Advanced Synthesis of p-NH2-Bn-NOTA for High-Purity Radiopharmaceutical Applications
Introduction to Next-Generation Chelator Synthesis
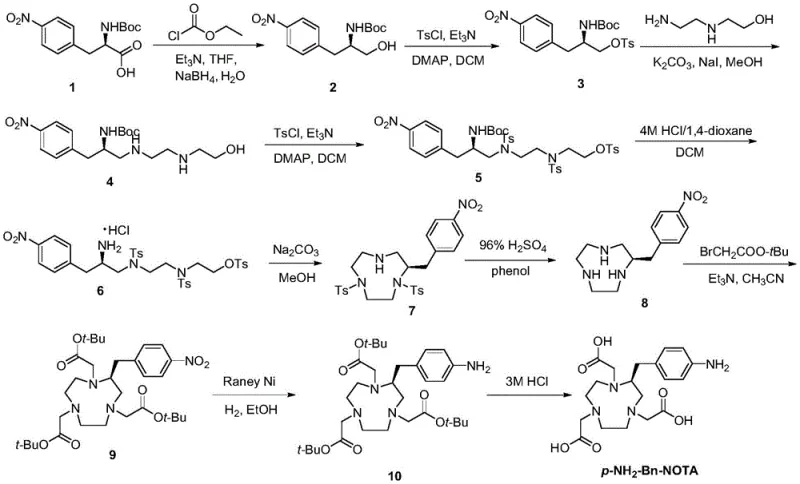
The rapidly evolving landscape of nuclear medicine, particularly Positron Emission Tomography (PET), demands bifunctional chelating agents of exceptional purity and thermodynamic stability. Patent CN114805231A introduces a groundbreaking synthetic methodology for p-NH2-Bn-NOTA, a critical precursor for radiopharmaceutical labeling, which fundamentally addresses the severe limitations of prior art. This innovation leverages a sophisticated sequence of carboxylic acid reduction, bimolecular nucleophilic substitution, and controlled cyclization to achieve a total yield of approximately 33%, a staggering improvement over the historical benchmark of 0.86%. By systematically eliminating the reliance on hazardous borane reagents and optimizing purification protocols, this technology offers a robust pathway for the reliable pharmaceutical intermediate supplier seeking to enhance their portfolio with high-value radiolabeling components.
For R&D Directors and Procurement Managers, the implications of this patent extend far beyond simple yield metrics; it represents a paradigm shift in process safety and economic viability. The traditional reliance on toxic borane complexes not only inflated operational costs due to specialized handling requirements but also introduced significant environmental liabilities. In contrast, the disclosed method utilizes sodium borohydride and standard organic solvents, creating a greener, more sustainable manufacturing footprint. This transition is pivotal for companies aiming to secure long-term supply chains for complex chelators, ensuring that the production of life-saving diagnostic agents remains uninterrupted by regulatory or safety bottlenecks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of NOTA derivatives like p-NH2-Bn-NOTA has been plagued by inefficient reaction pathways that rely heavily on dangerous and expensive reagents. Prior art strategies, such as those reported by Claude F. and Mohammad H. Ansari, typically necessitate the use of borane (BH3) for the critical reduction of carbonyl groups to amines or alcohols. Borane is not only highly toxic and pyrophoric, requiring stringent safety protocols and specialized equipment, but it is also cost-prohibitive for large-scale operations. Furthermore, these conventional routes often suffer from abysmal overall yields, frequently hovering below 1%, primarily due to the formation of complex impurity profiles that are difficult to separate.
The purification challenges associated with these legacy methods are equally daunting, often mandating extensive column chromatography which is notoriously difficult to scale industrially. The combination of low throughput, high reagent costs, and complex downstream processing creates a fragile supply chain vulnerable to disruptions. For a procurement manager, this translates to unpredictable lead times and volatile pricing for high-purity bifunctional chelating agents. Additionally, the environmental impact of disposing of boron-containing waste streams adds another layer of regulatory complexity, making these traditional routes increasingly untenable for modern, compliance-driven manufacturing facilities.
The Novel Approach
The innovative route disclosed in CN114805231A dismantles these barriers by introducing a safer, high-efficiency synthetic strategy centered on sodium borohydride reduction and optimized cyclization conditions. Instead of hazardous borane, the process employs a mixed anhydride method using ethyl chloroformate and sodium borohydride to reduce the carboxylic acid moiety, achieving yields as high as 86% in the initial step alone. This substitution drastically reduces raw material costs and eliminates the need for specialized hazard containment, thereby streamlining the entire production workflow. The subsequent steps utilize bimolecular nucleophilic substitutions and intramolecular cyclizations that proceed under moderate conditions, avoiding the extreme dilution often required for macrocycle formation.
Furthermore, the novel approach incorporates robust purification techniques, such as liquid-liquid extraction and recrystallization, which are inherently more scalable than column chromatography. The strategic use of tosyl protecting groups allows for precise control over the alkylation sequence, minimizing side reactions and ensuring the structural integrity of the triazacyclononane core. By achieving a cumulative yield of roughly 33%, this method offers a substantial increase in material throughput, directly addressing the supply constraints faced by the radiopharmaceutical industry. This technological leap ensures that the production of p-NH2-Bn-NOTA is not only economically feasible but also environmentally responsible, aligning perfectly with the goals of cost reduction in radiopharmaceutical manufacturing.
Mechanistic Insights into Safe Reduction and Cyclization
The cornerstone of this improved synthesis lies in the mechanistic elegance of the initial reduction step, where the carboxylic acid group of the nitro-phenylalanine derivative is converted into a primary alcohol. Mechanistically, this involves the in situ formation of a mixed anhydride intermediate upon reaction with ethyl chloroformate and triethylamine at low temperatures (0°C). This activated species is then selectively reduced by sodium borohydride, a mild yet effective hydride donor that avoids the over-reduction or side reactions common with stronger reducing agents. The presence of water in the reaction mixture is carefully controlled to facilitate the decomposition of the borate complexes, ensuring a clean conversion to the alcohol product without compromising the nitro group or the Boc-protected amine.
Following the construction of the linear polyamine chain through sequential tosylation and nucleophilic substitution, the critical macrocyclization step forms the nine-membered triazacyclononane ring. This step is facilitated by the use of concentrated sulfuric acid and phenol at elevated temperatures (100-105°C), which promotes the intramolecular nucleophilic attack of the secondary amines on the electrophilic centers. The choice of phenol as a co-solvent or additive is crucial, as it helps to solubilize the polar intermediates and stabilize the transition state, leading to a respectable 85% yield for this challenging transformation. This mechanistic understanding allows for precise tuning of reaction parameters to maximize purity and minimize the formation of oligomeric byproducts.
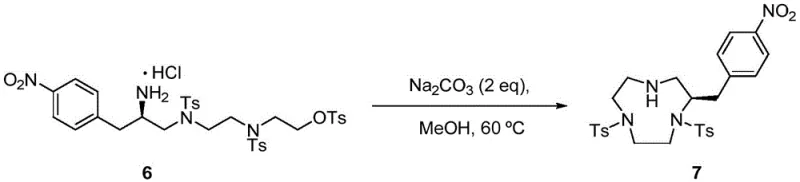
Impurity control is rigorously maintained throughout the synthesis by leveraging the differential solubility of intermediates and the stability of the tosyl protecting groups. For instance, the removal of tosyl groups is achieved under specific acidic conditions that do not affect the integrity of the newly formed macrocycle or the pendant nitrobenzyl arm. The final alkylation with tert-butyl bromoacetate is performed under basic conditions using triethylamine, ensuring selective N-alkylation of the ring nitrogens without ester hydrolysis. This level of chemoselectivity is vital for producing high-purity bifunctional chelating agents, as even trace impurities can interfere with the subsequent radiolabeling efficiency or alter the biodistribution of the final PET tracer.
How to Synthesize p-NH2-Bn-NOTA Efficiently
The synthesis of p-NH2-Bn-NOTA described in this patent offers a clear, step-by-step protocol that balances high yield with operational simplicity, making it ideal for both laboratory optimization and industrial scale-up. The process begins with the safe reduction of the starting amino acid derivative, followed by chain elongation and ring closure, culminating in the final deprotection to reveal the free carboxylic acids and the aniline functionality. Each step has been optimized to minimize waste and maximize recovery, utilizing common solvents like dichloromethane, methanol, and ethanol which are easily recycled. Detailed standardized synthetic steps see the guide below.
- Perform carboxylic acid reduction using ethyl chloroformate and sodium borohydride to safely convert the nitro-phenylalanine derivative to the corresponding alcohol.
- Execute sequential tosylation and nucleophilic substitution reactions to build the polyamine chain, followed by acid-mediated deprotection.
- Complete the macrocyclization using sulfuric acid and phenol, followed by alkylation with tert-butyl bromoacetate and final catalytic hydrogenation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route translates into tangible strategic advantages that go beyond mere technical specifications. The elimination of toxic borane reagents significantly lowers the barrier to entry for manufacturing, as it removes the need for expensive hazard mitigation infrastructure and specialized waste disposal contracts. This simplification of the safety profile allows for more flexible production scheduling and reduces the risk of shutdowns due to regulatory non-compliance or safety incidents. Consequently, suppliers adopting this method can offer more competitive pricing structures, driven by lower operational expenditures and higher material efficiency.
- Cost Reduction in Manufacturing: The shift from low-yield borane chemistry to high-yield sodium borohydride reduction results in a dramatic decrease in the cost of goods sold. By improving the overall yield from less than 1% to over 30%, the amount of raw material required to produce a kilogram of final product is drastically reduced, leading to substantial cost savings. Additionally, the avoidance of column chromatography in favor of extraction and crystallization reduces solvent consumption and labor costs, further enhancing the economic viability of the process. These efficiencies allow for a more aggressive pricing strategy in the competitive market for radiopharmaceutical precursors.
- Enhanced Supply Chain Reliability: The use of commercially available, non-hazardous reagents ensures a stable and resilient supply chain that is less susceptible to geopolitical or logistical disruptions. Unlike specialized borane complexes which may have limited suppliers and long lead times, sodium borohydride and ethyl chloroformate are commodity chemicals with robust global availability. This reliability is critical for maintaining continuous production schedules and meeting the just-in-time delivery requirements of pharmaceutical clients. Furthermore, the scalability of the process means that supply can be rapidly ramped up to meet surges in demand without compromising quality or safety.
- Scalability and Environmental Compliance: The process design inherently supports commercial scale-up of complex chelators by utilizing unit operations that are standard in the fine chemical industry. The absence of high-dilution conditions and the use of benign solvents simplify the engineering requirements for large-scale reactors, facilitating a smoother transition from pilot plant to full commercial production. From an environmental perspective, the reduction in toxic waste generation aligns with increasingly stringent global environmental regulations, reducing the carbon footprint and liability associated with chemical manufacturing. This sustainability angle is becoming a key differentiator for suppliers partnering with major pharmaceutical corporations committed to green chemistry initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of p-NH2-Bn-NOTA, based on the detailed disclosures within the patent literature. Understanding these nuances is essential for stakeholders evaluating the feasibility of integrating this intermediate into their radiopharmaceutical development pipelines. The answers provided reflect the specific advantages of the new methodology over traditional approaches, focusing on yield, safety, and purity.
Q: How does the new synthesis method improve upon traditional p-NH2-Bn-NOTA production?
A: The new method replaces toxic and expensive borane reagents with safer sodium borohydride, increasing total yield from 0.86% to approximately 33% while simplifying purification.
Q: Is this synthesis route suitable for large-scale commercial manufacturing?
A: Yes, the process avoids high-dilution conditions and hazardous reagents, utilizing standard extraction and crystallization techniques that are highly scalable for industrial production.
Q: What are the purity specifications achievable with this method?
A: The method employs rigorous purification steps including column chromatography and recrystallization, ensuring high-purity intermediates suitable for GMP radiopharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable p-NH2-Bn-NOTA Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality bifunctional chelating agents play in the advancement of next-generation diagnostic imaging. Our technical team has thoroughly analyzed the synthetic route disclosed in CN114805231A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this efficient process to market. We are committed to delivering p-NH2-Bn-NOTA with stringent purity specifications and rigorous QC labs testing to ensure it meets the exacting standards of the global radiopharmaceutical industry. Our state-of-the-art facilities are equipped to handle the specific reagents and conditions of this novel route safely and efficiently.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis can benefit your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to our supply source. We encourage you to contact us directly to obtain specific COA data and route feasibility assessments tailored to your volume requirements. Let us collaborate to secure a stable, cost-effective supply of this vital intermediate for your PET imaging applications.
