Revolutionizing Isoquinoline Production: A Green Photochemical Route for Commercial Scale-Up
Introduction to Advanced Isoquinoline Synthesis Technology
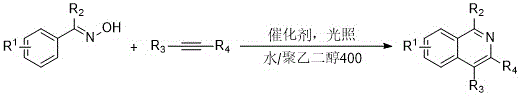
The pharmaceutical and fine chemical industries are currently undergoing a paradigm shift towards sustainable manufacturing processes, driven by both regulatory pressures and the economic necessity of reducing waste. In this context, the technology disclosed in patent CN111039867A represents a significant breakthrough in the synthesis of 3,4-disubstituted isoquinoline derivatives. These heterocyclic compounds are pivotal scaffolds in medicinal chemistry, serving as core structures for a vast array of bioactive molecules and drug candidates. The patent introduces a novel green synthesis method that leverages room-temperature illumination to promote transition metal-catalyzed C-H coupling reactions. By utilizing a benign solvent system comprising water and polyethylene glycol 400, this approach effectively addresses the longstanding challenges of toxicity and high energy consumption associated with traditional isoquinoline production. For R&D directors and procurement strategists, understanding the implications of this technology is crucial for optimizing supply chains and enhancing the sustainability profile of their product portfolios.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the isoquinoline ring system has relied heavily on classical cyclization strategies that often demand harsh reaction conditions and generate substantial chemical waste. Traditional methods frequently necessitate the use of volatile organic compounds (VOCs) as solvents, which pose significant safety hazards and environmental liabilities during large-scale manufacturing. Furthermore, many conventional pathways require the pre-activation of substrates or the use of stoichiometric amounts of hazardous oxidants to drive the reaction to completion. These factors not only inflate the cost of goods sold (COGS) due to expensive reagent consumption but also complicate the downstream purification processes. The generation of toxic byproducts necessitates rigorous waste treatment protocols, adding layers of operational complexity and cost that can erode profit margins in competitive markets. Additionally, the requirement for elevated temperatures or pressures in thermal catalysis increases energy expenditure and limits the compatibility with sensitive functional groups, often leading to lower overall yields and purity issues.
The Novel Approach
In stark contrast to these legacy methods, the innovation described in CN111039867A offers a streamlined, environmentally benign alternative that operates under mild conditions. The core of this advancement lies in the utilization of visible light or UV illumination to drive the catalytic cycle at room temperature, thereby eliminating the need for thermal energy input. The reaction employs a unique solvent system of water and polyethylene glycol 400, which not only enhances the solubility of organic substrates but also ensures the process is non-flammable and low-toxicity. Crucially, this method functions as an oxidative C-H coupling reaction that does not require the addition of external oxidants; instead, the oxygen source is managed internally, resulting in water as the sole byproduct. This dramatic improvement in atom economy means that nearly all atoms in the starting materials are incorporated into the final product, minimizing waste generation. The simplicity of the operational procedure, combined with excellent functional group tolerance, allows for the rapid assembly of diverse isoquinoline libraries, making it an ideal candidate for both drug discovery and commercial manufacturing.
Mechanistic Insights into Photochemical C-H Coupling
The mechanistic foundation of this synthesis relies on a sophisticated interplay between transition metal catalysis and photo-excitation. The reaction initiates with the coordination of the phenyl oxime substrate to the transition metal center, typically a Rhodium, Ruthenium, Cobalt, or Iridium complex. Under illumination, the catalyst-substrate complex absorbs photon energy, facilitating the activation of the inert C-H bond on the aromatic ring through a concerted metalation-deprotonation (CMD) pathway or a radical mechanism depending on the specific metal center employed. This activation step is critical as it bypasses the high energy barriers associated with thermal C-H activation, allowing the reaction to proceed efficiently at ambient temperatures. Once the C-H bond is activated, the non-terminal alkyne inserts into the metal-carbon bond, forming a metallacycle intermediate. Subsequent reductive elimination releases the desired 3,4-disubstituted isoquinoline product and regenerates the active catalytic species. The absence of external oxidants suggests that the catalytic cycle is self-sustaining through the internal redox properties of the oxime directing group, which acts as both a template for regioselectivity and an internal oxidant source.
From an impurity control perspective, the mildness of the reaction conditions plays a pivotal role in ensuring high product purity. Traditional thermal methods often promote side reactions such as polymerization of alkynes or over-oxidation of sensitive functional groups due to the high energy environment. By maintaining the reaction at room temperature, the kinetic energy available for these parasitic pathways is significantly reduced, thereby suppressing the formation of complex impurity profiles. Furthermore, the use of water and PEG 400 as solvents provides a unique polarity environment that can stabilize charged intermediates and suppress non-polar side reactions. The high selectivity observed in the patent examples, with yields consistently reaching high levels across various substrates, indicates a robust mechanism that tolerates electron-donating and electron-withdrawing groups alike. This predictability is essential for pharmaceutical manufacturers who must adhere to strict regulatory guidelines regarding impurity thresholds in active pharmaceutical ingredients (APIs).
How to Synthesize 3,4-Disubstituted Isoquinoline Derivatives Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to the specific parameters outlined in the patent to ensure reproducibility and optimal yield. The process begins with the precise weighing of the phenyl oxime compound and the non-terminal alkyne, which serve as the foundational building blocks for the isoquinoline scaffold. These substrates are then introduced into a clean quartz reactor, a material choice that is critical for maximizing light transmission and ensuring efficient photo-excitation of the catalyst. The addition of the transition metal catalyst, such as pentamethylcyclopentadienyl rhodium acetate or dichloro(pentamethylcyclopentadienyl)rhodium dimer, must be done under controlled conditions to prevent premature deactivation. The solvent system, a 1:1 mixture of water and polyethylene glycol 400, is then added to create a homogeneous or micro-heterogeneous reaction medium that facilitates mass transfer. The detailed standardized synthesis steps for this process are provided in the guide below.
- Combine phenyl oxime compounds, non-terminal alkynes, and a transition metal catalyst (such as Rhodium or Cobalt complexes) in a mixture of water and polyethylene glycol 400 (1: 1 ratio) within a quartz reactor.
- Subject the reaction mixture to illumination using a high-pressure mercury lamp, xenon lamp, or LED source at room temperature for approximately 24 hours to facilitate the C-H coupling reaction.
- Upon completion, extract the product using ethyl acetate and saturated brine, remove the solvent under reduced pressure, and purify the residue via silica gel column chromatography to obtain the target isoquinoline derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this photochemical synthesis method offers tangible strategic benefits that extend beyond mere technical novelty. The shift away from traditional thermal processes and hazardous solvents directly translates into a more resilient and cost-effective supply chain. By eliminating the need for expensive and potentially scarce external oxidants, the raw material costs are inherently lowered, providing a buffer against market volatility in reagent pricing. The use of water and PEG 400, which are commodity chemicals with stable global supply chains, further mitigates the risk of supply disruptions compared to specialized organic solvents. Moreover, the simplified workup procedure, which involves standard extraction and chromatography without the need for complex quenching of hazardous oxidants, reduces the labor hours and equipment downtime associated with batch processing. These operational efficiencies accumulate to provide significant cost savings over the lifecycle of the product, enhancing the overall competitiveness of the manufacturing operation.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound, primarily driven by the elimination of costly reagents and energy inputs. Traditional C-H activation methods often rely on stoichiometric oxidants like silver salts or copper salts, which are not only expensive but also generate heavy metal waste that requires costly disposal. By removing the need for these additives, the direct material costs are drastically reduced. Additionally, the ability to run the reaction at room temperature eliminates the energy costs associated with heating reactors to high temperatures or maintaining cryogenic conditions for sensitive steps. The high atom economy ensures that the maximum amount of raw material is converted into saleable product, minimizing the cost of wasted feedstock. These factors combined result in a leaner manufacturing process with a lower break-even point, allowing for more aggressive pricing strategies in the marketplace.
- Enhanced Supply Chain Reliability: Supply chain continuity is paramount for pharmaceutical manufacturers, and this method enhances reliability through the use of robust and readily available starting materials. Phenyl oximes and non-terminal alkynes are commercially accessible building blocks with well-established production networks, reducing the dependency on custom-synthesized precursors that may have long lead times. The tolerance of the reaction to various functional groups means that a wider range of substrate suppliers can be qualified, diversifying the supply base and reducing single-source risks. Furthermore, the stability of the catalyst systems described allows for potential recycling or extended usage, decreasing the frequency of catalyst replenishment orders. This stability ensures that production schedules can be maintained consistently without unexpected delays caused by reagent shortages or quality variations in sensitive materials.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges, particularly regarding heat management and waste treatment, but this photochemical approach is inherently scalable due to its mild conditions. The absence of exothermic runaway risks associated with strong oxidants makes the scale-up safer and more predictable, facilitating the transition from gram-scale discovery to multi-ton commercial production. From an environmental compliance standpoint, the use of green solvents and the generation of water as the only byproduct significantly simplify the permitting process and reduce the burden on wastewater treatment facilities. This alignment with green chemistry principles not only future-proofs the manufacturing site against tightening environmental regulations but also enhances the corporate sustainability profile, which is increasingly valued by investors and customers alike.
Frequently Asked Questions (FAQ)
The following questions address common inquiries regarding the technical feasibility and commercial viability of this synthesis method, based on the detailed disclosures within the patent documentation. Understanding these aspects is vital for stakeholders evaluating the integration of this technology into their existing manufacturing frameworks. The answers provided reflect the specific advantages and operational parameters defined in the intellectual property, ensuring accuracy and relevance for technical decision-makers.
Q: What are the primary environmental advantages of this isoquinoline synthesis method?
A: This method utilizes water and polyethylene glycol 400 as green solvents instead of toxic organic solvents. Furthermore, the reaction proceeds at room temperature under illumination without the need for additional external oxidants, producing only water as a byproduct, which significantly improves atom economy and reduces waste disposal costs.
Q: Which catalysts are compatible with this photochemical C-H coupling reaction?
A: The patent discloses a broad scope of transition metal catalysts including various Rhodium complexes (e.g., pentamethylcyclopentadienyl rhodium acetate), Ruthenium complexes, Cobalt complexes, and Iridium complexes, allowing for flexibility in catalyst selection based on substrate specificity and cost considerations.
Q: Does this method require harsh reaction conditions or substrate pre-activation?
A: No, one of the key innovations is that the reaction occurs at room temperature under illumination, eliminating the need for high heat or pressure. Additionally, it does not require pre-activation of the substrates, simplifying the operational procedure and reducing energy consumption compared to traditional thermal methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4-Disubstituted Isoquinoline Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the green synthesis technologies described in CN111039867A and are fully equipped to leverage them for your project needs. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory concept to market-ready product is seamless. Our state-of-the-art facilities are designed to handle complex photochemical reactions with precision, supported by stringent purity specifications and rigorous QC labs that guarantee the highest quality standards for every batch. We understand that consistency and reliability are the cornerstones of a successful supply chain, and our dedicated team is committed to delivering products that meet your exact requirements.
We invite you to collaborate with us to explore how this innovative route can optimize your specific isoquinoline derivative projects. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating exactly how this green method can improve your bottom line. We encourage you to reach out today to discuss your specific COA data requirements and to receive comprehensive route feasibility assessments that will empower your R&D and supply chain strategies. Let us be your partner in driving efficiency and sustainability in your chemical manufacturing operations.
