Advanced Manufacturing of 4-Amino-1-piperidinepropanol: A Scalable Route for Pharmaceutical Intermediates
The chemical landscape for functionalized piperidine derivatives is constantly evolving, driven by the dual demands of pharmaceutical innovation and emerging applications in carbon capture technology. Patent CN115677567A introduces a robust and highly efficient synthetic methodology for producing 4-amino-1-piperidinepropanol, a versatile cyclic diamine structure that has garnered significant attention for its potential utility in CO2 capture systems and as a critical building block in medicinal chemistry. This specific patent delineates a streamlined three-step protocol that circumvents the complexities often associated with introducing hydroxyalkyl chains onto heterocyclic cores while simultaneously installing a primary amine functionality. By leveraging a combination of nucleophilic substitution, oxime formation, and subsequent hydride reduction, the disclosed method achieves a remarkable balance between operational simplicity and chemical efficiency. For industrial stakeholders, this represents a pivotal advancement, offering a pathway to secure high-purity intermediates without the burden of excessive purification steps or exotic reagents that typically inflate production costs and extend lead times.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of amino-functionalized piperidines bearing hydroxyalkyl side chains has been plagued by significant challenges regarding regioselectivity and functional group compatibility. Traditional routes often rely on multi-step protection and deprotection strategies to prevent unwanted side reactions between the nucleophilic nitrogen and the electrophilic sites on the alkylating agents, or between the reducing agents and the hydroxyl groups. Furthermore, direct alkylation of piperidones can frequently result in over-alkylation or polymerization if reaction conditions are not meticulously controlled, leading to complex impurity profiles that are difficult to separate. The lack of disclosed, scalable methods in existing literature prior to this invention suggests that previous attempts suffered from low overall yields or required harsh conditions that compromised the integrity of the sensitive amine and alcohol functionalities. These inefficiencies translate directly into higher manufacturing costs and unreliable supply chains for downstream users who require consistent quality for their own synthesis campaigns.
The Novel Approach
In stark contrast to these cumbersome historical precedents, the methodology outlined in CN115677567A employs a direct and atom-economical strategy that maximizes throughput while minimizing waste. The process initiates with a highly efficient N-alkylation of 4-piperidone using 3-chloro-1-propanol, facilitated by cesium carbonate as a base and potassium iodide as a catalyst in 3-pentanone. This specific combination of reagents promotes rapid conversion to 1-(3-hydroxypropyl)-4-piperidone under reflux conditions, effectively installing the necessary carbon backbone in a single operation. 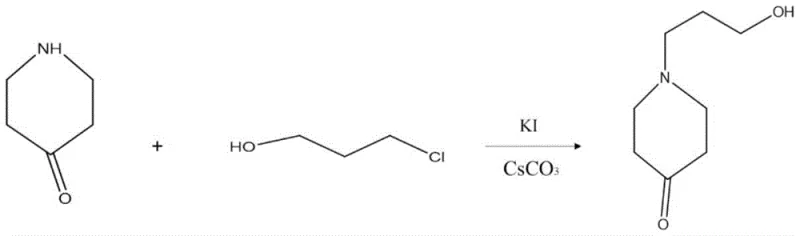 The subsequent transformation involves converting the ketone moiety into an oxime, which serves as a superior precursor for the final reduction step compared to the ketone itself. This strategic choice of intermediate allows for a cleaner reduction profile, ultimately delivering the target 4-amino-1-piperidinepropanol with reported yields ranging from 60% to 70% and purity levels between 85% and 90%. The elimination of protecting groups and the use of common industrial solvents underscore the practical viability of this novel approach for large-scale manufacturing.
The subsequent transformation involves converting the ketone moiety into an oxime, which serves as a superior precursor for the final reduction step compared to the ketone itself. This strategic choice of intermediate allows for a cleaner reduction profile, ultimately delivering the target 4-amino-1-piperidinepropanol with reported yields ranging from 60% to 70% and purity levels between 85% and 90%. The elimination of protecting groups and the use of common industrial solvents underscore the practical viability of this novel approach for large-scale manufacturing.
Mechanistic Insights into LiAlH4-Mediated Oxime Reduction
The cornerstone of this synthetic sequence lies in the final reduction step, where the 1-(3-hydroxypropyl)-4-piperidone oxime is converted into the corresponding amine. This transformation utilizes lithium aluminum hydride (LiAlH4), a potent reducing agent known for its ability to cleave the nitrogen-oxygen bond of oximes effectively. The mechanism proceeds through the coordination of the aluminum species to the oxime nitrogen, followed by hydride transfer which generates an intermediate imine or aluminum-complexed amine species that is subsequently hydrolyzed during the quenching phase. Conducting this reaction in tetrahydrofuran (THF) at low temperatures (0°C to 5°C) is critical for controlling the exothermic nature of the hydride addition and preventing potential side reactions such as the reduction of other sensitive functional groups or the degradation of the piperidine ring. 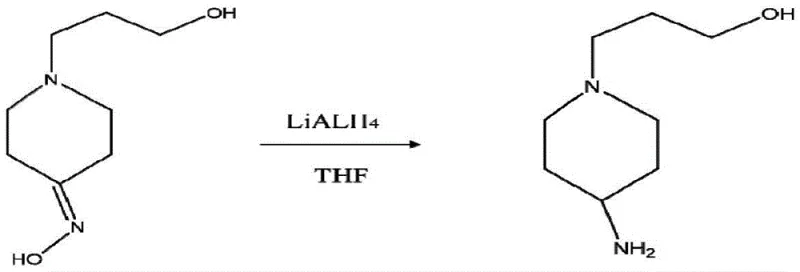 This precise thermal control ensures that the reduction proceeds selectively to the primary amine without affecting the terminal hydroxyl group, a common pitfall in less optimized protocols. The careful quenching procedure involving water and sodium hydroxide further ensures the safe decomposition of excess hydride and the liberation of the free amine product in a form suitable for isolation.
This precise thermal control ensures that the reduction proceeds selectively to the primary amine without affecting the terminal hydroxyl group, a common pitfall in less optimized protocols. The careful quenching procedure involving water and sodium hydroxide further ensures the safe decomposition of excess hydride and the liberation of the free amine product in a form suitable for isolation.
From an impurity control perspective, the choice of the oxime intermediate plays a vital role in defining the final purity profile. Unlike direct reductive amination which can suffer from equilibrium issues and the formation of secondary amines, the oxime route provides a discrete, isolable intermediate that can be purified prior to the final reduction. This two-stage approach (oximation followed by reduction) allows for the removal of unreacted ketone or alkylation by-products before the critical C-N bond formation is finalized. Furthermore, the use of sodium acetate in the oximation step buffers the reaction medium, preventing acid-catalyzed degradation of the piperidine ring and ensuring high conversion rates. The result is a final product with a defined impurity spectrum that is easier to manage and characterize, meeting the stringent requirements of pharmaceutical grade intermediates where trace contaminants can have significant downstream effects.
How to Synthesize 4-Amino-1-piperidinepropanol Efficiently
The synthesis of this valuable piperidine derivative is achieved through a logical sequence of three distinct chemical transformations that are amenable to standard reactor setups found in most fine chemical facilities. The process begins with the alkylation of the piperidone core, followed by functional group interconversion to the oxime, and concludes with a hydride reduction. Each step has been optimized in the patent data to balance reaction kinetics with safety and ease of workup, utilizing solvents like 3-pentanone and ethanol that are both effective and economically viable. The following guide outlines the critical operational parameters derived from the patent examples, providing a roadmap for technical teams to replicate this high-yielding process.
- Perform N-alkylation of 4-piperidone with 3-chloro-1-propanol using cesium carbonate and potassium iodide in 3-pentanone under reflux.
- Convert the resulting 1-(3-hydroxypropyl)-4-piperidone to its oxime derivative using hydroxylamine hydrochloride and sodium acetate in aqueous ethanol.
- Reduce the oxime intermediate to the final amine product using lithium aluminum hydride in tetrahydrofuran at low temperature.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible benefits that extend beyond mere chemical elegance. The reliance on commodity chemicals such as 4-piperidone, 3-chloro-1-propanol, and standard inorganic bases like cesium carbonate significantly mitigates the risk of raw material shortages. Unlike processes that depend on proprietary catalysts or scarce organometallic complexes, this method utilizes reagents that are widely available from multiple global suppliers, ensuring a resilient supply chain that is less susceptible to geopolitical disruptions or single-source bottlenecks. The simplicity of the workup procedures, which involve standard filtration and solvent evaporation techniques, further reduces the operational overhead and equipment requirements, making it an attractive option for contract manufacturing organizations looking to optimize their asset utilization.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the elimination of expensive protecting group chemistry and the use of cost-effective catalysts like potassium iodide. By streamlining the synthesis into just three steps with high overall yields, the consumption of solvents and energy per kilogram of product is drastically reduced compared to longer, more convoluted routes. The avoidance of transition metal catalysts also removes the need for costly and time-consuming heavy metal scavenging steps, which are often required to meet regulatory limits in pharmaceutical applications. This inherent efficiency translates into a lower cost of goods sold, allowing buyers to negotiate more favorable pricing structures while maintaining healthy margins.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes significantly to supply continuity. The use of thermally stable solvents and reagents that tolerate minor variations in temperature or stoichiometry means that batch-to-batch consistency is easier to maintain. This reliability reduces the incidence of failed batches or off-spec material, which are major causes of supply delays in the fine chemical industry. Furthermore, the short reaction times reported in the patent examples indicate a high throughput capability, enabling manufacturers to respond quickly to fluctuations in demand without the need for extensive inventory buffering.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward due to the absence of hazardous high-pressure steps or cryogenic conditions beyond standard cooling. The solvents employed, such as ethanol and 3-pentanone, have well-established recovery and recycling protocols, facilitating compliance with increasingly strict environmental regulations regarding volatile organic compound (VOC) emissions. The generation of inorganic salts as by-products is manageable through standard wastewater treatment processes, minimizing the environmental footprint of the manufacturing site and reducing the costs associated with waste disposal and regulatory reporting.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating the feasibility of integrating this intermediate into their supply chains, we have compiled answers to common inquiries based on the specific technical disclosures in the patent. These responses address critical aspects regarding purity standards, reagent selection, and process safety, providing a clear picture of what to expect when sourcing or manufacturing this compound. Understanding these details is essential for conducting accurate risk assessments and ensuring that the material meets the specific requirements of your downstream applications.
Q: What is the expected purity of 4-amino-1-piperidinepropanol using this method?
A: According to patent CN115677567A, the synthesized product achieves a purity range of 85% to 90% with an overall yield between 60% and 70%.
Q: Which reducing agent is preferred for the final step?
A: Lithium aluminum hydride (LiAlH4) is the preferred catalyst for the reduction of the oxime intermediate, conducted in tetrahydrofuran at 0°C to 5°C.
Q: What solvents are utilized throughout the synthesis process?
A: The process utilizes 3-pentanone for the initial alkylation, 95% aqueous ethanol for oximation, and tetrahydrofuran (THF) for the final reduction step.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Amino-1-piperidinepropanol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your drug development and specialty chemical programs. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to full-scale manufacturing is seamless and efficient. We are committed to delivering 4-amino-1-piperidinepropanol with stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical instrumentation to verify every batch against the highest industry standards. Our dedication to quality assurance means that you can rely on us to provide material that consistently meets the demanding criteria required for pharmaceutical and advanced chemical applications.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs. By leveraging our technical expertise and production capabilities, we can offer a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized synthesis of 4-amino-1-piperidinepropanol can add immediate value to your operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
