Revolutionizing Antidepressant Intermediate Production via Safe Orthoester-Mediated Cyclization Technology
Introduction to Next-Generation Antidepressant Intermediate Synthesis
The global pharmaceutical landscape is increasingly demanding sustainable and scalable manufacturing routes for complex active pharmaceutical ingredients (APIs) and their precursors. In this context, the technological breakthroughs detailed in patent CN101107228A represent a significant paradigm shift in the production of (Z)-1-phenyl-1-(N,N-diethylaminocarbonyl)-2-phthalimidomethylcyclopropane, a critical intermediate for potent antidepressant medications. This patent discloses a novel synthetic strategy that fundamentally reimagines the activation of hydroxymethyl cyclopropane derivatives, moving away from hazardous halogenation towards a more benign orthoester-mediated activation protocol. By leveraging the unique reactivity of iminium salts generated in situ, this methodology offers a robust solution to long-standing challenges in process safety and environmental compliance. For R&D directors and process chemists, understanding the nuances of this transformation is essential for developing next-generation manufacturing protocols that align with modern green chemistry principles while maintaining rigorous quality standards.
Furthermore, the implications of this technology extend far beyond the laboratory bench, offering tangible benefits for supply chain resilience and cost efficiency in high-volume production environments. The elimination of corrosive chlorinating agents and volatile halogenated solvents addresses critical pain points for procurement managers who are tasked with sourcing raw materials that meet increasingly stringent regulatory frameworks. As the industry moves towards stricter controls on emissions and waste disposal, adopting a synthesis route that inherently minimizes hazardous byproducts becomes a strategic imperative. This report delves deep into the mechanistic underpinnings and commercial viability of this innovative approach, providing a comprehensive analysis for stakeholders involved in the sourcing and manufacturing of high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of phthalimidomethyl cyclopropane derivatives has relied heavily on classical halogenation strategies, typically employing thionyl chloride (SOCl2) to convert hydroxymethyl precursors into their corresponding chloromethyl analogues. While chemically effective, this conventional pathway presents severe operational and environmental drawbacks that hinder its suitability for modern large-scale manufacturing. The primary concern is the generation of stoichiometric amounts of sulfur dioxide (SO2) gas, a toxic and corrosive byproduct that necessitates sophisticated and expensive gas scrubbing infrastructure to ensure worker safety and regulatory compliance. Additionally, these traditional methods often require the use of low-boiling halogenated hydrocarbon solvents, such as dichloroethane, which are subject to intense scrutiny due to their ozone-depleting potential and carcinogenic properties. The handling of such solvents increases the complexity of solvent recovery systems and elevates the overall cost of goods sold (COGS) through increased waste treatment fees and safety monitoring requirements.
Beyond the environmental hazards, the multi-step nature of the conventional process introduces significant inefficiencies in terms of throughput and yield optimization. The isolation of the unstable chloromethyl intermediate often leads to material degradation and impurity accumulation, which complicates downstream purification efforts. For supply chain heads, reliance on such archaic chemistry translates to longer lead times and higher vulnerability to supply disruptions caused by regulatory changes on hazardous reagents. The need for strict temperature control during the exothermic chlorination step further limits the scalability of the process, often requiring specialized reactor configurations that are not universally available in standard CDMO facilities. Consequently, there is a pressing industry need for a streamlined alternative that circumvents these bottlenecks while delivering superior product quality and process reliability.
The Novel Approach
The innovative methodology described in the patent data offers a transformative solution by replacing the hazardous chlorination step with a mild and highly efficient orthoester-mediated activation. In this novel route, the hydroxymethyl cyclopropane precursor is reacted directly with an orthoester, such as triethyl orthoformate, in the presence of a protic acid like methanesulfonic acid. This reaction sequence generates a reactive iminium salt intermediate in situ, which serves as a superior electrophile for the subsequent nucleophilic substitution with phthalimide. By bypassing the formation of discrete alkyl halides, this approach completely eliminates the evolution of sulfur dioxide gas and removes the necessity for handling corrosive thionyl chloride. The result is a significantly cleaner reaction profile that reduces the burden on environmental health and safety (EHS) departments and lowers the capital expenditure required for pollution control equipment.
Moreover, this new process enables a telescoped "one-pot" operation where the activation and substitution steps can be performed sequentially in the same reactor vessel without isolating the intermediate. This consolidation of unit operations drastically reduces processing time, solvent consumption, and labor costs, directly addressing the cost reduction imperatives of procurement teams. The use of methanesulfonic acid as the proton source provides excellent solubility characteristics and reaction control, allowing the process to proceed under milder thermal conditions compared to traditional halogenation. For manufacturers seeking a reliable pharmaceutical intermediates supplier, this technology represents a mature and scalable platform that ensures consistent batch-to-batch quality while adhering to the highest standards of sustainable manufacturing practices.
Mechanistic Insights into Orthoester-Mediated Iminium Salt Formation
To fully appreciate the technical superiority of this synthesis, one must examine the mechanistic details of the iminium salt formation, which serves as the cornerstone of this novel pathway. The reaction initiates with the protonation of the orthoester by the strong protic acid, generating a highly electrophilic oxocarbenium species. This activated species then interacts with the hydroxyl group of the cyclopropane substrate, facilitating the departure of the hydroxyl moiety as water and establishing a stabilized cationic center. Unlike the transient and often unstable carbocations formed in acid-catalyzed dehydration, the resulting iminium ion is resonance-stabilized by the adjacent nitrogen atom of the diethylamide group, preventing unwanted rearrangement or polymerization side reactions. This stability is crucial for maintaining the stereochemical integrity of the Z-configuration, ensuring that the final product meets the stringent chiral purity requirements demanded by regulatory agencies for antidepressant APIs.
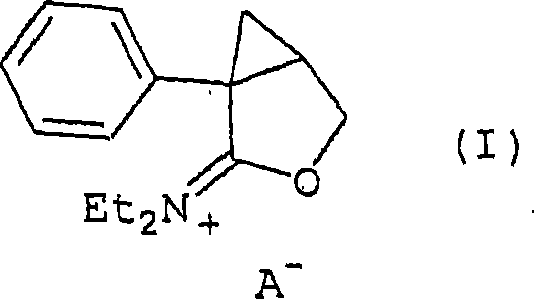
The structure of this key intermediate, depicted in the provided visual data, highlights the cationic nature of the species where A- represents the conjugate base of the protonic acid, preferably the methanesulfonate anion. The presence of this non-nucleophilic counterion is vital, as it prevents premature substitution or elimination reactions that could degrade the sensitive cyclopropane ring system. Once formed, this iminium salt acts as a potent electrophile that readily undergoes nucleophilic attack by the phthalimide anion. The mechanism proceeds via an SN2-type displacement where the phthalimide nitrogen attacks the methylene carbon, displacing the orthoester-derived leaving group. This concerted mechanism ensures high regioselectivity and minimizes the formation of bis-substituted impurities, which are common pitfalls in less controlled alkylation processes. Understanding this mechanistic nuance allows process chemists to fine-tune reaction parameters such as acid stoichiometry and addition rates to maximize yield and minimize impurity profiles.
Furthermore, the choice of methanesulfonic acid over other mineral acids is dictated by its dual role as both a catalyst and a solubilizing agent. The methanesulfonate anion provides a balanced nucleophilicity that stabilizes the transition state without competing with the phthalimide nucleophile. This delicate balance is essential for achieving the high conversion rates reported in the patent examples, where yields exceeding 93% are routinely observed. For R&D directors evaluating process feasibility, this mechanistic clarity provides confidence in the robustness of the chemistry, as the reaction pathway is well-defined and less susceptible to variations in raw material quality. The ability to generate such a reactive intermediate under mild conditions (20-40°C) also underscores the energy efficiency of the process, contributing to a lower carbon footprint for the overall manufacturing campaign.
How to Synthesize (Z)-Phthalimidomethylcyclopropane Efficiently
Implementing this advanced synthesis route requires precise control over reaction conditions to ensure the successful generation and consumption of the reactive iminium intermediate. The process begins with the preparation of the activation mixture, where the hydroxymethyl precursor is dissolved in a suitable solvent such as toluene, followed by the addition of triethyl orthoformate. The careful addition of methanesulfonic acid at controlled temperatures is critical to manage the exotherm and prevent localized overheating that could degrade the sensitive cyclopropane ring. Once the iminium salt is fully generated, indicated by the completion of the activation phase, the reaction mixture is concentrated to remove volatile byproducts before being redissolved in a polar aprotic solvent like DMF for the substitution step. This solvent switch is a key operational detail that optimizes the solubility of the potassium phthalimide nucleophile, ensuring homogeneous reaction conditions.
- Activate the hydroxymethyl precursor using triethyl orthoformate and methanesulfonic acid to generate the reactive iminium salt intermediate in situ.
- Introduce potassium phthalimide directly into the reaction mixture to effect nucleophilic substitution without isolating the unstable intermediate.
- Quench the reaction with water, filter the precipitated crystals, and dry under reduced pressure to obtain high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this orthoester-mediated technology offers profound strategic advantages that extend well beyond simple chemical yield improvements. The most significant benefit lies in the drastic simplification of the waste management profile, as the complete elimination of sulfur dioxide generation removes the need for costly caustic scrubbing systems and associated maintenance protocols. This reduction in hazardous waste streams translates directly into substantial cost savings in terms of waste disposal fees and regulatory reporting burdens, allowing manufacturing sites to operate with greater flexibility and lower overhead. Additionally, the removal of halogenated solvents from the process bill of materials mitigates the risk of supply chain disruptions caused by environmental regulations restricting the production or transport of such chemicals, thereby enhancing the long-term security of supply for critical intermediates.
From a scalability perspective, the ability to telescope the activation and substitution steps into a single vessel operation significantly reduces the equipment footprint and cycle time required for production. This intensification of the process allows existing manufacturing facilities to increase throughput without the need for major capital investment in new reactor trains, offering a rapid path to commercial scale-up for high-purity pharmaceutical intermediates. The improved safety profile, characterized by the absence of corrosive gases and flammable halogenated solvents, also leads to lower insurance premiums and reduced downtime for safety inspections. Furthermore, the high purity of the crude product obtained through this method minimizes the need for extensive downstream purification, such as repeated recrystallizations or chromatography, which are often the most expensive and time-consuming stages of API manufacturing. These cumulative efficiencies create a compelling economic case for transitioning to this greener, more robust synthetic platform.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route, derived from the specific advantages outlined in the patent literature. These insights are intended to clarify the operational benefits and feasibility of adopting this technology for large-scale production of antidepressant intermediates. By understanding the specific solutions this method offers to traditional pain points, stakeholders can make informed decisions about integrating this chemistry into their existing manufacturing portfolios.
Q: How does this new method improve safety compared to traditional thionyl chloride routes?
A: The novel process eliminates the use of thionyl chloride, thereby preventing the generation of hazardous sulfur dioxide gas and removing the need for complex gas scrubbing systems.
Q: What are the solvent advantages of the orthoester-mediated pathway?
A: This method avoids low-boiling halogenated hydrocarbons like dichloroethane, utilizing safer alternatives such as toluene and DMF, which simplifies solvent recovery and waste treatment.
Q: Can this process be scaled for commercial API manufacturing?
A: Yes, the ability to perform the activation and substitution in a single vessel (one-pot) significantly reduces processing time and equipment footprint, facilitating efficient commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (Z)-Phthalimidomethylcyclopropane Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced, green synthesis routes requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN101107228A are fully realized in a commercial setting. Our state-of-the-art facilities are equipped to handle the specific solvent systems and reaction conditions required for this orthoester-mediated process, maintaining stringent purity specifications through our rigorous QC labs. We understand that consistency is paramount in the pharmaceutical supply chain, and our robust quality management systems guarantee that every batch of (Z)-1-phenyl-1-(N,N-diethylaminocarbonyl)-2-phthalimidomethylcyclopropane meets the highest international standards for identity, strength, and purity.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this innovative technology for their antidepressant development programs. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis that quantifies the specific economic benefits of switching to this greener route for your specific volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments tailored to your project timelines. Let us help you optimize your supply chain, reduce your environmental footprint, and accelerate your time to market with our reliable supply of high-quality pharmaceutical intermediates.
