack of the enol oxygen onto the activated allene system, facilitated by hydrogen bonding from the solvent. This step constructs the strained four-membered ring while restoring the conjugated enone system of the coumarin core. The high regioselectivity observed is attributed to the specific electronic bias imparted by the spiro-center and the allenyl ether moiety, which directs the cyclization exclusively to the desired [4,6,6] framework. Understanding this mechanistic pathway is essential for process chemists, as it highlights the importance of maintaining anhydrous conditions during the copper catalysis step while ensuring sufficient proton source availability in the final thermal step to drive the equilibrium towards the product.
How to Synthesize 7-Substituted-3,4,4,7-tetrahydrocyclobutanocoumarin-5-one Efficiently
To implement this synthesis effectively, operators must adhere to specific parameters regarding stoichiometry and solvent selection to maximize yield and purity. The process begins with the careful addition of PhI(OAc)2 to a solution of the phenol and propargyl alcohol at controlled temperatures to manage the exotherm of the oxidation. Following isolation of the spiro-dienone, the copper-catalyzed step requires precise molar ratios of diisopropylamine and paraformaldehyde to ensure complete conversion to the allene without over-reaction. The detailed standardized synthesis steps, including specific quenching procedures and purification protocols, are outlined below to ensure reproducibility across different laboratory settings.
- Oxidize 4-substituted phenol with propargyl alcohol using PhI(OAc)2 to generate the spiro-dienone intermediate.
- React the spiro-dienone with paraformaldehyde and diisopropylamine under CuBr catalysis to form the hydroxymethyl allenyl ether.
- Heat the allenyl ether intermediate in a protic solvent like TFE at 90-120°C to induce cyclization into the final fused ring product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented process offers substantial benefits that directly address the pain points of procurement and supply chain management in the fine chemical sector. The reliance on commodity chemicals such as p-cresol, propargyl alcohol, and paraformaldehyde ensures that raw material costs remain low and supply is stable, mitigating the risks associated with sourcing exotic or custom-synthesized starting materials. Furthermore, the elimination of expensive noble metal catalysts in the final cyclization step significantly reduces the cost of goods sold (COGS) and simplifies the removal of metal impurities, a critical factor for pharmaceutical grade intermediates. The robustness of the reaction conditions, which tolerate a range of temperatures and do not require inert atmospheres for the final step, translates to lower energy consumption and reduced operational complexity in a manufacturing plant.
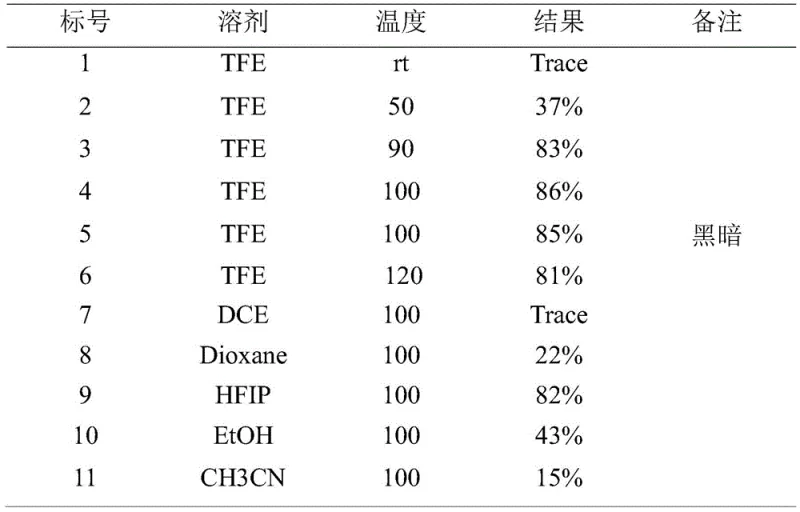
The data presented in the patent underscores the economic viability of using 2,2,2-Trifluoroethanol (TFE) as the solvent for the key cyclization step. As illustrated in the optimization table, TFE delivers superior yields compared to conventional solvents like DCE or Dioxane, which produce only trace amounts or moderate yields respectively. While fluorinated solvents can sometimes be costly, the dramatic increase in yield (up to 86%) and the potential for solvent recovery and reuse make this a cost-effective choice for high-value intermediate production. This efficiency gain means less waste generation and higher throughput per batch, directly enhancing the supply chain reliability for downstream customers. Additionally, the high purity achieved (98% by HPLC) minimizes the need for extensive recrystallization or preparative HPLC, further streamlining the manufacturing timeline and reducing lead times for delivering high-purity pharmaceutical intermediates to clients.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the scalability and specific reaction conditions of this synthesis. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for technical teams evaluating this route for potential adoption.
Q: What is the key advantage of this synthesis route for [4,6,6] fused rings?
A: The primary advantage lies in its modularity and high regioselectivity. Unlike traditional methods that struggle with constructing the strained cyclobutane ring fused to the coumarin system, this patent utilizes a tandem allene-mediated cyclization that proceeds with excellent yields (up to 86%) and high purity (98% HPLC), significantly simplifying the construction of complex natural product scaffolds like Kingianin A analogs.
Q: Which solvent system provides the optimal yield for the cyclization step?
A: According to the optimization data in patent CN109535120B, 2,2,2-Trifluoroethanol (TFE) is the superior solvent for the final thermal cyclization. Screening results demonstrate that while non-polar solvents like DCE yield only trace amounts, TFE facilitates the reaction efficiently at 100°C, achieving yields as high as 86%, likely due to its unique hydrogen-bonding donor capability which stabilizes the transition state.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process is highly amenable to scale-up. It relies on commercially available starting materials such as p-cresol and propargyl alcohol, avoids expensive transition metal catalysts in the final step, and utilizes robust thermal conditions. The simplicity of the work-up procedures, involving standard extraction and column chromatography, ensures that the process can be translated from gram-scale laboratory synthesis to multi-kilogram production with minimal technical barriers.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 7-Substituted-3,4,4,7-tetrahydrocyclobutanocoumarin-5-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced intermediates play in accelerating drug discovery pipelines. Our team of expert process chemists has thoroughly analyzed the methodology described in CN109535120B and is fully equipped to translate this laboratory-scale innovation into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our state-of-the-art facilities are designed to handle the specific requirements of hypervalent iodine chemistry and copper catalysis safely, while our rigorous QC labs guarantee that every batch meets stringent purity specifications required for clinical and commercial applications.
We invite you to collaborate with us to leverage this cutting-edge synthesis for your next project. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements, helping you optimize your budget without compromising on quality. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how we can become your trusted partner in the supply of complex fused ring intermediates.
