Advanced Synthesis of 1,2,3-Triazole Cinnamic Acid Derivatives for Commercial Scale-Up
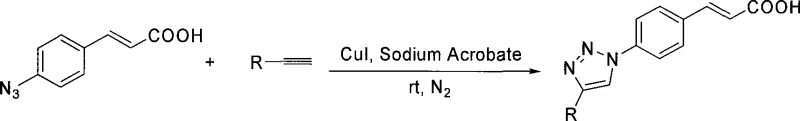
The pharmaceutical and agrochemical industries continuously seek robust synthetic routes for heterocyclic scaffolds that offer both biological efficacy and manufacturing feasibility. Patent CN101519380A introduces a significant advancement in this domain by detailing a method for preparing compounds containing 1,2,3-triazole cinnamic acid moieties. This technology leverages the principles of Click Chemistry, specifically the Copper(I)-catalyzed Azide-Alkyne Cycloaddition (CuAAC), to couple 4-azidocinnamic acid with various terminal alkynes. The resulting 1,2,3-triazole ring serves as a critical pharmacophore, known for its metabolic stability and ability to mimic peptide bonds, making these derivatives highly valuable as intermediates for antifungal, antiviral, and antitumor agents. By shifting from harsh thermal conditions to a mild, catalytic environment, this patent addresses long-standing challenges in triazole synthesis, offering a pathway that is not only chemically elegant but also commercially viable for the production of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,2,3-triazoles relied on the thermal Huisgen 1,3-dipolar cycloaddition between organic azides and alkynes. This conventional approach suffers from severe drawbacks that hinder its utility in modern process chemistry. Primarily, the thermal reaction lacks regioselectivity, invariably producing a difficult-to-separate mixture of both 1,4-disubstituted and 1,5-disubstituted triazole isomers. This lack of stereocontrol necessitates complex and costly downstream purification steps, such as repeated recrystallizations or preparative HPLC, which drastically reduce overall process efficiency and increase solvent waste. Furthermore, thermal cycloadditions often require elevated temperatures and prolonged reaction times, posing safety risks associated with the handling of energetic azide compounds under heat. These factors collectively result in higher production costs and inconsistent batch quality, making the traditional thermal route unsuitable for the cost-sensitive manufacturing of bulk pharmaceutical intermediates.
The Novel Approach
The methodology disclosed in CN101519380A represents a paradigm shift by employing a copper-catalyzed system that operates under exceptionally mild conditions. By utilizing cuprous iodide (CuI) and sodium ascorbate as an additive system, the reaction proceeds efficiently at room temperature under a nitrogen atmosphere. This catalytic cycle enforces strict regioselectivity, yielding exclusively the 1,4-disubstituted 1,2,3-triazole product without the formation of the 1,5-isomer. The operational simplicity is remarkable; the process involves mixing the 4-azidocinnamic acid, the terminal alkyne, and the catalyst system, followed by a straightforward aqueous workup and extraction. This elimination of isomeric byproducts simplifies the purification landscape, allowing for high-purity isolation via standard silica gel chromatography. The ability to run the reaction at ambient temperature also significantly reduces energy consumption and mitigates safety risks associated with thermal runaway, positioning this novel approach as a superior alternative for sustainable chemical manufacturing.
Mechanistic Insights into Cu-Catalyzed Azide-Alkyne Cycloaddition
The core of this synthetic strategy lies in the mechanistic efficiency of the Cu(I)-catalyzed cycle. The reaction initiates with the coordination of the terminal alkyne to the copper(I) species, facilitated by the presence of sodium ascorbate which acts as a reducing agent to maintain the copper in its active +1 oxidation state and prevent oxidation to inactive Cu(II). This interaction generates a copper-acetylide intermediate, which is significantly more nucleophilic than the free alkyne. The azide group of the 4-azidocinnamic acid then attacks this activated alkyne complex, leading to the formation of a six-membered copper-metallacycle intermediate. This metallacycle subsequently undergoes ring contraction to form the triazole ring, followed by protonolysis to release the final 1,4-disubstituted product and regenerate the copper catalyst. This mechanism explains the observed high regioselectivity, as the steric and electronic constraints of the copper-metallacycle favor the formation of the 1,4-isomer over the 1,5-isomer, ensuring a clean reaction profile.
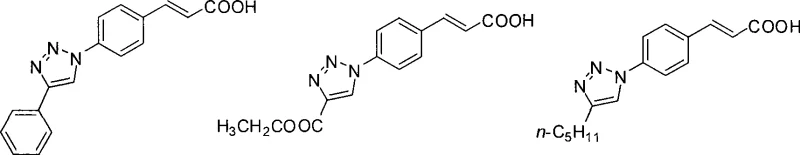
From an impurity control perspective, this mechanism offers distinct advantages for process chemists aiming for high-purity outputs. Because the catalytic cycle effectively suppresses the formation of regioisomers, the primary impurity profile is limited to unreacted starting materials rather than structurally similar byproducts. The patent data indicates that varying the R-group on the alkyne (such as phenyl, ester, or alkyl chains) does not compromise the selectivity of the cyclization. For instance, the reaction with phenylacetylene, ethyl propiolate, and heptyne all proceed to form the corresponding triazoles with the cinnamic acid moiety intact. This robustness suggests that the electronic nature of the alkyne substituent has minimal impact on the critical cyclization step, allowing for a broad substrate scope without the need for extensive re-optimization of reaction parameters for each new derivative.
How to Synthesize 1,2,3-Triazole Cinnamic Acid Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale execution. The process begins by charging the reactor with 4-azidocinnamic acid and the chosen terminal alkyne in a molar ratio ranging from 1:1 to 1:1.2. To this mixture, catalytic amounts of cuprous iodide and sodium ascorbate are added, typically at 10 mol% relative to the azide. The reaction vessel is purged with nitrogen to create an inert atmosphere, preventing oxidative degradation of the catalyst or reactants. The mixture is then stirred at room temperature for a period ranging from 24 to 72 hours, depending on the reactivity of the specific alkyne employed. Upon completion, indicated by TLC analysis, the reaction is quenched with water and extracted with ethyl acetate. The organic layer is washed, dried over anhydrous sodium sulfate, and concentrated. Final purification is achieved through silica gel column chromatography, yielding the target 1,2,3-triazole cinnamic acid derivatives as high-purity solids.
- Charge 4-azidocinnamic acid, terminal alkyne, sodium ascorbate, and cuprous iodide into a reaction flask under nitrogen protection.
- Stir the reaction mixture at room temperature for 24 to 72 hours, monitoring progress via TLC until completion.
- Quench with water, extract with ethyl acetate, dry the organic phase, and purify the crude product using silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this catalytic methodology offers tangible economic and logistical benefits. The most significant advantage is the drastic simplification of the purification process. By eliminating the formation of regioisomeric impurities, the need for expensive and time-consuming separation techniques is removed, leading to substantial cost savings in solvent usage and labor hours. Furthermore, the use of commodity chemicals such as cuprous iodide and sodium ascorbate ensures that catalyst costs remain negligible compared to precious metal alternatives like palladium or rhodium. The mild reaction conditions also translate to lower energy overheads, as no heating or cooling beyond ambient temperature is required, contributing to a reduced carbon footprint and lower utility costs per kilogram of product manufactured.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by its atom economy and operational simplicity. Since the reaction is highly selective, raw material utilization is maximized, minimizing the waste of expensive alkyne or azide precursors. The absence of high-temperature requirements removes the need for specialized heating infrastructure and reduces the risk of thermal degradation of sensitive functional groups, which can often lead to batch failures in conventional synthesis. Additionally, the straightforward aqueous workup allows for the potential recovery and recycling of solvents, further driving down the variable costs associated with large-scale production runs.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials enhances supply chain resilience. 4-Azidocinnamic acid can be synthesized from cheap and abundant 4-nitrocinnamic acid via reduction and diazotization, ensuring a stable upstream supply. The terminal alkynes required, such as phenylacetylene or simple aliphatic alkynes, are commodity chemicals available from multiple global suppliers, reducing the risk of single-source bottlenecks. The robustness of the reaction conditions means that production is less susceptible to fluctuations in environmental controls or minor variations in reagent quality, ensuring consistent delivery schedules and reliable inventory management for downstream API manufacturers.
- Scalability and Environmental Compliance: Scaling this process from grams to tons is inherently safer and more compliant with modern environmental regulations. The room temperature operation eliminates the hazards associated with exothermic runaways common in thermal azide chemistry. Moreover, the high selectivity reduces the generation of hazardous chemical waste streams, simplifying effluent treatment and disposal. The process aligns well with Green Chemistry principles by utilizing safer solvents (ethyl acetate/water systems) and catalytic reagents, making it easier for manufacturing sites to maintain ISO 14001 compliance and meet the increasingly stringent sustainability goals of multinational pharmaceutical clients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for evaluating the technology's fit within your existing manufacturing portfolio. Understanding these details is crucial for R&D teams planning tech transfers and for procurement teams assessing the long-term viability of this supply route.
Q: What are the primary advantages of this Cu-catalyzed method over traditional thermal cycloaddition?
A: Unlike traditional thermal Huisgen cycloadditions which require high temperatures and produce mixtures of 1,4- and 1,5-regioisomers, this Cu-catalyzed method operates at room temperature and exhibits perfect regioselectivity for the 1,4-disubstituted triazole. This eliminates complex separation processes, significantly reducing solvent consumption and processing time while improving overall yield consistency.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the process is highly amenable to scale-up. The reaction utilizes inexpensive catalysts (CuI) and additives (sodium ascorbate) under mild conditions (room temperature, atmospheric pressure), removing the need for specialized high-pressure or high-temperature reactors. The simple aqueous workup and standard chromatographic purification further facilitate transfer from laboratory to pilot and commercial plant scales.
Q: What represents the key impurity profile concern in this synthesis?
A: The primary advantage of this specific protocol is the suppression of regioisomeric impurities. In non-catalyzed reactions, the formation of the 1,5-isomer is a major purification challenge. Here, the copper catalyst directs the cyclization exclusively to the 1,4-position. The main residual impurities typically stem from unreacted starting materials (azide or alkyne), which are easily removed during the extraction and silica gel purification steps described in the patent examples.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2,3-Triazole Cinnamic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the drug development pipeline. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless. We adhere to stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of 1,2,3-triazole cinnamic acid meets the exacting standards required for pharmaceutical applications. Our commitment to quality assurance ensures that our clients receive materials that are fully characterized and ready for immediate use in subsequent synthetic steps.
We invite you to collaborate with us to optimize your supply chain for these valuable heterocyclic building blocks. Our experts are prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a reliable source of complex intermediates backed by deep chemical expertise and a proven track record of delivering excellence in the fine chemical sector.
